Abstract
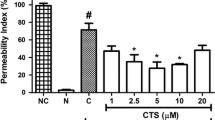
Over the past 100 years, Salvia miltiorrhiza f. alba (Lamiaceae) (RSMA) roots have been used to cure thromboangiitis obliterans (TAO) in local clinics. This study aimed to confirm the anti-thrombotic efficacy of 12 phenolic acids obtained from RSMA and to clarify the possible underlying mechanisms. The results of quantitative real-time polymerase chain reaction (qRT-PCR) and enzyme-linked immunosorbent assay (ELISA) experiments demonstrated that most of the phenolic acids markedly inhibited PAI-1 protein and mRNA levels but increased t-PA protein and mRNA levels in TNF-α-induced EA.hy926 cells (P < 0.05 or 0.001), with lithospermic acid displaying the strongest effect. In vitro anticoagulation and antiplatelet aggregation assays showed that lithospermic acid and salvianolic acid B significantly prolonged prothrombin time (PT), activated partial thromboplastin time (APTT), decreased fibrinogen concentration (FIB), and inhibited platelet aggregation induced by adenosine diphosphate (ADP) in rat blood. Both lithospermic acid and salvianolic acid B markedly down-regulated the expression of factor Xa and factor IIa on the external surface of EA.hy926 cells and demonstrated significant anti-factor IIa and anti-factor Xa activity using chromogenic substrates in vitro. Western blot results revealed that both lithospermic acid and salvianolic acid B also significantly inhibited the expression of TF, p-p65, p-p38, and pJNK proteins induced by TNF-α. These results indicated that all of the phenolic acids appeared to have some anti-thrombotic activity, with salvianolic acid B and lithospermic acid markedly decreasing the chance of thrombosis by regulating the NF-κB/JNK/p38 MAPK signaling pathway in response to TNF-α.





Similar content being viewed by others
References
Amiral J, Laroche M, Seghatchian J (2018) A new assay for global fibrinolysis capacity (GFC): investigating a critical system regulating hemostasis and thrombosis and other extravascular functions. Transfus Apher Sci 57(1):118–126. https://doi.org/10.1016/j.transci.2018.02.020
Breitenstein A, Camici GG, Tanner FC (2009) Tissue factor: beyond coagulation in the cardiovascular system. Clin Sci 118(3–4):159–172. https://doi.org/10.1042/CS20080622
Edén D, Panagiotou G, Mokhtari D, Eriksson JW, Åberg M, Siegbahn A (2019) Adipocytes express tissue factor and FVII and are procoagulant in a TF/FVIIa-dependent manner. Ups J Med Sci 124(3):158–167. https://doi.org/10.1080/03009734.2019.1645248
Esmon CT (2004) Crosstalk between inflammation and thrombosis. Maturitas 61(1–2):122–131
Fan HY, Fu FH, Yang MY, Xu H, Zhang AH, Liu K (2010) Antiplatelet and antithrombotic activities of salvianolic acid A. Thromb Res 126(1):E17–E22. https://doi.org/10.1016/j.thromres.2010.04.006
Fanjul-Fernández M, Folgueras AR, Cabrera S, López-Otín C (2010) Matrix metalloproteinases: evolution, gene regulation and functional analysis in mouse models. Biochim Biophys Acta 1803(1):3–19. https://doi.org/10.1016/j.bbamcr.2009.07.004
Fei YX, Wang SQ, Yang LJ, Qiu YY, Li YZ, Liu WY, Xi T, Fang WR, Li YM (2017) Salvia miltiorrhiza Bunge (Danshen) extract attenuates permanent cerebral ischemia through inhibiting platelet activation in rats. J Ethnopharmacol 207:57–66. https://doi.org/10.1016/j.jep.2017.06.023
Hanslik A, Kitzmüller EE, Tran US, Thom K, Karapetian H, Prutsch N, Voitl J, Michel-Behnke I, Newall F, Male C (2017) Anti-activated factor II assay for monitoring unfractionated heparin in children: results of the HEARTCAT study. J Thromb Haemost 15(1):38–46. https://doi.org/10.1111/jth.13539
Hsu T, Young MR, Cmarik J, Colburn NH (2000) Activator protein 1 (AP-1) and nuclear factor κB (NF-κB)–dependent transcriptional events in carcinogenesis. Free Radic Biol Med 28(9):1338–1348. https://doi.org/10.1016/S0891-5849(00)00220-3
Huebner BR, Moore EE, Moore HB, Stettler GR, Nunns GR, Lawson P, Sauaia A, Kelher M, Banerjee A, Silliman CC (2018) Thrombin provokes degranulation of platelet α-granules leading to the release of active plasminogen activator inhibitor-1 (PAI-1). Shock 50(6):671–676. https://doi.org/10.1097/SHK.0000000000001089
Kadri OE, Surblyte M, Chandran VD, Voronov RS (2019) Is the endothelial cell responsible for the thrombus core and shell architecture? Med Hypotheses 129:109–244. https://doi.org/10.1016/j.mehy.2019.109244
Koupenova M, Clancy L, Corkrey HA, Freedman JE (2018) Circulating platelets as mediators of immunity, inflammation, and thrombosis. Circ Res 122(2):337–351. https://doi.org/10.1161/CIRCRESAHA.117.310795
Kovács B, Bereczky Z, Oláh Z, Gindele R, Kerényi A, Selmeczi A, Boda Z, Muszbek L (2013) The superiority of anti-FXa assay over anti-FIIa assay in detecting heparin-binding site antithrombin deficiency. Am J Clin Pathol 140(5):675–679. https://doi.org/10.1309/AJCPVY4Z9XZMFOTH
Ku SK, Yoon EK, Lee W, Kwon S, Lee T, Bae JS (2016) Antithrombotic and antiplatelet activities of pelargonidin in vivo and in vitro. Arch Pharm Res 39(3):398–408. https://doi.org/10.1007/s12272-016-0708-x
Lee W, Lee J, Kulkarni R, Kim M, Hwang JS, Na M, Bae J (2016) Antithrombotic and antiplatelet activities of small-molecule alkaloids from Scolopendra subspinipes mutilans. Sci Rep 6:21956. https://doi.org/10.1038/srep21956
Liu HM, Ma SL, Xia HR, Lou HX, Zhu FL, Sun LR (2018) Anti-inflammatory activities and potential mechanisms of phenolic acids isolated from Salvia miltiorrhiza f. alba roots in THP-1 macrophages. J Ethnopharmacol 222:201–207. https://doi.org/10.1016/j.jep.2018.05.008
Lopes-Bezerra LM, Filler SG (2003) Endothelial cells, tissue factor and infectious diseases. Braz J Med Biol Res 36(8):987–991. https://doi.org/10.1590/S0100-879X2003000800004
Nar H, Bauer M, Schmid A, Stassen J, Wienen W, Priepke HWM, Kauffmann IK, Ries UJ, Hauel NH (2001) Structural basis for inhibition promiscuity of dual specific thrombin and factor Xa blood coagulation inhibitors. Structure 9(1):29–37. https://doi.org/10.1016/S0969-2126(00)00551-7
Obermayer G, Afonyushkin T, Binder CJ (2018) Oxidized low-density lipoprotein in inflammation-driven thrombosis. J Thromb Haemost 16(3):418–428. https://doi.org/10.1111/jth.13925
Pergola GD, Mitrio VD, Giorgino F, Sciaraffia M, Minenna A, Bari LD, Pannacciulli N, Giorgino R (1997) Increase in both pro-thrombotic and anti-thrombotic factors in obese premenopausal women: relationship with body fat distribution. Int J Obes 21(7):527–535. https://doi.org/10.1038/sj.ijo.0800435
Prabhakara N, Smyth SS (2013) Inflammation and thrombosis in cardiovascular disease. Curr Opin Hematol 20(5):457–463. https://doi.org/10.1097/MOH.0b013e328364219d
Rauch U, Osende JI, Fuster V, Badimon JJ, Fayad Z, Chesebro JH (2001) Thrombus formation on atherosclerotic plaques: pathogenesis and clinical consequences. Ann Intern Med 134(3):224. https://doi.org/10.7326/0003-4819-134-3-200102060-00014
Sae-Kwang Ku, Lee I-C, Bae J-S (2014) Antithrombotic activities of oroxylin A in vitro and in vivo. Arch Pharm Res 37(5):679–686. https://doi.org/10.1007/s12272-013-0233-0
Sioulis A, Malindretos P, Makedou A, Makris P, Grekas D (2009) Coagulation factors as biological risk markers of endothelial dysfunction. Association with the thrombotic episodes of chronic hemodialysis patients. Hippokratia 13(4):237–241. https://doi.org/10.1093/fampra/cmp051
Tsantarliotou MP, Lavrentiadou SN, Psalla DA, Margaritis IE, Kritsepi MG, Zervos IA, Latsari MI, Sapanidou VG, Taitzoglou IA, Sinakos ZM (2019) Suppression of plasminogen activator inhibitor-1 (PAI-1) activity by crocin ameliorates lipopolysaccharide-induced thrombosis in rats. Food Chem Toxicol 125:190–197. https://doi.org/10.1016/j.fct.2019.01.001
Ulfhammer E, Larsson P, Karlsson L, Hrafnkelsdóttir T, Bokarewa M, Tarkowski A, Jern S (2006) TNF-alpha mediated suppression of tissue type plasminogen activator expression in vascular endothelial cells is NF-kappa B- and p38 MAPK-dependent. J Thromb Haemost 4(8):1781–1789. https://doi.org/10.1111/j.1538-7836.2006.02035.x
Ward AO, Caputo M, Angelini GD, George SJ, Zakkar M (2017) Activation and inflammation of the venous endothelium in vein graft disease. Atherosclerosis 265:266–274. https://doi.org/10.1016/j.atherosclerosis.2017.08.023
Winckers K, Cate HT, Hacken TM (2013) The role of tissue factor pathway inhibitor in atherosclerosis and arterial thrombosis. Blood Rev 27(3):119–132. https://doi.org/10.1016/j.blre.2013.03.001
Xue X, Jiang LS, Duenninger E, Muenzel M, Guan S, Fazakas A, Cheng FZ, Illnitzky J, Keil T, Yu JT (2018) Impact of chronic kidney disease on Watchman implantation: experience with 300 consecutive left atrial appendage closures at a single center. Heart Vessels 33(9):1068–1075. https://doi.org/10.1007/s00380-018-1157-x
Zhang Y, Zhu HL, Sun LR (2017) Phenolic acids from the roots of Salvia miltiorrhiza var alba. Chin Med Mater 40(4):855–858. https://doi.org/10.13863/j.issn1001-4454.2017.04.021
Zhang ZR, Ji JB, Zhang DW, Ma MQ, Sun LR (2020) Protective effects and potential mechanism of salvianolic acid B on sodium laurate-induced thromboangiitis obliterans in rats. Phytomedicine. https://doi.org/10.1016/j.phymed.2019.153110s
Zhou Z, Liu Y, Miao AD, Wang SQ (2005) Salvianolic acid B attenuates plasminogen activator inhibitor type 1 production in TNF-alpha treated human umbilical vein endothelial cells. J Cell Biochem 96(1):109–116. https://doi.org/10.1002/jcb.20567
Zhu HL, Zhang DW, Sun LR, Ji JB, Wu K (2012) Effects of total salvianolic acid from Salvia miltiorrhiza f. alba on thromboangiitis obliterans in rats. Chin Tradit Herbal Drugs 43(8):1565–1569
Acknowledgements
Natural Science Foundation of Shandong Province (No. ZR2019MH088) provided assistance in the research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No conflict of interests should be declared by the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zheng, X., Liu, H., Ma, M. et al. Anti-thrombotic activity of phenolic acids obtained from Salvia miltiorrhiza f. alba in TNF-α-stimulated endothelial cells via the NF-κB/JNK/p38 MAPK signaling pathway. Arch. Pharm. Res. 44, 427–438 (2021). https://doi.org/10.1007/s12272-021-01325-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-021-01325-7