Abstract
Purpose
Phosphatidylinositol-3 kinases (PI3Ks) are involved in regulating cell growth, proliferation, differentiation, apoptosis and survival. p110α and p110β, two ubiquitously expressed isoforms of PI3K signalling, are involved in growth factor mediated signaling and survival by generating second messengers. Earlier, we have generated GFP-fusion proteins of p110α and p110β and expressed them in normal and cancer cell-lines to investigate their subcellular localization and their role in various activities. Here, we sought to examine the role of p110α and p110β isoforms in protecting MCF-7 breast cancer cells against oxidative stress.
Material methods
We performed cytotoxicity assays, DNA transfection, Plasmid DNA preparation, western blotting, flourscence microscopy and statistical analysis.
Results
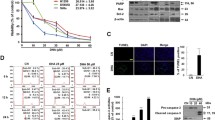
To know whether p110α and p110β are involved in protecting MCF-7 breast cancer cells against oxidative stress, we subjected MCF-7 cells to H2O2 treatment and observed a dose dependent decrease in cell viability and a marked increase in the levels of pro-apoptotic markers which include PARP, Bcl-2, Bax and procaspase-9. We then over-expressed recombinant GFP-fusion p110α and p110β proteins in MCF-7 cells and observed a significant decrease in apoptosis and a concomitant increase in pAkt levels.
Conclusion
We report the involvement of p110α and p110β isoforms of Class 1A PI3K signalling in rescue from oxidative stress-induced apoptosis in MCF-7 cells in Akt dependent manner.





Similar content being viewed by others
References
Domin J, Waterfield M. Using structure to define the function of phosphoinositide 3-kinase family members. FEBS Lett. 1997;410:91–5.
Engelman JA, Luo J, Cantley LC. The evolution of phosphatidylinositol 3-kinases as regulators of growth and metabolism. Nat Rev Genet. 2006;7:606–19.
Vanhaesebroeck B, Waterfield M. Signaling by distinct classes of phosphoinositide 3-kinases. Exp Cell Res. 1999;253:239–54.
Liu P, Cheng H, Roberts TM, Zhao JJ. Targeting the phosphoinositide 3-kinase pathway in cancer. Nat Rev Drug Discov. 2009;8:627–44.
Vanhaesebroeck B, Guillermet-Guibert J, Graupera M, Bilanges B. The emerging mechanisms of isoform-specific PI3K signalling. Nat Rev Mol Cell Biol. 2010;11:329–41.
Alessi DR, James SR, Downes CP, Holmes AB, Gaffney PR, Reese CB, et al. Characterization of a 3-phosphoinositide-dependent protein kinase which phosphorylates and activates protein kinase Bα. Curr Biol. 1997;7:261–9.
Sarbassov DD, Guertin DA, Ali SM, Sabatini DM. Phosphorylation and regulation of Akt/PKB by the rictor-mTOR complex. Science. 2005;307:1098–101.
Stephens L, Anderson K, Stokoe D, Erdjument-Bromage H, Painter GF, Holmes AB, et al. Protein kinase B kinases that mediate phosphatidylinositol 3, 4, 5-trisphosphate-dependent activation of protein kinase B. Science. 1998;279:710–4.
Vivanco I, Sawyers CL. The phosphatidylinositol 3-kinase–AKT pathway in human cancer. Nat Rev Cancer. 2002;2:489–501.
Yamaguchi H, Wang H-G. The protein kinase PKB/Akt regulates cell survival and apoptosis by inhibiting Bax conformational change. Oncogene. 2001;20:7779–86.
Zhang X, Tang N, Hadden TJ, Rishi AK. Akt. FoxO and regulation of apoptosis. Biochim Biophys Acta (BBA) Mol Cell Res. 2011;1813:1978–86.
Cantley LC. The phosphoinositide 3-kinase pathway. Science. 2002;296:1655–7.
Singh P, Dar MS, Dar MJ. p110α and p110β isoforms of PI3K signaling: are they two sides of the same coin? FEBS Letters. 2016.
Singh P, Dar MS, Singh G, Jamwal G, Sharma PR, Ahmad M, et al. Dynamics of GFP-Fusion p110α and p110β isoforms of PI3K signaling pathway in normal and cancer cells. J Cell Biochem. 2016;117:2864–74.
Kumar A, Redondo-Muñoz J, Perez-García V, Cortes I, Chagoyen M, Carrera AC. Nuclear but not cytosolic phosphoinositide 3-kinase beta has an essential function in cell survival. Mol Cell Biol. 2011;31:2122–33.
Shayesteh L, Lu Y, Kuo W-L, Baldocchi R, Godfrey T, Collins C, et al. PIK3CA is implicated as an oncogene in ovarian cancer. Nat Genet. 1999;21:99–102.
Levine DA, Bogomolniy F, Yee CJ, Lash A, Barakat RR, Borgen PI, et al. Frequent mutation of the PIK3CA gene in ovarian and breast cancers. Clin Cancer Res. 2005;11:2875–8.
Velho S, Oliveira C, Ferreira A, Ferreira AC, Suriano G, Schwartz S, et al. The prevalence of PIK3CA mutations in gastric and colon cancer. Eur J Cancer. 2005;41:1649–54.
Campbell IG, Russell SE, Choong DY, Montgomery KG, Ciavarella ML, Hooi CS, et al. Mutation of the PIK3CA gene in ovarian and breast cancer. Cancer Res. 2004;64:7678–81.
Zhao L, Vogt PK. Helical domain and kinase domain mutations in p110α of phosphatidylinositol 3-kinase induce gain of function by different mechanisms. Proc Natl Acad Sci. 2008;105:2652–7.
Thorpe LM, Yuzugullu H, Zhao JJ. PI3K in cancer: divergent roles of isoforms, modes of activation and therapeutic targeting. Nat Rev Cancer. 2015;15:7–24.
Samuels Y, Ericson K. Oncogenic PI3K and its role in cancer. Curr Opin Oncol. 2006;18:77–82.
Carracedo A, Pandolfi P. The PTEN–PI3K pathway: of feedbacks and cross-talks. Oncogene. 2008;27:5527–41.
Samuels Y, Wang Z, Bardelli A, Silliman N, Ptak J, Szabo S, et al. High frequency of mutations of the PIK3CA gene in human cancers. Science. 2004;304:554.
Yuan T, Cantley L. PI3K pathway alterations in cancer: variations on a theme. Oncogene. 2008;27:5497–510.
Kaufmann SH, Desnoyers S, Ottaviano Y, Davidson NE, Poirier GG. Specific proteolytic cleavage of poly (ADP-ribose) polymerase: an early marker of chemotherapy-induced apoptosis. Cancer Res. 1993;53:3976–85.
Tewari M, Quan LT, O’Rourke K, Desnoyers S, Zeng Z, Beidler DR, et al. Yama/CPP32β, a mammalian homolog of CED-3, is a CrmA-inhibitable protease that cleaves the death substrate poly (ADP-ribose) polymerase. Cell. 1995;81:801–9.
Zhou H, Li X-M, Meinkoth J, Pittman RN. Akt regulates cell survival and apoptosis at a postmitochondrial level. J Cell Biol. 2000;151:483–94.
Matheny RW, Adamo ML. PI3K p110α and p110β have differential effects on Akt activation and protection against oxidative stress-induced apoptosis in myoblasts. Cell Death Differ. 2010;17:677–88.
Acknowledgements
This work was supported by a grant from the Council of Scientific and Industrial Research (CSIR), Government of India, New Delhi, under HCP0008. This manuscript represents CSIR-IIIM communication number (IIIM/2255/2018).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
About this article
Cite this article
Singh, P., Bano, N., Hossain, M.M. et al. p110α and p110β isoforms of PI3K are involved in protection against H2O2 induced oxidative stress in cancer cells. Breast Cancer 26, 378–385 (2019). https://doi.org/10.1007/s12282-018-0933-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12282-018-0933-x