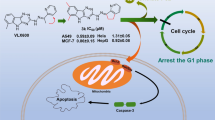
Abstract
A new series of bis(aryl or aralkyl) diselenides 5a–5q was synthesized by selenylation from aryl halide (or aralkyl halide) for development of new anticancer agents. The process involves the reaction of aryl halides (or aralkyl halides) with selenium, hydrazine hydrate under atmospheric pressure in the presence of sodium hydroxide, to afford diorganodiselenides. These new compounds showed antiproliferative activities against breast cancer (MCF-7) cells in CCK-8 assays, and could be promising candidates for chemotherapy of carcinomas. Among 17 synthesized compounds for inhibiting the growth of these cell lines, 1,2-bis(chloropyridazinyl) diselenide 5a showed the highest potency. This result suggests the potential anticancer activity of compound 5a.
Graphical Abstract






Similar content being viewed by others
References
Bhasin, K.K., and J. Singh. 2002. A novel and convenient synthesis towards 2-pyridylselenium compounds: X-ray crystal structure of 4,4′-dimethyl-2, 2′-dipyridyl diselenide and tris (2-pyridylseleno) methane. Journal of Organometallic Chemistry 658: 71–76.
Bhasin, K.K., B.S. Bhandal, J. Singh, N. Singh, K.N. Singh, and P. Singh. 2002. A new and convenient route to 2,2′-dipyridyl diselenide/ditelluride and some of their alkyl/aryl derivatives through BF3-complexed pyridyl carbanion. Synthetic Communications 32(9): 1319–1325.
Bhasin, K.K., N. Singh, R. Kumar, D.G. Deepali, S.K. Mehta, T.M. Klapoetke, and M.J. Crawford. 2004. A convenient synthesis of some symmetrical and unsymmetrical diarylmethyl sulfur and selenium compounds: X-ray crystal structure of diphenylmethylseleno-2-propene and bis[p-chlorophenyl (phenyl)methyl] diselenide. Journal of Organometallic Chemistry 689: 3327–3334.
Bortolatto, C.F., C.R. Jesse, E.A. Wilhelm, L.R. Ribeiro, L.M. Rambo, L.F.F. Royes, S.S. Roman, and C.W. Nogueira. 2011. protective effect of 2,2′-dithienyl diselenide on kainic acid-induced neurotoxicity in rat hippocampus. Neuroscience 193: 300–309.
Brunton, L.L., B.A. Chabner, and B.C. Knollmann. 2011. Goodman & Gilman’s the pharmacological basis of therapeutics, 12th ed, 1694–1698. New York: McGraw-Hill.
Chen, Y., F. Tian, M. Song, and S. Lu. 2007. Improved method for the synthesis of organic diselenides from organic halides under atmospheric pressure. Synthetic Communications 37: 2687–2692.
Glaser, V., B. Moritz, A. Schmits, A.L. Dafré, E.M. Nazari, Y.M.R. Müller, L. Feksa, M.R. Straliottoa, A.F. de Bem, M. Farina, J.B.T. da Rocha, and A. Latini. 2013. Protective effects of diphenyl diselenide in a mouse model of brain toxicity. Chemico-Biological Interactions 206: 18–26.
Jung, J.H., E.J. Kim, H.K. Chung, H.J. Park, S.Y. Jeong, and E.K. Choi. 2012. c-Myc down-regulation is involved in proteasome inhibitor-mediated enhancement of radiotherapeutic efficacy in non-small cell lung cancer. International Journal of Oncology 40: 385–390.
Kim, C., S.B. Kim, and M.S. Park. 2014. Synthesis of novel 3-allylseleno-6-alkylthiopyridazines: Their anticancer activity against MCF-7 cells. Archives of Pharmacal Research 37: 452–458.
Lee, S.H., and M.S. Park. 2011. New organoselenium compounds: synthesis of potential anticancer alkylselenoallylthiopyridazine derivatives. Yakhak Hoeji 55: 32–38.
Lee, S.H., S.B. Kim, and M.S. Park. 2011. One-pot syntheric method of symmetrical diaryl diselenides using Na2Se2. Journal of the Korean Chemical Society 55: 546–549.
Maciel, E.N., E.M.M. Flores, J.B.T. Rocha, and V. Folmer. 2003. Comparative deposition of diphenyl diselenide in liver, kidney, and brain of mice. Bulletin of Environmental Contamination and Toxicology 70: 470–476.
Melo, M.T., I.M. de Oliveira, I. Grivicich, T.N. Guecheva, J. Saffi, J.A.P. Henriques, and R.M. Rosa. 2013. Diphenyl diselenide protects cultured MCF-7 cells against tamoxifen-induced oxidative DNA damage. Biomedicine & Pharmacotherapy 67: 329–335.
Ming-De, R., Z. Hua-Rong, F. Wei-Qiang, and Z. Xun-Jun. 1995. First example of selenium transfer reaction of primary selenoamides and selenourea. Novel synthesis of dialkyl diselenides from alkyl halides. Journal of Organometallic Chemistry 485: 19–24.
Müller, J., and A. Terfort. 2006. Synthesis of pure aromatic, aliphatic, and araliphatic diselenides. Inorganica Chimica Acta 359: 4821–4827.
Mugesh, G., W.W. Mont, and H. Sies. 2001. Chemistry of biologically important synthetic organoselenium compounds. Chemical Review 101: 2125–2179.
Nedel, F., V.F. Campos, D. Alves, A.J.A. Mcbride, O.A. Dellagostin, T. Collares, L. Savegnago, and F.K. Seixas. 2012. Substituted diaryl diselenides: Cytotoxic and apoptotic effect in human colon adenocarcinoma cells. Life Sciences 91: 345–352.
Nogueira, C.W., and J.B.T. Rocha. 2011. Toxicology and pharmacology of selenium: Emphasis on synthetic organoselenium compounds. Archives of Toxicology 85: 1313–1359.
Park, E.H., and M.S. Park. 2007. Synthesis of potential anticancer 6-allylthio-3-aminopyridazine derivatives. Journal of the Korean Chemical Society 51(3): 244–250.
Posser, T., M.T. de Paula, J.L. Franco, R.B. Leal, and J.B.T. da Rocha. 2011. Diphenyl diselenide induces apoptotic cell death and modulates ERK1/2 phosphorylation in human neuroblastoma SH-SY5Y cells. Archives of Toxicology 85: 645–651.
Rosa, R.M., D.J. Moura, A.C.R. Silva, J. Saffi, and J.A.P. Henriques. 2007. Antioxidant activity of diphenyl diselenide prevents the genotoxicity of several mutagens in Chinese hamster V79 cells. Mutation Research 631: 44–54.
Scianowski, J., Rafinski, Z., and Wojtczak, A. 2006. Synthesis and reactions of new optically active terpene dialkyl diselenides. European Journal of Organic Chemistry 2006(14): 3216–3225.
Acknowledgments
This research was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT and Future Planning (No. 2012050022). We would like to acknowledge Professor Joohee Jung (College of Pharmacy, Duksung Women’s University) for the CCK-8 assays of anti-proliferative activities.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, C., Lee, J. & Park, MS. Synthesis of new diorganodiselenides from organic halides: their antiproliferative effects against human breast cancer MCF-7 cells. Arch. Pharm. Res. 38, 659–665 (2015). https://doi.org/10.1007/s12272-014-0407-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-014-0407-4