Abstract
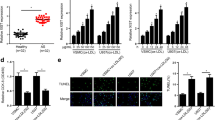
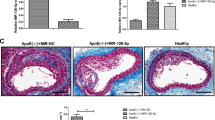
Abnormal function of endothelial cells (ECs) is an important reason for vascular endothelial remodeling and atherosclerotic plaque formation in patients with atherosclerosis (AS). Here, we report for the first time that the vascular ECs with apoptosis resistance phenotype (ARECs) exist in peripheral blood of AS patients. Our research data showed that the switch of regulation modes between HIF-1α and Bax operated by lncRNA-ASLNC18810 is the direct cause for the formation of ARECs. When ASLNC18810 is low or missing, HIF-1α indirectly negatively regulates the Bax in post-transcription through HIF-1α/miR-559/Bax pathway which makes ECs acquire apoptosis resistance and form ARECs. The functional experiments results showed that ASLNC18810 could effectively eliminate the anti-apoptotic properties of ARECs by blocking the HIF-1α/miR559/Bax pathway and maintaining HIF-1α/Bax pathway. In a word, our study shows that ASLNC18810 has full potential to become a biological target for the prevention and treatment of atherosclerotic plaques by regulating ARECs.
Graphical abstract
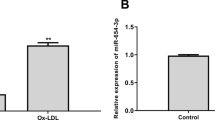
ASLNC18810 was significantly upregulated in ECs compared to ARECs. With high level of ASLNC18810 in ECs, ASLNC18810 binds to miR-559 as a miRNA sponge and suppresses the inhibition effect of miR-559 on Bax protein, this direct positive transcriptional regulation between HIF-1α and Bax endows the apoptotic property in ECs induced by Ox-LDL. However, with low expression of ASLNC18810 in ARECs, the post-transcriptional regulation of Bax by miR-559 dominates and the indirect negative regulation between HIF-1α and Bax endows the anti-apoptotic property of ARECs. To sum up, low ASLNC18810 expression-mediated switching of HIF-1α/Bax pathway to HIF-1α/miR-559/Bax pathway is the internal reason for ECs to obtain apoptosis resistance and the formation of ARECs under the ox-LDL induction.







Similar content being viewed by others
Abbreviations
- AS:
-
Atherosclerosis
- ECs:
-
Endothelial cells
- ARECs:
-
Apoptosis-resistant endothelial cells
- Ox-LDL:
-
Oxidized low-density lipoprotein
- LncRNA:
-
Long non-coding RNA
- MiRNA:
-
MicroRNA
- HIF-1α:
-
Hypoxia inducible factor-1α
- Bax:
-
BCL2-associated X protein
- HFD:
-
High-fat diet
- RT-qPCR:
-
Quantitative reverse transcription PCR
- GFP:
-
Green fluorescent protein
- TFBS:
-
Transcription factor binding site
- PI:
-
Propidium Iodide
- MOI:
-
Multiplicity of infection
- DMEM:
-
Dulbecco’s modification of Eagle’s medium
- NC:
-
Negative control
- IF:
-
Immunofluorescence
- cDNA:
-
Complementary DNA
- UTR:
-
Untranslated regions
- EPCs:
-
Endothelial progenitor cells
References
Gimbrone, M. A., Jr., & García-Cardeña, G. (2016). Endothelial cell dysfunction and the pathobiology of atherosclerosis. Circulation Research, 118, 620–636.
Hinterdobler, J., Schunkert, H., Kessler, T., & Sager, H. B. (2021). Impact of acute and chronic psychosocial stress on vascular inflammation. Antioxidants & Redox Signaling, 35, 1531–1550.
Roth, L., Van der Donckt, C., EminiVeseli, B., Van Dam, D., De Deyn, P. P., Martinet, W., Herman, A. G., & De Meyer, G. R. Y. (2019). Nitric oxide donor molsidomine favors features of atherosclerotic plaque stability and reduces myocardial infarction in mice. Vascular Pharmacology, 118–119, 106561.
Malekmohammad, K., Sewell, R. D. E., & Rafieian-Kopaei, M. (2019). Antioxidants and atherosclerosis: Mechanistic aspects. Biomolecules, 9, 301.
Masri, F. A., Xu, W., Comhair, S. A., Asosingh, K., Koo, M., Vasanji, A., Drazba, J., Anand-Apte, B., & Erzurum, S. C. (2007). Hyperproliferative apoptosis-resistant endothelial cells in idiopathic pulmonary arterial hypertension. American journal of physiology. Lung cellular and molecular physiology, 293, L548-554.
Sakao, S., Taraseviciene-Stewart, L., Lee, J. D., Wood, K., Cool, C. D., & Voelkel, N. F. (2005). Initial apoptosis is followed by increased proliferation of apoptosis-resistant endothelial cells. FASEB journal : Official publication of the Federation of American Societies for Experimental Biology, 19, 1178–1180.
Liu, B., & Li, Y. (2015). Impact of conditional miRNA126 overexpression on apoptosis-resistant endothelial cell production. PloS one, 10, e0126661.
Camaré, C., Pucelle, M., Nègre-Salvayre, A., & Salvayre, R. (2017). Angiogenesis in the atherosclerotic plaque. Redox Biology, 12, 18–34.
Fasolo, F., Di Gregoli, K., Maegdefessel, L., & Johnson, J. L. (2019). Non-coding RNAs in cardiovascular cell biology and atherosclerosis. Cardiovascular Research, 115, 1732–1756.
Indolfi, C., Iaconetti, C., Gareri, C., Polimeni, A., & De Rosa, S. (2019). Non-coding RNAs in vascular remodeling and restenosis. Vascular Pharmacology, 114, 49–63.
Kumar, S., Williams, D., Sur, S., Wang, J. Y., & Jo, H. (2019). Role of flow-sensitive microRNAs and long noncoding RNAs in vascular dysfunction and atherosclerosis. Vascular Pharmacology, 114, 76–92.
Wang, Q., Yu, Y., Zhang, P., Chen, Y., Li, C., Chen, J., Wang, Y., & Li, Y. (2017). The crucial role of activin A/ALK4 pathway in the pathogenesis of Ang-II-induced atrial fibrosis and vulnerability to atrial fibrillation. Basic Research in Cardiology, 112, 47.
Bäck, M., Yurdagul, A., Jr., Tabas, I., Öörni, K., & Kovanen, P. T. (2019). Inflammation and its resolution in atherosclerosis: Mediators and therapeutic opportunities. Nature reviews. Cardiology, 16, 389–406.
Fan, Z., Zhang, Y., Xiao, D., Ma, J., Liu, H., Shen, L., Zhang, M., & He, B. (2020). Long noncoding RNA UC.98 stabilizes atherosclerotic plaques by promoting the proliferation and adhesive capacity in murine aortic endothelial cells. Acta Biochimica et Biophysica Sinica, 52, 141–149.
Viktorinova, A. (2019). Potential clinical utility of macrophage colony-stimulating factor, monocyte chemotactic protein-1 and myeloperoxidase in predicting atherosclerotic plaque instability. Discovery Medicine, 28, 237–245.
Kavurma, M. M., Rayner, K. J., & Karunakaran, D. (2017). The walking dead: Macrophage inflammation and death in atherosclerosis. Current Opinion in Lipidology, 28, 91–98.
Wang, W., Liu, W., Fidler, T., Wang, Y., Tang, Y., Woods, B., Welch, C., Cai, B., Silvestre-Roig, C., Ai, D., Yang, Y. G., Hidalgo, A., Soehnlein, O., Tabas, I., Levine, R. L., Tall, A. R., & Wang, N. (2018). Macrophage inflammation, erythrophagocytosis, and accelerated atherosclerosis in Jak2 (V617F) mice. Circulation Research, 123, e35–e47.
Chen, L., Yang, W., Guo, Y., Chen, W., Zheng, P., Zeng, J., & Tong, W. (2017). Exosomal lncRNA GAS5 regulates the apoptosis of macrophages and vascular endothelial cells in atherosclerosis. PloS One, 12, e0185406.
Kattoor, A. J., Kanuri, S. H., & Mehta, J. L. (2019). Role of Ox-LDL and LOX-1 in atherogenesis. Current Medicinal Chemistry, 26, 1693–1700.
Cao, Y., Jiang, Z., Zeng, Z., Liu, Y., Gu, Y., Ji, Y., Zhao, Y., & Li, Y. (2016). Bcl-2 silencing attenuates hypoxia-induced apoptosis resistance in pulmonary microvascular endothelial cells. Apoptosis : An International Journal on Programmed Cell Death, 21, 69–84.
Lévy, M., Maurey, C., Celermajer, D. S., Vouhé, P. R., Danel, C., Bonnet, D., & Israël-Biet, D. (2007). Impaired apoptosis of pulmonary endothelial cells is associated with intimal proliferation and irreversibility of pulmonary hypertension in congenital heart disease. Journal of the American College of Cardiology, 49, 803–810.
Lu, Y., Thavarajah, T., Gu, W., Cai, J., & Xu, Q. (2018). Impact of miRNA in Atherosclerosis. Arteriosclerosis, Thrombosis, and Vascular Biology, 38, e159–e170.
Wang, Z., Wang, R., Wang, K., & Liu, X. (2018). Upregulated long noncoding RNA Snhg1 promotes the angiogenesis of brain microvascular endothelial cells after oxygen-glucose deprivation treatment by targeting miR-199a. Canadian Journal of Physiology and Pharmacology, 96, 909–915.
Yu, X. H., Deng, W. Y., Chen, J. J., Xu, X. D., Liu, X. X., Chen, L., Shi, M. W., Liu, Q. X., Tao, M., & Ren, K. (2020). LncRNA kcnq1ot1 promotes lipid accumulation and accelerates atherosclerosis via functioning as a ceRNA through the miR-452-3p/HDAC3/ABCA1 axis. Cell Death & Disease, 11, 1043.
Funding
This work was supported by the National Natural Science Foundation of China (81900293), Shanghai Sailing Program (19YF1431800).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics Approval
All human studies were carried out under informed consent, with data collection approved by the Ethics Committee of Xinhua Hospital affiliated to Shanghai Jiao Tong University School of Medicine. All institutional and national guidelines for the care and use of laboratory animals were followed and approved by the appropriate institutional committees.
Conflict of Interest
The authors declare no competing interests.
Additional information
Associate Editor Junjie Xiao oversaw the review of this article
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, Q., Wan, YY., Li, N. et al. The Mechanism Underlying the Regulation of LncRNA-ASLNC18810 Involved in the Abnormal Function of Vascular Endothelial Cell in Atherosclerosis: Its Function as a microRNA (miRNA) Sponge for miR-559. J. of Cardiovasc. Trans. Res. 15, 1010–1023 (2022). https://doi.org/10.1007/s12265-022-10230-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12265-022-10230-3