Abstract
Purpose
To establish an optimal surveillance schedule after surgery for breast cancer, patients included in an institutional database were retrospectively investigated with respect to the first metastatic site and timing of recurrence.
Patients and methods
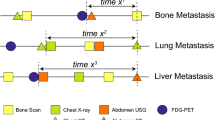
We investigated 11,676 pT1-4pN0-2M0 breast cancer patients treated from 1985 to 2009 and followed up until June 2014. Our surveillance protocol included physician visits and examinations with bone scans, liver echography, chest roentgenography and laboratory tests. We evaluated the liver, bones, lungs and pleura as surveillance covering sites (SCS) in addition to parameters such as time points exceeding 80 % with respect to the accumulated percentage of patients of recurrence and the number of surveillance per one recurrence (NSR), calculated by dividing the number of patients at risk of recurrence at the start of a particular time frame by the number of patients of recurrence at SCS within that period.
Results
There were a total of 1,962 recurrent patients, including 601 patients with locoregional recurrence, nine patients with recurrence in the opposite breast, 1,349 patients with recurrence at distant sites and three unknown patients. The number of patients with the bones, lungs, liver and pleura as the first site of recurrence was 447, 324, 144 and 69, respectively, and 72.9 % of the distant metastatic lesions belonged to SCS. The five-year overall survival rate after recurrence among the patients with single recurrent site was longer than that observed among the patients with multiple sites of recurrence (43.3 vs 25.3 %; p < 0.0001). In addition, more than 80 % of the patients of liver metastasis were detected within 5 years after surgery, while 80 % of the patients of pleura metastasis were detected within 10 years. The NSR was below 200 for the 10-year period, as was the NSR of the patients with lymph node metastasis and a positive hormone receptor status. In contrast, the NSR of the patients with a negative hormone receptor status was above 200 after 5 years.
Conclusions
In this study, the prognosis of the patients with a single site of recurrence was superior to that of the patients with multiple sites. Curable patients with distant metastases included those with single metastatic sites. The optimal surveillance schedule should be established taking into consideration that the incidence of metastasis differs among metastatic sites during follow-up.



Similar content being viewed by others
References
American Society of Clinical Oncology, Breast cancer surveillance expert panel of the American Society of Clinical Oncology. Recommended breast cancer surveillance guidelines. J Clin Oncol. 1997;15(5):2149–56.
Rutgers EJ, van Slooten EA, Kluck HM. Follow-up after treatment of primary breast cancer. Br J Surg. 1989;76(2):187–90.
Pivot X, Asmar L, Hortobagyi GN, Theriault R, Pastorini F, Buzdar A. A retrospective study of first indicators of breast cancer recurrence. Oncology. 2000;58(3):185–90.
Del Turco MR, Palli D, Cariddi A, Ciatto S, Pacini P, Distante V. Intensive diagnostic follow-up after treatment of primary breast cancer: a randomized trial. JAMA. 1994;271(20):1593–7.
Khatcheressian JL, Hurley P, Bantug E, Esserman LJ, Grunfeld E, Halberg F, et al. Breast cancer follow-up and management after primary treatment: American Society of Clinical Oncology clinical practice guideline update. J Clin Oncol. 2013;31(7):961–5.
Dawood S, Broglio K, Buzdar AU, Hortobagyi GN, Giordano SH. Prognosis of women with metastatic breast cancer by HER2 status and trastuzumab treatment: an institutional-based review. J Clin Oncol. 2010;28(1):92–8.
Andre F, Slimane K, Bachelot T, Dunant A, Namer M, Barrelier A, et al. Breast cancer with synchronous metastases: trends in survival during a 14-year period. J Clin Oncol. 2004;22(16):3302–8.
Iwata H. Future treatment strategies for metastatic breast cancer: curable or incurable? Breast Cancer. 2012;19(3):200–5.
Kasumi F, Takahashi K, Nishimura S, Iijima K, Miyagi U, Tada K, et al CIH-Tokyo Experience with Breast-Conserving Surgery without Radiotherapy: 6.5 Year Follow-Up Results of 1462 Patients. Breast J. 12suppl2:s181–s190. 2006.
Makita M, Iwase T, Tada T, Takahashi K, Tada K, Nishimura S, et al. The site and timing of the first recurrence of breast cancer. Jpn J Breast Cancer. 2004;19(4):343–51 in Japanese.
Japanese Breast Cancer Society. The general rules for clinical and pathological recording of breast cancer. Breast Cancer. 2005;12:s1–14.
Corbin KS, Hellman S, Weichselbaum RR. Extracranial oligometastases: a subset of metastases curable with stereotactic radiotherapy. J Clin Oncol. 2013;31(11):1384–90.
Niibe Y, Hayakawa K. Oligometastases and oligo-recurrence: the new era of cancer therapy. Jpn J Clin Oncol. 2010;40:107–11.
Saphner T, Tormey DC, Gray R. Annual hazard rates of recurrence for breast cancer after primary therapy. J Clin Oncol. 1996;14:2738–46.
Insa A, Lluch A, Prosper F, Marugan I, Martinez-Agullo A, Garcia-Conde J. Prognostic factors predicting survival from first recurrence in patients with metastatic breast cancer: analysis of 439 patients. Breast Cancer Res Treat. 1999;56(1):67–78.
Tsuji W, Teramukai S, Ueno M, Toi M, Inamoto T. Prognostic factors for survival after first recurrence in breast cancer: a retrospective analysis of 252 recurrent cases at a single institution. Breast Cancer. 2014;21(1):86–95.
Kim H, Choi DH, Park W, Huh SJ, Nam SJ, Lee JE, et al. Prognostic factors for survivals from first relapse in breast cancer patients: analysis of deceased patients. Radiat Oncol J. 2013;31(4):222–7.
Yoshimoto M, Tada T, Saito M, Takahashi K, Uchida Y, Kasumi F. Surgical treatment of hepatic metastases from breast cancer. Breast Cancer Res Treat. 2000;59(2):177–84.
Iwase T, Nakagawa K, Tsuchiya S, Okumura S, Sato Y, Kasumi F, et al. Long-term results of the resected cases with pulmonary metastasis from breast cancer. Jpn J Breast Cancer. 1994;9(1):4–12 in Japanese.
Andrews DW, Scott CB, Sperduto PW, Flanders AE, Gaspar LE, Schell MC, et al. Whole brain radiation therapy with or without stereotactic radiosurgery boost for patients with one to three brain metastases: phase III results of the RTOG 9508 randomised trial. Lancet. 2004;363(22):1665–72.
Andre F, Cabioglu N, Assi H, Sabourin JC, Delaloge S, Sahin A. et al Expression of chemokine receptors predicts the site of metastatic relapse in patients with axillary node positive primary breast cancer. Ann Oncol. 2006;17(6):945–51.
Kennecke H, Yerushalmi R, Woods R, Cheang MC, Voduc D, Speers CH, et al. Metastatic behavior of breast cancer subtypes. J Clin Oncol. 2010;28(20):3271–7.
Pennant M, Takwoingi Y, Pennant L, Davenport C, Fry-Smith A, Eisinga A, et al. A systematic review of positron emission tomography (PET) and positron emission tomography/computed tomography (PET/CT) for the diagnosis of breast cancer recurrence. Health Technol Assess. 2010;14(50):1–103.
Acknowledgments
The authors thank Miss Rie Gokan for her valuable help administrating our institutional database.
Conflict of interest
No authors have any conflicts of interest to declare in association with this study.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Makita, M., Sakai, T., Ogiya, A. et al. Optimal surveillance for postoperative metastasis in breast cancer patients. Breast Cancer 23, 286–294 (2016). https://doi.org/10.1007/s12282-014-0571-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12282-014-0571-x