Abstract
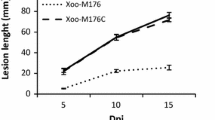
To identify novel virulence genes, a mutant library of Xanthomonas citri subsp. citri 29-1 was produced using EZ-Tn5 transposon and the mutants were inoculated into susceptible grapefruit. Forty mutants with altered virulence phenotypes were identified. Nine of the mutants showed a complete loss of citrus canker induction, and the other 31 mutants resulted in attenuated canker symptoms. Southern blot analysis revealed that each of the mutants carried a single copy of Tn5. The flanking sequence was identified by plasmid rescue and 18 different ORFs were identified in the genome sequence. Of these 18 ORFs, seven had not been previously associated with the virulence of X. citri subsp. citri and were therefore confirmed by complementation analysis. Real-time PCR analysis showed that the seven genes were upregulated when the bacteria were grown in citrus plants, suggesting that the expression of these genes was essential for canker development.
Similar content being viewed by others
References
Atkinson, M.R. and Ninfa, A.J. 1998. Role of the GlnK signal transduction protein in the regulation of nitrogen assimilation in Escherichia coli. Mol. Microbiol. 29, 431–447.
Alegria, M.C., Docena, C., Khater, L., Ramos, C.H., da Silva, A.C., and Farah, C.S. 2004. New protein-protein interactions identified for the regulatory and structural components and substrates of the type III secretion system of the phytopathogen Xanthomonas axonopodis pathovar citri. J. Bacteriol. 186, 6186–6197.
AI-Saadi, A., Reddy, J.D., Duan, Y.P., Brunings, A.M., Yuan, Q., and Gabriel, D.W. 2007. All five host-range variants of Xanthomonas citri carry one pthA homolog with 17.5 repeats that determines pathogenicity on citrus, but none determine host-range variation. Mol. Plant Microbe Interact. 20, 934–943.
Baptista, J.C., Machado, M.A., Homem, R.A., Torres, P.S., Vojnov, A.A., and do Amaral, A.M. 2010. Mutation in the xpsD gene of Xanthomonas axonopodis pv. citri affects cellulose degradation and virulence. Genet. Mol. Biol. 33, 146–153.
Brunings, A.M. and Gabriel, D.W. 2003. Xanthomonas citri: breaking the surface. Mol. Plant Pathol. 4, 141–157.
Cui, Q. and Karplus, M. 2001. Triosephosphate isomerase: a theoretical comparison of alternative pathways. J. Am. Chem. Soc. 123, 2284–2290.
Del Sorbo, G., Schoonbeek, H., and De Waard, M.A. 2000. Fungaltransporters involved in efflux of natural toxic compounds and fungicides. Fungal Genet. Biol. 30, 1–15.
Diamant, S. and Goloubinoff, P. 1998. Temperature-controlled activity of DnaK-DnaJ-GrpE chaperones: protein-folding arrest and recovery during and after heat shock depends on the substrate protein and the GrpE concentration. Biochemistry 37, 9688–9694.
Dunger, G., Arabolaza, A.L., Gottig, N., Orellano, E.G., and Ottado, J. 2005. Participation of Xanthomonas axonopodis pv. citri hrp cluster in citrus canker and non host plant responses. Plant Pathol. 54, 781–788.
Dunger, G., Garofalo, C.G., Gottig, N., Garavaglia, B.S., Rosa, M.C.P., Farah, C.S., Orellano, E.G., and Ottado, J. 2012. Analysis of three Xanthomonas axonopodis pv. citri effector proteins in pathogenicity and their interactions with host plant proteins. Mol. Plant Pathol. 13, 865–876.
Galan, J.E. and Collmer, A. 1999. Type III secretion machines: bacterial devices for protein delivery into host cells. Science 284, 1322–1328.
Gottig, N., Garavaglia, B.S., Garofalo, C.G., Orellano, E.G., and Ottado, J.A. 2009. Filamentous hemagglutinin-like protein of Xanthomonas axonopodis pv. citri, the phytopathogen responsible for citrus canker, is involved in bacterial virulence. PLoS One 4, e4358.
Gottwald, T.R., Hughes, G., Graham, J.H., Sun, X., and Riley, T. 2001. The citrus canker epidemic in Florida: The scientific basis of regulatory eradication policy for an invasive species. Phytopathology 91, 30–34.
Knowles, J.R. 1991. Enzyme catalysis: not different, just better. Nature 350, 121–124.
Krysan, P.J., Young, J.C., and Sussman, M.R. 1999. T-DNA as an insertional mutagen in Arabidopsis. Plant Cell 11, 2283–2290.
Laia, M.L., Moreira, L.M., Dezajacomo, J., Brigati, J.B., Ferreira, C.B., Ferro, M.I.T., Silva, A.C.R., Ferro, J.A., and Oliveira, J.C.F. 2009. New genes of Xanthomonas citri subsp. citri involved in pathogenesis and adaptation revealed by a transposon-based mutant library. BMC Microbiol. 9, 12.
Li, J.Y. and Wang, N. 2011. Genome-wide mutagenesis of Xanthomonas axonopodis pv. citri reveals novel genetic determinants and regulation mechanisms of biofilm formation. PLoS One 6, e21804.
Malamud, F., Conforte, V.P., Rigano, L.A., Castagnaro, A.P., Marano, M.R., Morais do Amaral, A., and Vojnov, A.A. 2012. HrpM is involved in glucan biosynthesis, biofilm formation and pathogenicity in Xanthomonas citri subsp. citri. Mol. Plant. Pathol. 13, 1010–1018.
Mitchell, P. 1967. Translocations through natural membranes. Adv. Enzymol. 29, 33–87.
Pao, S.S., Paulsen, I.T., and Saier, M.H.Jr. 1998. Major facilitator superfamily. Microbiol. Mol. Biol. Rev. 62, 1–34.
Perales-Calvo, J., Muga, A., and Moro, F. 2010. Role of DnaJ G/Frich domain in conformational recognition and binding of protein substrates. J. Biol. Chem. 285, 34231–3439.
Pruvost, O., Boher, C., Brocherieux, C., Nicole, M., and Chiroleu, F. 2002. Survival of Xanthomonas axonopodis pv. citri in leaf lesions under subtropical environmental conditions and simulated splash dispersal of inoculum. Phytopathology 92, 336–346.
Qian, W., Jia, Y., Ren, S., He, Y., Feng, J., Lu, L., Sun, Q., Ying, G., Tang, D., and Tang, H. 2005. Comparative and functional genomic analyses of the pathogenicity of phytopathogen Xanthomonas campestris pv. campestris. Genome Res. 15, 757–767.
Rigano, L.A., Siciliano, F., Enrique, R., Sendín, L., Filippone, P., Torres, P.S., Qüesta, J., Dow, J.M., Castagnaro, and other authors. 2007. Biofilm formation, epiphytic fitness, and canker development in Xanthomonas axonopodis pv. citri. Mol. Plant Microbe Interact. 20, 1222–1230.
Sgro, G.G., Ficarra, F.A., Dunger, G., Scarpec, T.E., Valle, E.M., Cortadi, A., Orellano, E.G., Gottig, N., and Ottado, J. 2012. Contribution of a harpin protein from Xanthomonas axonopodis pv. citri to pathogen virulence. Mol. Plant Pathol. 13, 1047–1059.
Shiotani, H., Fujikawa, T., Ishihara, H., Tsuyumu, S., and Ozaki, K.A. 2007. pthA homolog from Xanthomonas axonopodis pv. citri responsible for host-specific suppression of virulence. J. Bacteriol. 189, 3271–3279.
da Silva, A.C., Ferro, J.A., Reinach, F.C., Farah, C.S., Furlan, L.R., Quaggio, R.B., Monteiro-Vitorello, C.B., Van Sluys, M.A., Almeida, N.F., and other authors. 2002. Comparison of the genomes of two Xanthomonas pathogens with differing host specificities. Nature 417, 459–463.
de Souza, T.A., Soprano, A.S., de Lira, N.P., Quaresma, A.J., Pauletti, B.A., Paes Leme, A.F., and Benedetti, C.E. 2012. The TAL effector PthA4 interacts with nuclear factors involved in RNA-dependent processes including a HMG protein that selectively binds poly (U) RNA. PLoS One 7, e32305.
Stall, R.E. and Civerolo, E.L. 1991. Research relating to the recent outbreak of citrus canker in Florida. Ann. Rev. Phytopathol. 29, 399–420.
Stock, A.M., Robinson, V.L., and Goudreau, P.N. 2000. Two-component signal transduction. Annu. Rev. Biochem. 69, 183–215.
Tang, D.J., He, Y.Q., Feng, J.X., He, B.R., Jiang, B.L., Lu, G.T., Chen, B., and Tang, J.L. 2005. Xanthomonas campestris pv. campestris possesses a single gluconeogenic pathway that is required for virulence. J. Bacteriol. 187, 6231–6237.
Wang, L., Makino, S., Subedee, A., and Bogdanove, A.J. 2007. Novel candidate virulence factors in rice pathogen Xanthomonas oryzae pv. oryzicola as revealed by mutational analysis. Appl. Environ. Microbiol. 73, 8023–8027.
Wang, J., So, B., Kim, J., Park, Y., Lee, B., and Kang, H. 2008. Genome- wide identification of pathogenicity genes in Xanthomonas oryzae pv. oryzae by transposon mutagenesis. Plant Pathol. 57, 1136–1145.
Warnecke, T. 2012. Loss of the DnaK-DnaJ-GrpE chaperone system among the Aquificales. Mol. Biol. Evol. 29, 3485–3495.
Yan, Q. and Wang, N. 2011. The ColR/ColS two-component system plays multiple roles in the pathogenicity of the citrus canker pathogen Xanthomonas citri subsp. citri. J. Bacteriol. 193, 1590–1599.
Yan, Q. and Wang, N. 2012. High-throughput screening and analysis of genes of Xanthomonas citri subsp. citri involved in citrus canker symptom development. Mol. Plant Microbe Interact. 25, 69–84.
Yang, W., Liu, Y., Chen, L., Qian, G., Liu, H., Hu, B., and Liu, F. 2008. Involvement of gluconeogenic pathway in virulence of Xanthomonas oryzae pv. oryzae. J. Phytopathol. 156, 174–180.
Ye, G., Hong, N., Zou, L.F., Zou, H.S., Zakria, M., Wang, G.P., and Chen, G.Y. 2013. Tale-based genetic diversity of chinese isolates of the citrus canker pathogen Xanthomonas citri subsp. citri. Plant Dis. 97, 1187–1194.
Yurgel, S.N., Rice, J., Mulder, M., and Kahn, M.L. 2010. GlnB/GlnK PII proteins and regulation of the Sinorhizobium meliloti Rm1021 nitrogen stress response and symbiotic function. J. Bacteriol. 192, 2473–2481.
Zou, H.S., Song, X., Zou, L.F., Yuan, L., Li, Y.R., Guo, W., Che, Y.Z., Zhao, W.X., Chen, G.Y., and Duan, Y.P. 2012. EcpA, an extracellular protease, is a specific virulence factor required by Xanthomonas oryzae pv. oryzicola but not by X. oryzae pv. oryzae in rice. Microbiology 158, 2372–2383.
Zou, H.S., Yuan, L., Guo, W., Li, Y.R., Che, Y.Z., Zou, L.F., and Chen, G.Y. 2011. Construction of a Tn5-tagged mutant library of Xanthomonas oryzae pv. oryzicola as an invaluable resource for functional genomics. Curr. Microbiol. 2, 908–916.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supplemental material for this article may be found at http://www.springerlink.com/content/120956.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Song, X., Guo, J., Ma, Wx. et al. Identification of seven novel virulence genes from Xanthomonas citri subsp. citri by Tn5-based random mutagenesis. J Microbiol. 53, 330–336 (2015). https://doi.org/10.1007/s12275-015-4589-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12275-015-4589-3