Abstract
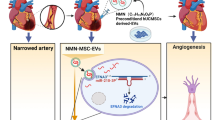
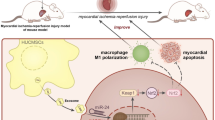
Our research intended to investigate the roles of mesenchymal stem cell (MSC)-derived extracellular vesicles (EVs) in acute myocardial infarction (AMI) via delivery of microRNA (miR)-302d-3p. AMI mouse models were established. EVs isolated from MSCs with miR-302d-3p mimic were injected near the infarct area or co-cultured with hypoxic cardiomyocytes to evaluate their effects. The expression of NF-κB pathway-related genes and inflammatory factors was determined. AMI mice exhibited downregulated miR-302d-3p and elevated MD2 and BCL6 levels. BCL6 was negatively targeted by miR-302d-3p and could bind to MD2 promoter to upregulate MD2 expression. MSCs-EVs, MSCs-EVs carrying miR-302d-3p, or BCL6 or MD2 silencing inactivated the NF-κB pathway and alleviated infarcted area, myocardial fibrosis, inflammation, apoptosis, and cardiac dysfunction in AMI mice. Besides, MSCs-EVs, MSCs-EVs carrying miR-302d-3p, or BCL6 or MD2 silencing diminished viability and inflammation but augmented apoptosis of hypoxic cardiomyocytes. Conclusively, MSCs-EVs carrying miR-302d-3p repressed inflammation and cardiac remodeling after AMI via BCL6/MD2/NF-κB axis.








Similar content being viewed by others
References
Anderson, J. L., & Morrow, D. A. (2017). Acute myocardial infarction. New England Journal of Medicine, 376(21), 2053–2064. https://doi.org/10.1056/NEJMra1606915
Reed G. W., Rossi, J. E., & Cannon, C. P. (2017). Department of error. Lancet, 389(10065):156. https://doi.org/10.1016/S0140-6736(17)30007-7
Wang, C., & Jing, Q. (2018). Non-coding RNAs as biomarkers for acute myocardial infarction. Acta Pharmacologica Sinica, 39(7), 1110–1119. https://doi.org/10.1038/aps.2017.205
Lalem, T., & Devaux, Y. (2019). Circulating microRNAs to predict heart failure after acute myocardial infarction in women. Clin Biochem, 70, 1–7. https://doi.org/10.1016/j.clinbiochem.2019.05.011
Khodayari, S., Khodayari, H., Amiri, A. Z., Eslami, M., Farhud, D., Hescheler, J., et al. (2019). Inflammatory microenvironment of acute myocardial infarction prevents regeneration of heart with stem cells therapy. Cell Physiol Biochem, 53(5), 887–909. https://doi.org/10.33594/000000180
Schirone, L., Forte, M., Palmerio, S., Yee, D., Nocella, C., Angelini, F., et al. (2017). A review of the molecular mechanisms underlying the development and progression of cardiac remodeling. Oxid Med Cell Longev, 2017, 3920195. https://doi.org/10.1155/2017/3920195
Wang, C., Hou, J., Du, H., Yan, S., Yang, J., Wang, Y., et al. (2019). Anti-depressive effect of Shuangxinfang on rats with acute myocardial infarction: Promoting bone marrow mesenchymal stem cells mobilization and alleviating inflammatory response. Biomed Pharmacother, 111, 19–30. https://doi.org/10.1016/j.biopha.2018.11.113
Huang, P., Wang, L., Li, Q., Xu, J., Xu, J., Xiong, Y., et al. (2019). Combinatorial treatment of acute myocardial infarction using stem cells and their derived exosomes resulted in improved heart performance. Stem Cell Research & Therapy, 10(1), 300. https://doi.org/10.1186/s13287-019-1353-3
van Niel, G., D’Angelo, G., & Raposo, G. (2018). Shedding light on the cell biology of extracellular vesicles. Nature Reviews Molecular Cell Biology, 19(4), 213–228. https://doi.org/10.1038/nrm.2017.125
Lucchetti, D., Fattorossi, A., & Sgambato, A. (2019). Extracellular vesicles in oncology: Progress and pitfalls in the methods of isolation and analysis. Biotechnology Journal, 14(1), e1700716. https://doi.org/10.1002/biot.201700716
Nakamura, Y., Miyaki, S., Ishitobi, H., Matsuyama, S., Nakasa, T., Kamei, N., et al. (2015). Mesenchymal-stem-cell-derived exosomes accelerate skeletal muscle regeneration. FEBS Letters, 589(11), 1257–1265. https://doi.org/10.1016/j.febslet.2015.03.031
Liu, W., Zhuang, J., Jiang, Y., Sun, J., Prinz, R. A., Sun, J., et al. (2019). Toll-like receptor signalling cross-activates the autophagic pathway to restrict Salmonella typhimurium growth in macrophages. Cellular Microbiology, 21(12), e13095. https://doi.org/10.1111/cmi.13095
Gu, Y., Luo, M., Li, Y., Su, Z., Wang, Y., Chen, X., et al. (2019). Bcl6 knockdown aggravates hypoxia injury in cardiomyocytes via the P38 pathway. Cell Biology International, 43(2), 108–116. https://doi.org/10.1002/cbin.11028
Wang, X., Guo, D., Li, W., Zhang, Q., Jiang, Y., Wang, Q., et al. (2020). Danshen (Salvia miltiorrhiza) restricts MD2/TLR4-MyD88 complex formation and signalling in acute myocardial infarction-induced heart failure. Journal of Cellular and Molecular Medicine, 24(18), 10677–10692. https://doi.org/10.1111/jcmm.15688
Liu, J., Jiang, M., Deng, S., Lu, J., Huang, H., Zhang, Y., et al. (2018). miR-93–5p-containing exosomes treatment attenuates acute myocardial infarction-induced myocardial damage. Mol Ther Nucleic Acids, 11, 103–115. https://doi.org/10.1016/j.omtn.2018.01.010
Zhu, J., Lu, K., Zhang, N., Zhao, Y., Ma, Q., Shen, J., et al. (2018). Myocardial reparative functions of exosomes from mesenchymal stem cells are enhanced by hypoxia treatment of the cells via transferring microRNA-210 in an nSMase2-dependent way. Artif Cells Nanomed Biotechnol, 46(8), 1659–1670. https://doi.org/10.1080/21691401.2017.1388249
Boersma, E., Mercado, N., Poldermans, D., Gardien, M., Vos, J., & Simoons, M. L. (2003). Acute myocardial infarction. Lancet, 361(9360), 847–858. https://doi.org/10.1016/S0140-6736(03)12712-2
Xiong, Y. Y., Gong, Z. T., Tang, R. J., & Yang, Y. J. (2021). The pivotal roles of exosomes derived from endogenous immune cells and exogenous stem cells in myocardial repair after acute myocardial infarction. Theranostics, 11(3), 1046–1058. https://doi.org/10.7150/thno.53326
Wang, N., Chen, C., Yang, D., Liao, Q., Luo, H., Wang, X., et al. (2017). Mesenchymal stem cells-derived extracellular vesicles, via miR-210, improve infarcted cardiac function by promotion of angiogenesis. Biochimica et Biophysica Acta, Molecular Basis of Disease, 1863(8), 2085–2092. https://doi.org/10.1016/j.bbadis.2017.02.023
He, J. G., Li, H. R., Han, J. X., Li, B. B., Yan, D., Li, H. Y., et al. (2018). GATA-4-expressing mouse bone marrow mesenchymal stem cells improve cardiac function after myocardial infarction via secreted exosomes. Science and Reports, 8(1), 9047. https://doi.org/10.1038/s41598-018-27435-9
Firoozi, S., Pahlavan, S., Ghanian, M. H., Rabbani, S., Barekat, M., Nazari, A., et al. (2020). Mesenchymal stem cell-derived extracellular vesicles alone or in conjunction with a SDKP-conjugated self-assembling peptide improve a rat model of myocardial infarction. Biochemical and Biophysical Research Communications, 524(4), 903–909. https://doi.org/10.1016/j.bbrc.2020.02.009
Huang, P., Wang, L., Li, Q., Tian, X., Xu, J., Xu, J., et al. (2020). Atorvastatin enhances the therapeutic efficacy of mesenchymal stem cells-derived exosomes in acute myocardial infarction via up-regulating long non-coding RNA H19. Cardiovascular Research, 116(2), 353–367. https://doi.org/10.1093/cvr/cvz139
Liu, J., Chen, T., Lei, P., Tang, X., & Huang, P. (2019). Exosomes released by bone marrow mesenchymal stem cells attenuate lung injury induced by intestinal ischemia reperfusion via the TLR4/NF-kappaB pathway. International Journal of Medical Sciences, 16(9), 1238–1244. https://doi.org/10.7150/ijms.35369
Pramanik K. C., Makena M. R., Bhowmick K. & Pandey M. K. (2018). Advancement of NF-kappaB signaling pathway: A novel target in pancreatic cancer. Int J Mol Sci, 19(12):https://doi.org/10.3390/ijms19123890
Ge, Z. W., Wang, B. C., Hu, J. L., Sun, J. J., Wang, S., Chen, X. J., et al. (2019). IRAK3 gene silencing prevents cardiac rupture and ventricular remodeling through negative regulation of the NF-kappaB signaling pathway in a mouse model of acute myocardial infarction. Journal of Cellular Physiology, 234(7), 11722–11733. https://doi.org/10.1002/jcp.27827
Jiang, Y., Feng, Y. P., Tang, L. X., Yan, Y. L., & Bai, J. W. (2019). The protective role of NR4A3 in acute myocardial infarction by suppressing inflammatory responses via JAK2-STAT3/NF-kappaB pathway. Biochemical and Biophysical Research Communications, 517(4), 697–702. https://doi.org/10.1016/j.bbrc.2019.07.116
Ma, C., Jiang, Y., Zhang, X., Chen, X., Liu, Z., & Tian, X. (2018). Isoquercetin ameliorates myocardial infarction through anti-inflammation and anti-apoptosis factor and regulating TLR4-NF-kappaB signal pathway. Molecular Medicine Reports, 17(5), 6675–6680. https://doi.org/10.3892/mmr.2018.8709
Kim, J. Y., Shin, K. K., Lee, A. L., Kim, Y. S., Park, H. J., Park, Y. K., et al. (2014). MicroRNA-302 induces proliferation and inhibits oxidant-induced cell death in human adipose tissue-derived mesenchymal stem cells. Cell Death Dis, 5, e1385. https://doi.org/10.1038/cddis.2014.344
Chen, Y. L., Xu, Q. P., Guo, F., & Guan, W. H. (2017). MicroRNA-302d downregulates TGFBR2 expression and promotes hepatocellular carcinoma growth and invasion. Experimental and Therapeutic Medicine, 13(2), 681–687. https://doi.org/10.3892/etm.2016.3970
Alimov, I., Menon, S., Cochran, N., Maher, R., Wang, Q., Alford, J., et al. (2019). Bile acid analogues are activators of pyrin inflammasome. Journal of Biological Chemistry, 294(10), 3359–3366. https://doi.org/10.1074/jbc.RA118.005103
Sun, N., Zhang, L., Zhang, C., & Yuan, Y. (2020). miR-144–3p inhibits cell proliferation of colorectal cancer cells by targeting BCL6 via inhibition of Wnt/beta-catenin signaling. Cell Mol Biol Lett, 25, 19. https://doi.org/10.1186/s11658-020-00210-3
Wei, P., Chen, H., Lin, B., Du, T., Liu, G., He, J., et al. (2021). Inhibition of the BCL6/miR-31/PKD1 axis attenuates oxidative stress-induced neuronal damage. Exp Neurol, 335, 113528. https://doi.org/10.1016/j.expneurol.2020.113528
Zhu, B., Zhang, R., Li, C., Jiang, L., Xiang, M., Ye, Z., et al. (2019). BCL6 modulates tissue neutrophil survival and exacerbates pulmonary inflammation following influenza virus infection. Proc Natl Acad Sci U S A, 116(24), 11888–11893. https://doi.org/10.1073/pnas.1902310116
Huang, C., & Melnick, A. (2015). Mechanisms of action of BCL6 during germinal center B cell development. Sci China Life Sci, 58(12), 1226–1232. https://doi.org/10.1007/s11427-015-4919-z
Tsunoda, F., Koba, S., Hirano, T., Ban, Y., Iso, Y., Suzuki, H., et al. (2004). Association between small dense low-density lipoprotein and postprandial accumulation of triglyceride-rich remnant-like particles in normotriglyceridemic patients with myocardial infarction. Circulation Journal, 68(12), 1165–1172. https://doi.org/10.1253/circj.68.1165
Koba, S., Hirano, T., Murayama, S., Kotani, T., Tsunoda, F., Iso, Y., et al. (2003). Small dense LDL phenotype is associated with postprandial increases of large VLDL and remnant-like particles in patients with acute myocardial infarction. Atherosclerosis, 170(1), 131–140. https://doi.org/10.1016/s0021-9150(03)00245-4
Funding
This study was supported by the Guidance Project of Social Development Plan of Science and Technology Bureau of Qiqihar in 2019 (No. SFZD-2019139).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics Approval
Animal experiments were conducted under the approval of the Animal Ethics Committee of The Third Affiliated Hospital of Qiqihar Medical University and in strict accordance with the recommendations of the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health. Extensive efforts were made to ensure minimal suffering of the included animals.
Conflict of Interest
The authors declare no competing interests.
Additional information
Communicated by Associate Editor Yihua Bei oversaw the review of this article.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Liu, Y., Guan, R., Yan, J. et al. Mesenchymal Stem Cell-Derived Extracellular Vesicle-Shuttled microRNA-302d-3p Represses Inflammation and Cardiac Remodeling Following Acute Myocardial Infarction. J. of Cardiovasc. Trans. Res. 15, 754–771 (2022). https://doi.org/10.1007/s12265-021-10200-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12265-021-10200-1