Abstract
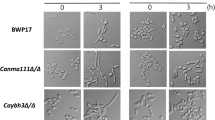
Candida albicans is an opportunistic human pathogen that exists as yeast, hyphal or pseudohyphal forms depending on pH, nutrients, and temperature. The morphological transition from yeast to hyphae, which is required for the complete virulence of C. albicans, is controlled by many transcription factors that activate or repress hypha-specific genes. The C. albicans transcriptional factor Cas5, a key regulator of genes involved in cell wall integrity, affects the susceptibility of C. albicans to fluconazole, an inhibitor of ergosterol synthesis. In this study, we found that deletion of CAS5 in C. albicans decreased the expression levels of a set of ergosterol biosynthesis genes, such as ERG2, ERG3, ERG5, ERG6, ERG11, and ERG24, resulting in the accumulation of lanosterol and zymosterol, which are intermediate metabolites in the ergosterol biosynthesis pathway. Interestingly, it was observed that the cas5Δ/Δ mutant could not maintain the yeast form under non-hypha-inducing conditions, while the CAS5-overexpressing cells could not form hyphae under hypha-inducing conditions. Consistent with these observations, the cas5Δ/Δ mutant highly expressed hypha-specific genes, ALS3, ECE1, and HWP1, under non-hypha-inducing conditions. In addition, CAS5 transcription was significantly downregulated immediately after hyphal initiation in the wild-type strain. Furthermore, the cas5Δ/Δ mutant reduced the transcription of NRG1, which encodes a major repressor of hyphal morphogenesis, while Cas5 overexpression increased the transcription of NRG1 under hypha-inducing conditions. Collectively, this study suggests the potential role of Cas5 as a repressor of hypha-specific genes during yeast-form growth of C. albicans.
Similar content being viewed by others
Change history
01 November 2021
An Erratum to this paper has been published: https://doi.org/10.1007/s12275-021-0326-2
References
Arthington-Skaggs, B.A., Jradi, H., Desai, T., and Morrison, C.J. 1999. Quantitation of ergosterol content: novel method for determination of fluconazole susceptibility of Candida albicans. J. Clin. Microbiol.37, 3332–3337.
Bhattacharya, S., Sae-Tia, S., and Fries, B.C. 2020. Candidiasis and mechanisms of antifungal resistance. Antibiotics9, 312.
Braun, B.R., Kadosh, D., and Johnson, A.D. 2001. NRG1, a repressor of filamentous growth in C. albicans, is down-regulated during filament induction. EMBO J.20, 4753–4761.
Bruno, V.M., Kalachikov, S., Subaran, R., Nobile, C.J., Kyratsous, C., and Mitchell, A.P. 2006. Control of the C. albicans cell wall damage response by transcriptional regulator Cas5. PLoS Pathog.2, e21.
Calderone, R.A. and Fonzi, W.A. 2001. Virulence factors of Candida albicans. Trends Microbiol.9, 327–335.
Care, R.S., Trevethick, J., Binley, K.M., and Sudbery, P.E. 1999. The MET3 promoter: a new tool for Candida albicans molecular genetics. Mol. Microbiol.34, 792–798.
Chamilos, G., Nobile, C.J., Bruno, V.M., Lewis, R.E., Mitchell, A.P., and Kontoyiannis, D.P. 2009. Candida albicans Cas5, a regulator of cell wall integrity, is required for virulence in murine and Toll mutant fly models. J. Infect. Dis.200, 152–157.
Fonzi, W.A. and Irwin, M.Y. 1993. Isogenic strain construction and gene mapping in Candida albicans. Genetics134, 717–728.
Ghannoum, M.A. and Rice, L.B. 1999. Antifungal agents: mode of action, mechanisms of resistance, and correlation of these mechanisms with bacterial resistance. Clin. Microbiol. Rev.12, 501–517.
Gow, N.A.R., Brown, A.J.P., and Odds, F.C. 2002. Fungal morphogenesis and host invasion. Curr. Opin. Microbiol.5, 366–371.
Gow, N.A., van de Veerdonk, F.L., Brown, A.J., and Netea, M.G. 2012. Candida albicans morphogenesis and host defence: discriminating invasion from colonization. Nat. Rev. Microbiol.10, 112–122.
Kadosh, D. and Johnson, A.D. 2005. Induction of the Candida albicans filamentous growth program by relief of transcriptional repression: a genome-wide analysis. Mol. Biol. Cell.16, 2903–2912.
Kim, J.Y. 2016. Human fungal pathogens: why should we learn? J. Microbiol.54, 145–148.
Kruppa, M. 2009. Quorum sensing and Candida albicans. Mycoses52, 1–10.
LaFayette, S.L., Collins, C., Zaas, A.K., Schell, W.A., Betancourt-Quiroz, M., Gunatilaka, A.A.L., Perfect, J.R., and Cowen, L.E. 2010. PKC signaling regulates drug resistance of the fungal pathogen Candida albicans via circuitry comprised of Mkc1, calcineurin, and Hsp90. PLoS Pathog.6, e1001069.
Lee, H.J., Kim, J.M., Kang, W.K., Yang, H., and Kim, J.Y. 2015. The NDR kinase Cbk1 downregulates the transcriptional repressor Nrg1 through the mRNA-binding protein Ssd1 in Candida albicans. Eukaryot. Cell14, 671–683.
Lu, Y., Su, C., Unoje, O., and Liu, H. 2014. Quorum sensing controls hyphal initiation in Candida albicans through Ubr1-mediated protein degradation. Proc. Natl. Acad. Sci. USA111, 1975–1980.
Lu, Y., Su, C., Wang, A., and Liu, H. 2011. Hyphal development in Candida albicans requires two temporally linked changes in promoter chromatin for initiation and maintenance. PLoS Biol.9, e1001105.
Martin, S.W. and Konopka, J.B. 2004. Lipid raft polarization contributes to hyphal growth in Candida albicans. Eukaryot. Cell3, 675–684.
Mazu, T.K., Bricker, B.A., Flores-Rozas, H., and Ablordeppey, S.Y. 2016. The mechanistic targets of antifungal agents: an overview. Mini Rev. Med. Chem.16, 555–578.
Murad, A.M., Leng, P., Straffon, M., Wishart, J., Macaskill, S., MacCallum, D., Schnell, N., Talibi, D., Marechal, D., Tekaia, F., et al. 2001. NRG1 represses yeast-hypha morphogenesis and hyphaspecific gene expression in Candida albicans. EMBO J.20, 4742–4752.
Pukkila-Worley, R., Peleg, A.Y., Tampakakis, E., and Mylonakis, E. 2009. Candida albicans hyphal formation and virulence assessed using a Caenorhabditis elegans infection model. Eukaryot. Cell8, 1750–1758.
Sudbery, P.E. 2011. Growth of Candida albicans hyphae. Nat. Rev. Microbiol.9, 737–748.
Vasicek, E.M., Berkow, E.L., Bruno, V.M., Mitchell, A.P., Wiederhold, N.P., Barker, K.S., and Rogers, P.D. 2014. Disruption of the transcriptional regulator Cas5 results in enhanced killing of Candida albicans by Fluconazole. Antimicrob. Agents Chemother.58, 6807–6818.
Walther, A. and Wendland, J. 2003. An improved transformation protocol for the human fungal pathogen Candida albicans. Curr. Genet.42, 339–343.
Xie, J.L., Qin, L., Miao, Z., Grys, B.T., Diaz, J.C., Ting, K., Krieger, J.R., Tong, J., Tan, K., Leach, M.D., et al. 2017. The Candida albicans transcription factor Cas5 couples stress responses, drug resistance and cell cycle regulation. Nat. Commun.8, 499.
Xiong, K., Su, C., Sun, Q., and Lu, Y. 2021. Efg1 and Cas5 orchestrate cell wall damage response to caspofungin in Candida albicans. Antimicrob. Agents Chemother.65, e01584–20.
Acknowledgements
This research was supported by the National Research Foundation of Korea, Grant No. NRF 2020R1A2C101025412 to J.-Y. Kim and Grant No. NRF2018R1A5A1025077 (Advanced Research Center Program) to H.A. Kang.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors have no conflict of interest to report.
Additional information
Supplemental material for this article may be found at http://www.springerlink.com/content/120956.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Kim, JM., Moon, H.Y., Lee, D.W. et al. The transcription factor Cas5 suppresses hyphal morphogenesis during yeast-form growth in Candida albicans. J Microbiol. 59, 911–919 (2021). https://doi.org/10.1007/s12275-021-1326-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12275-021-1326-y