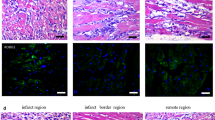
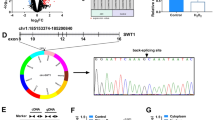
Abstract
Myocardial ischemia/reperfusion (MI/R) injury induces myocardial damage and dysfunction. Increasing evidence has confirmed that circular RNAs (circRNAs) play crucial roles in regulating MI/R. Mmu-circ-0001380 has identified to be highly expressed in myocardium of MI/R mouse model. However, its biological function and molecular mechanism in MI/R injury are still unclear. Here, we demonstrated that knockdown of cric-0001380 attenuated myocardial injury of MI/R mice. In vitro, silence of circ-0001380 significantly enhanced viability, and inhibited apoptosis and oxidative stress in HL-1 cells under oxygen–glucose deprivation/reoxygenation (OGD/R). Mmu-miR-106b-5p interacted with circ-0001380, and suppressed the expression of pleckstrin homology domain and leucine rich repeat protein phosphatase 2 (Phlpp2). The miR-106b-5p/Phlpp2 axis mediated the effect of circ-0001380 on OGD/R-induced apoptosis through regulating the phosphorylation of p38, and further involved in regulating the viability and oxidative stress of HL-1 cells. In conclusion, circ-0001380 downregulation relieves MI/R injury via regulating the miR-106b-5p/Phlpp2 axis.
Graphical Abstract
The present study indicates that mmu-circ-0001380 exacerbates the myocardial ischemia/reperfusion injury through modulating the miR-106b-5p/Phlpp2 axis in vitro and in vivo.







Similar content being viewed by others
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Abbreviations
- MI/R:
-
Myocardial ischemia/reperfusion
- circRNA:
-
Circular RNA
- OGD/R:
-
Oxygen-glucose deprivation/reoxygenation
- AMI:
-
Acute myocardial infarction
- ROS:
-
Reactive oxygen species
- ceRNA:
-
Competitive endogenous RNAs
- Phlpp2:
-
PH domain and leucine rich repeat protein phosphatase 2
- MDA:
-
Malonaldehyde
- SOD:
-
Superoxide dismutase
- GSH-Px:
-
Glutathione peroxidase
- CK-MB:
-
Creatine kinase myocardial band
- LDH:
-
Lactate dehydrogenase
- I/R:
-
Ischemia/reperfusion
- LVEDD:
-
Left ventricular end-diastolic dimension
- LVESD:
-
Left ventricular end systolic diameter
- LVEF:
-
Left ventricular ejection fraction
- LVFS:
-
Left ventricular fraction shortening
- cDNA:
-
Complementary DNA
- gDNA:
-
Genomic DNA
References
Davidson SM, Ferdinandy P, Andreadou I, Bøtker HE, Heusch G, Ibáñez B, et al. Multitarget strategies to reduce myocardial ischemia/reperfusion injury: JACC review topic of the week. J Am Coll Cardiol. 2019;73(1):89–99. https://doi.org/10.1016/j.jacc.2018.09.086.
Dong Y, Chen H, Gao J, Liu Y, Li J, Wang J. Molecular machinery and interplay of apoptosis and autophagy in coronary heart disease. J Mol Cell Cardiol. 2019;136:27–41. https://doi.org/10.1016/j.yjmcc.2019.09.001.
Zong L, Wang W. CircANXA2 promotes myocardial apoptosis in myocardial ischemia-reperfusion injury via inhibiting miRNA-133 expression. Biomed Res Int. 2020;2020:8590861. https://doi.org/10.1155/2020/8590861.
Li Y, Liang P, Jiang B, Tang Y, Lv Q, Hao H, et al. CARD9 inhibits mitochondria-dependent apoptosis of cardiomyocytes under oxidative stress via interacting with Apaf-1. Free Radical Biol Med. 2019;141:172–81. https://doi.org/10.1016/j.freeradbiomed.2019.06.017.
Memczak S, Jens M, Elefsinioti A, Torti F, Krueger J, Rybak A, et al. Circular RNAs are a large class of animal RNAs with regulatory potency. Nature. 2013;495(7441):333–8. https://doi.org/10.1038/nature11928.
Dragomir M, Calin GA. Circular RNAs in Cancer - Lessons Learned From microRNAs. Front Oncol. 2018;8:179. https://doi.org/10.3389/fonc.2018.00179.
Suzuki H, Tsukahara T. A view of pre-mRNA splicing from RNase R resistant RNAs. Int J Mol Sci. 2014;15(6):9331–42. https://doi.org/10.3390/ijms15069331.
Salzman J, Chen RE, Olsen MN, Wang PL, Brown PO. Cell-type specific features of circular RNA expression. PLoS Genet. 2013;9(9):e1003777. https://doi.org/10.1371/journal.pgen.1003777.
Zhang HD, Jiang LH, Sun DW, Hou JC, Ji ZL. CircRNA: a novel type of biomarker for cancer. Breast Cancer (Tokyo, Japan). 2018;25(1):1–7. https://doi.org/10.1007/s12282-017-0793-9.
Wang J, Fu J, Xu C, Jia R, Zhang X, Zhao S. Circ_ZFP644 attenuates caerulein-induced inflammatory injury in rat pancreatic acinar cells by modulating miR-106b/Pias3 axis. Exp Mol Pathol. 2021;121:104644. https://doi.org/10.1016/j.yexmp.2021.104644.
Guarnerio J, Bezzi M, Jeong JC, Paffenholz SV, Berry K, Naldini MM, et al. Oncogenic role of fusion-circRNAs derived from cancer-associated chromosomal translocations. Cell. 2016;165(2):289–302. https://doi.org/10.1016/j.cell.2016.03.020.
Ye X, Hang Y, Lu Y, Li D, Shen F, Guan P, et al. CircRNA circ-NNT mediates myocardial ischemia/reperfusion injury through activating pyroptosis by sponging miR-33a-5p and regulating USP46 expression. Cell Death Discov. 2021;7(1):370. https://doi.org/10.1038/s41420-021-00706-7.
Sun Z, Yu T, Jiao Y, He D, Wu J, Duan W, et al. Expression profiles and ontology analysis of circular RNAs in a mouse model of myocardial ischemia/reperfusion injury. Biomed Res Int. 2020;2020:2346369. https://doi.org/10.1155/2020/2346369.
Rivera-Barahona A, Pérez B, Richard E, Desviat LR. Role of miRNAs in human disease and inborn errors of metabolism. J Inherit Metab Dis. 2017;40(4):471–80. https://doi.org/10.1007/s10545-017-0018-6.
Hansen TB, Jensen TI, Clausen BH, Bramsen JB, Finsen B, Damgaard CK, et al. Natural RNA circles function as efficient microRNA sponges. Nature. 2013;495(7441):384–8. https://doi.org/10.1038/nature11993.
Matboli M, Shafei AE, Agwa SHA, Elzahy SS, Anwar AK, Mansour AR, et al. Identification of novel molecular network expression in acute myocardial infarction. Curr Genomics. 2019;20(5):340–8. https://doi.org/10.2174/1389202920666190820142043.
Elgebaly SA, Christenson RH, Kandil H, Ibrahim M, Rizk H, El-Khazragy N, et al. Nourin-dependent miR-137 and miR-106b: Novel biomarkers for early diagnosis of myocardial ischemia in coronary artery disease patients. Diagnostics (Basel, Switzerland). 2021;11(4). https://doi.org/10.3390/diagnostics11040703.
Liu YM, Cao Y, Zhao PS, Wu LY, Lu YM, Wang YL, et al. CircCCNB1 silencing acting as a miR-106b-5p sponge inhibited GPM6A expression to promote HCC progression by enhancing DYNC1I1 expression and activating the AKT/ERK signaling pathway. Int J Biol Sci. 2022;18(2):637–51. https://doi.org/10.7150/ijbs.66915.
Hu X, Wang P, Qu C, Zhang H, Li L. Circular RNA Circ_0000677 promotes cell proliferation by regulating microRNA-106b-5p/CCND1 in non-small cell lung cancer. Bioengineered. 2021;12(1):6229–39. https://doi.org/10.1080/21655979.2021.1965697.
Xiong Y, Wang Y, Tian H, Li Y, Xu Q, He Z. Circ-PRKCI alleviates lipopolysaccharide-induced human kidney 2 cell injury by regulating miR-106b-5p/GAB1 axis. J Cardiovasc Pharmacol. 2021;78(4):523–33. https://doi.org/10.1097/fjc.0000000000001031.
Galasso M, Sandhu SK, Volinia S. MicroRNA expression signatures in solid malignancies. Cancer J (Sudbury, Mass). 2012;18(3):238–43. https://doi.org/10.1097/PPO.0b013e318258b5f4.
Filipowicz W, Bhattacharyya SN, Sonenberg N. Mechanisms of post-transcriptional regulation by microRNAs: are the answers in sight? Nat Rev Genet. 2008;9(2):102–14. https://doi.org/10.1038/nrg2290.
Sierecki E, Newton AC. Biochemical characterization of the phosphatase domain of the tumor suppressor PH domain leucine-rich repeat protein phosphatase. Biochemistry. 2014;53(24):3971–81. https://doi.org/10.1021/bi500428j.
Jin A, Li B, Li W, Xiao D. PHLPP2 downregulation protects cardiomyocytes against hypoxia-induced injury through reinforcing Nrf2/ARE antioxidant signaling. Chem-Biol Interact. 2019;314:108848. https://doi.org/10.1016/j.cbi.2019.108848.
Westphal S, Stoppe C, Gruenewald M, Bein B, Renner J, Cremer J, et al. Genome-wide association study of myocardial infarction, atrial fibrillation, acute stroke, acute kidney injury and delirium after cardiac surgery - a sub-analysis of the RIPHeart-Study. BMC Cardiovasc Disord. 2019;19(1):26. https://doi.org/10.1186/s12872-019-1002-x.
Zhou LY, Zhai M, Huang Y, Xu S, An T, Wang YH, et al. The circular RNA ACR attenuates myocardial ischemia/reperfusion injury by suppressing autophagy via modulation of the Pink1/ FAM65B pathway. Cell Death Differ. 2019;26(7):1299–315. https://doi.org/10.1038/s41418-018-0206-4.
Song YF, Zhao L, Wang BC, Sun JJ, Hu JL, Zhu XL, et al. The circular RNA TLK1 exacerbates myocardial ischemia/reperfusion injury via targeting miR-214/RIPK1 through TNF signaling pathway. Free Radical Biol Med. 2020;155:69–80. https://doi.org/10.1016/j.freeradbiomed.2020.05.013.
Tang J, Yao C, Liu Y, Yuan J, Wu L, Hosoi K, et al. Arsenic trioxide induces expression of BCL-2 expression via NF-κB and p38 MAPK signaling pathways in BEAS-2B cells during apoptosis. Ecotoxicol Environ Saf. 2021;222:112531. https://doi.org/10.1016/j.ecoenv.2021.112531.
Song N, Ma J, Meng XW, Liu H, Wang H, Song SY, et al. Heat shock protein 70 protects the heart from ischemia/reperfusion injury through inhibition of p38 MAPK signaling. Oxid Med Cell Longev. 2020;2020:3908641. https://doi.org/10.1155/2020/3908641.
Yu J, Xu QG, Wang ZG, Yang Y, Zhang L, Ma JZ, et al. Circular RNA cSMARCA5 inhibits growth and metastasis in hepatocellular carcinoma. J Hepatol. 2018;68(6):1214–27. https://doi.org/10.1016/j.jhep.2018.01.012.
Qiao M, Wang Y, Xu X, Lu J, Dong Y, Tao W, et al. Mst1 is an interacting protein that mediates PHLPPs’ induced apoptosis. Mol Cell. 2010;38(4):512–23. https://doi.org/10.1016/j.molcel.2010.03.017.
Weinreuter M, Kreusser MM, Beckendorf J, Schreiter FC, Leuschner F, Lehmann LH, et al. CaM Kinase II mediates maladaptive post-infarct remodeling and pro-inflammatory chemoattractant signaling but not acute myocardial ischemia/reperfusion injury. EMBO Mol Med. 2014;6(10):1231–1245. https://doi.org/10.15252/emmm.201403848.
Neri M, Riezzo I, Pascale N, Pomara C, Turillazzi E. Ischemia/reperfusion injury following acute myocardial infarction: A critical issue for clinicians and forensic pathologists. Mediators Inflamm. 2017;2017:7018393. https://doi.org/10.1155/2017/7018393.
Yang R, Xing L, Zheng X, Sun Y, Wang X, Chen J. The circRNA circAGFG1 acts as a sponge of miR-195-5p to promote triple-negative breast cancer progression through regulating CCNE1 expression. Mol Cancer. 2019;18(1):4. https://doi.org/10.1186/s12943-018-0933-7.
Chen Y, Yang F, Fang E, Xiao W, Mei H, Li H, et al. Circular RNA circAGO2 drives cancer progression through facilitating HuR-repressed functions of AGO2-miRNA complexes. Cell Death Differ. 2019;26(7):1346–64. https://doi.org/10.1038/s41418-018-0220-6.
Li H, Yang F, Hu A, Wang X, Fang E, Chen Y, et al. Therapeutic targeting of circ-CUX1/EWSR1/MAZ axis inhibits glycolysis and neuroblastoma progression. EMBO Mol Med. 2019;11(12):e10835. https://doi.org/10.15252/emmm.201910835.
Garikipati VNS, Verma SK, Cheng Z, Liang D, Truongcao MM, Cimini M, et al. Circular RNA CircFndc3b modulates cardiac repair after myocardial infarction via FUS/VEGF-A axis. Nat Commun. 2019;10(1):4317. https://doi.org/10.1038/s41467-019-11777-7.
Altesha MA, Ni T, Khan A, Liu K, Zheng X. Circular RNA in cardiovascular disease. J Cell Physiol. 2019;234(5):5588–600. https://doi.org/10.1002/jcp.27384.
Patop IL, Kadener S. circRNAs in Cancer. Curr Opin Genet Dev. 2018;48:121–7. https://doi.org/10.1016/j.gde.2017.11.007.
Han B, Chao J, Yao H. Circular RNA and its mechanisms in disease: From the bench to the clinic. Pharmacol Ther. 2018;187:31–44. https://doi.org/10.1016/j.pharmthera.2018.01.010.
Liang Y, Jie H, Liu Q, Li C, Xiao R, Xing X, et al. Knockout of circRNA single stranded interacting protein 1 (circRBMS1) played a protective role in myocardial ischemia-reperfusion injury though inhibition of miR-2355-3p/Mammalian Sterile20-like kinase 1 (MST1) axis. Bioengineered. 2022;13(5):12726–37. https://doi.org/10.1080/21655979.2022.2068896.
Yuan C, Lu J, Chen Z, Zhou Q. Circ-GTF2I/miR-590-5p Axis Aggravates Myocardial Ischemia-Reperfusion Injury by Regulating Kelch Repeat and BTB Domain-Containing Protein 7. Evid-Based Complement Alternat Med. 2022;2022:2327669. https://doi.org/10.1155/2022/2327669.
Hao L, Wang J, Bi SJ, Cheng C. Upregulation of long noncoding RNA FGD5-AS1 ameliorates myocardial ischemia/reperfusion injury via MicroRNA-106a-5p and MicroRNA-106b-5p. J Cardiovasc Pharmacol. 2021;78(1):e45–54. https://doi.org/10.1097/fjc.0000000000001036.
Liu Z, Yang D, Xie P, Ren G, Sun G, Zeng X, et al. MiR-106b and MiR-15b modulate apoptosis and angiogenesis in myocardial infarction. Cell Physiol Biochem. 2012;29(5–6):851–62. https://doi.org/10.1159/000258197.
Wang L, Wang J, Yu P, Feng J, Xu GE, Zhao X, et al. METTL14 is required for exercise-induced cardiac hypertrophy and protects against myocardial ischemia-reperfusion injury. Nat Commun. 2022;13(1):6762. https://doi.org/10.1038/s41467-022-34434-y.
Yan X, Li W, Yang L, Dong W, Chen W, Mao Y, et al. MiR-135a protects vascular endothelial cells against ventilator-induced lung injury by inhibiting PHLPP2 to activate PI3K/Akt pathway. Cell Physiol Biochem. 2018;48(3):1245–58. https://doi.org/10.1159/000492010.
Liu YY, Liu X, Zhou JG, Liang SJ. MicroRNA-302a promotes neointimal formation following carotid artery injury in mice by targeting PHLPP2 thus increasing Akt signaling. Acta Pharmacol Sin. 2021;42(4):550–9. https://doi.org/10.1038/s41401-020-0440-4.
Tang J, Yan B, Tang Y, Zhou X, Ji Z, Xu F. Baicalein ameliorates oxidative stress and brain injury after intracerebral hemorrhage by activating the Nrf2/ARE pathway via miR-106a-5p/PHLPP2 axis. Int J Neurosci. 2022;1–14. https://doi.org/10.1080/00207454.2022.2080676.
Funding
This research was supported by Dalian Municipal Central Hospital Gaofeng Plan of Dalian Key Medical Specialty ‘Dengfeng Plan’ at Department of Cardiology.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Ethics Approval
No human studies were carried out by the authors for this article. All institutional and national guidelines for the care and use of laboratory animals were followed and approved by the ethics committee on live animals of Dalian Municipal Central Hospital (No. 2017PS041K).
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Associate Editor Guoping Li oversaw the review of this article.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.

Fig. S1
Downregulation of miR-106b-5p inhibits cell viability and aggravates oxidative stress and apoptosis through regulating Phlpp2. A. Real-time PCR evaluated the expression of miR-106b-5p in HL-1 cells. B. The mRNA level of Phlpp2 was detected by real-time PCR. C. Phlpp2 protein expression was assessed using western blot. D. The knockdown efficiency of siPhlpp2 was examined using real-time PCR. E and F. The mRNA and protein expression of Phlpp2 was estimated in OGD/R-induced HL-1 cells. G. MDA content and SOD activity were evaluated using the corresponding kits. H. Cell viability was examined using MTT assay in each group. I. Cell apoptosis was detected by TUNEL assay. Scale bars = 50 μm. There were three biological replicates in each group. Data are presented as mean ± SD. * P < 0.05; ** P < 0.01; ns, P > 0.05. (PNG 562 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, L., Wang, C., Sun, Z. et al. Knockdown of Mmu-circ-0001380 Attenuates Myocardial Ischemia/Reperfusion Injury via Modulating miR-106b-5p/Phlpp2 Axis. J. of Cardiovasc. Trans. Res. 16, 1064–1077 (2023). https://doi.org/10.1007/s12265-023-10383-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12265-023-10383-9