Abstract
Background
This study aimed to assess their characteristics and clinical course of each histologic type of the behavior of phyllodes tumor (PT).
Methods
We retrospectively reviewed 124 patients with PTs who underwent surgical treatment from 2003 to 2011. PTs were classified as benign, borderline, and malignant using surgical specimens. The clinicopathological characteristics according to solitary and multiple lesions on ipsilateral breast and histological change after local recurrence were examined.
Results
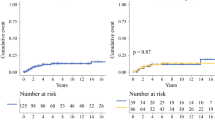
The median patient age was 42 years (range 12–72 years). The final pathologic diagnosis was benign PTs in 95 patients (76.6%), borderline PTs in 21 (16.9%), and malignant PTs in 8 (6.5%). The size of benign PTs [median 4.2 cm (range 1–21 cm)] was significantly smaller than that of malignant PTs [median 11.3 cm (range 6–27 cm)] (p < 0.001). Eight of the 95 benign PTs (8.4%) had multiple lesions, while all malignant PTs had solitary lesion. With a median follow-up period of 45 months, five patients with malignant PTs (62.5%) developed distant metastases while 13 patients with benign PTs (13.5%) and 6 patients with borderline PTs (28.6%) had local recurrence only without malignant transition. The median overall survival period of those with malignant PTs was 22.5 months (range 5–109 months).
Conclusions
Histologically benign and borderline PTs had a good prognosis without malignant transition even after developing repeat recurrence in contrast with malignant PTs that might be a solitary lesion.

Similar content being viewed by others
References
Parham KS. Management of non-metastatic phyllodes tumors of the breast: review of the literature. Surg Oncol. 2011;20:143–8. https://doi.org/10.1016/j.suronc.2011.04.007.
Tan PH, Tse GM, Lee A, et al. Fibroepithelial tumours. WHO classification of tumours of the breast. International agency for research on cancer. Lyon. 2012;4:142–7.
Salm R. Multifocal histogenesis of a cystosarcoma phyllodes. J Clin Patho. 1978;31:897–903.
Tan PH, Thike AA, Tan WJ, et al. Predicting clinical behavior of breast phyllodes tumours: a nomogram based on histological criteria and surgical margins. J Clin Pathol. 2012;65:69–76. https://doi.org/10.1136/jclinpath-2011-200368. (Epub 2011 Nov 2).
Chen WH, Cheng SP, Tzen CY, et al. Surgical treatment of phyllodes tumors of the breast: retrospective review of 172 cases. J Surg Oncol. 2005;91:185–94. https://doi.org/10.1002/jso.20334.
Sotheran W, Domjan J, Jeffrey M, et al. Phyllodes tumours of the breast—a retrospective study from 1982 to 2000 of 50 cases in Portsmouth. Ann R Coll Surg Engl. 2005;87:339–44. https://doi.org/10.1308/003588405X51128.
Abdalla HM, Sakr MA. Predictive factors of local recurrence and survival following primary surgical treatment of phyllodes tumors of the breast. J Egypt Natl Canc Inst. 2006;18:125–33.
Hassouna JB, Damak T, Gamoudi A, et al. Phyllodes tumors of the breast: a case series of 106 patients. Am J Surg. 2006;192:141–7. https://doi.org/10.1016/j.amjsurg.2006.04.007.
Granić M, Oprić D, Pupić G, et al. Surgical methods for the treatment of breast phyllodes tumors—a report of 319 cases. Acta Chir Iugosl. 2006;53:57–62.
Taira N, Takabatake D, Aogi K, et al. Phyllodes tumor of the breast: stromal overgrowth and histological classification are useful prognosis-predictive factors for local recurrence in patients with a positive surgical margin. Jpn J Clin Oncol. 2007;7:730–6. https://doi.org/10.1093/jjco/hym099.
Lenhard MS, Kahlert S, Himsl I, et al. Phyllodes tumour of the breast: clinical follow-up of 33 cases of this rare disease. Eur J Obstet Gynecol Reprod Biol. 2007;138:217–21. https://doi.org/10.1016/j.ejogrb.2007.08.002.
Bhargav PR, Mishra A, Agarwal G, Agarwal A, et al. Phyllodes tumour of the breast: clinicopathological analysis of recurrent vs. non-recurrent cases. Asian J Surg. 2009;32:224–8. https://doi.org/10.1016/S1015-9584(09)60398-5.
Jang JH, Choi MY, Lee SK, et al. Clinicopathologic risk factors for the local recurrence of phyllodes tumors of the breast. Ann Surg Oncol. 2012;19:2612–7. https://doi.org/10.1245/s10434-012-2307-5.
Sawalhi S, Al-Shatti M. Phyllodes tumor of the breast: a retrospective study of the impact of histopathological factors in local recurrence and distant metastasis. Ann Saudi Med. 2013;33:162–8. https://doi.org/10.5144/0256-4947.
Fou A, Schnabel FR, Hamele-Bena D, et al. Long-term outcomes of malignant phyllodes tumors patients: an institutional experience. Am J Surg. 2006;192:492–5. https://doi.org/10.1016/j.amjsurg.2006.06.017.
Barrio AV, Clark BD, Goldberg JI, et al. Clinicopathologic features and long-term outcomes of 293 phyllodes tumors of the breast. Ann Surg Oncol. 2007;14:2961–70. https://doi.org/10.1245/s10434-007-9439-z.
Abe M, Miyata S, Nishimura S, et al. Malignant transformation of breast fibroadenoma to malignant phyllodes tumor: long-term outcome of 36 malignant phyllodes tumors. Breast Cancer. 2011;18:268–72. https://doi.org/10.1007/s12282-009-0185-x.
Tan EY, Tan PH, Yong WS, et al. Recurrent phyllodes tumours of the breast: pathological features and clinical implications. ANZ J Surg. 2006;7:476–80. https://doi.org/10.1111/j.1445-2197.2006.03754.x.
Hajdu SI, Espinosa MH, Robbins GF. Recurrent cystosarcoma phyllodes: a clinicopathologic study of 32 cases. Cancer. 1978;38:1402–6.
Vidal M, Peg V, Galván P, et al. Gene expression based classifications of fibroadenomas and phyllodes tumours of the breast. Mol Oncol. 2015;9:1081–90. https://doi.org/10.1016/j.molonc.2015.01.003.
Guillot E, Couturaud B, Reyal F, et al. Management of phyllodes breast tumors. Breast J. 2011;17:129–37. https://doi.org/10.1111/j.1524-4741.2010.01045.x.
Treves N. A study of cystosarcoma phyllodes. Ann N Y Acad Sci. 1964;114:922–36.
Zurrida S, Bartoli C, Galimberti V, et al. Which therapy for unexpected phyllodes tumour of the breast? Eur J Cancer. 1992;28:654–7.
Rowe JJ, Prayson RA. Metastatic malignant phyllodes tumor involving the cerebellum. J Clin Neurosci. 2015;22:226–7. https://doi.org/10.1016/j.jocn.2014.09.004.
Kessinger A, Foley JF, Lemon HM, et al. Metastatic cystosarcoma phyllodes: a case report and review of the literature. J Surg Oncol. 1972;4:131–47.
Garg N, Moorthy N, Agrawal SK, et al. Delayed cardiac metastasis from phyllodes breast tumor presenting as cardiogenic shock. Tex Heart Inst J. 2011;38:441–4.
Yoshidaya F, Hayashi N, Takahashi K, et al. Malignant phyllodes tumor metastasized to the right ventricle: a case report. Surg Case Rep. 2015;1:121–6.
Telli ML, Horst KC, Guardino AE, et al. Phyllodes tumors of the breast: natural history, diagnosis, and treatment. J Natl Compr Cancer Netw. 2007;5:324–30.
Andi KC, Daniel HH, Andrew S, et al. Next-gen sequencing exposes frequent MED12 mutations and actionable therapeutic targets in phyllodes tumors. Mol Cancer Res. 2015;13:613–9. https://doi.org/10.1158/1541-7786.MCR-14-0578.
Su YL, Nancy MJ, Ajay R, et al. Genomic profiling of malignant phyllodes tumors reveals aberrations in FGFR1 and PI-3 kinase/RAS signaling pathways and provides insights into intratumoral heterogeneity. Mod Pathol. 2016;29:1012–27. https://doi.org/10.1038/modpathol.2016.97.
Desnedt C, Fumagalli D, Pietri E, et al. Uncovering the genomic heterogeneity of multifocal breast cancer. J Pathol. 2015;236:457–66. https://doi.org/10.1002/path.4540.
Acknowledgements
We thank all staff from breast center and departments of Pathology and Radiology, St. Luke’s International hospital, Tokyo, Japan for providing support to this study.
Funding
The authors have no fundings for this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
Ethical approval is in accordance with the Declaration of Helsinki.
Conflict of interest
The authors have declared no conflicts of interest.
About this article
Cite this article
Wada, A., Hayashi, N., Endo, F. et al. Repeat recurrence and malignant transition of phyllodes tumors of the breast. Breast Cancer 25, 736–741 (2018). https://doi.org/10.1007/s12282-018-0882-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12282-018-0882-4