Abstract
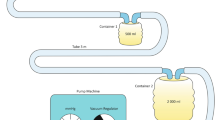
The present study was undertaken to develop the device that can be used to harvest adipose tissue for fat grafting with an airtight, minimal-invasive, and fast approach. The abdominal liposuction was performed in six women under local tumescent anesthesia. Fat from left abdomen was harvested by the classical 20-ml-syringe technique while fat from right abdomen was harvested by a reformative vacuum-assisted suction device (negative pressure, 25 to 35 kPa). This device includes a 1-L sterile canister, a cannula with 2.5 mm inner diameter, a conventional liposuction machine, and a one-way valve connected between the canister and the liposuction machine. The operative time of harvesting 200-ml fat grafts was recorded and calculated. The collected adipose tissue was used for adipose-derived stem cells (ADSCs) extraction and culture, glucose transport test (GTT), and bacteriological culture. First, we found that the operative time of harvesting 200-ml adipose tissue using 20-ml-syringe aspiration technique was 71.3 ± 4.3 min compared with 30 ± 3.5 min by the new device (p < 0.05). The sizes of the fat grafts obtained from both methods were similar. Second, the viability of harvested adipose tissue tested through GTT showed no significant difference between two harvesting methods (p > 0.05). Additionally, the samples harvested from both methods showed no bacterial growth. The results from the XTT test revealed that there was no significant difference on ADSCs growth between two methods (p > 0.05). In conclusion, the results support that the innovative device for adipose tissue harvesting is minimal-invasive, fast, and can prevent the fat grafts from contamination.




Similar content being viewed by others
References
Hsu VM, Stransky CA, Bucky LP, Percec I (2012) Fat grafting’s past, present, and future: why adipose tissue is emerging as a critical link to the advancement of regenerative medicine. Aesthet Surg J 32:892–899. https://doi.org/10.1177/1090820X12455658
Grahovac TL, Rubin JP (2012) An analysis of the experiences of 62 patients with moderate complications after full-face fat injection for augmentation. Plast Reconstr Surg 129:1369–1370. https://doi.org/10.1097/PRS.0b013e31824f4ffc
Simonacci F, Bertozzi N, Grieco MP, Grignaffini E, Raposio E (2017) Procedure, applications, and outcomes of autologous fat grafting. Ann Med Surg 20:49–60. https://doi.org/10.1016/j.amsu.2017.06.059
Yu NZ, Huang JZ, Zhang H, Wang Y, Wang XJ, Zhao R, Bai M, Long X (2015) A systemic review of autologous fat grafting survival rate and related severe complications. Chin Med J 128:1245–1251. https://doi.org/10.4103/0366-6999.156142
Alexander RW, Harrell DB (2013) Autologous fat grafting: use of closed syringe microcannula system for enhanced autologous structural grafting. Clin Cosmet Investig Dermatol 6:91–102. https://doi.org/10.2147/CCID.S40575
Koldaş T, Onel D, Ozden BC (2004) The use of a towel clamp as a complementary instrument for manual fat harvest. Ann Plast Surg 52:626–627
Xie Y, Zheng D, Li Q, Chen Y, Lei H, Pu LL (2010) The effect of centrifugation on viability of fat grafts: an evaluation with the glucose transport test. J Plast Reconstr Aesthet Surg 63:482–427. https://doi.org/10.1016/j.bjps.2008.11.056
Tuttle MS, Mostow E, Mukherjee P, Hu FZ, Melton-Kreft R, Ehrlich GD, Dowd SE, Ghannoum MA (2011) Characterization of bacterial communities in venous insufficiency wounds by use of conventional culture and molecular diagnostic methods. J Clin Microbiol 49:3812–3819. https://doi.org/10.1128/JCM.00847-11
Foroutan T, Farhadi A, Abroun S, Mohammad Soltani B (2018) Adipose derived stem cells affect mir-145 and p53 expressions of co-cultured hematopoietic stem cell. Cell J 19: 654-659. https://doi.org/10.22074/cellj.2018.4393
Lu F, Mizuno H, Uysal CA, Cai X, Ogawa R, Hyakusoku H (2008) Improved viability of random pattern skin flaps through the use of adipose-derived stem cells. Plast Reconstr Surg 121:50–58. https://doi.org/10.1097/01.prs.0000293876.10700.b8
Tan M, Xu X, Ohba M, Ogawa W, Cui MZ (2003) Thrombin rapidly induces protein kinase D phosphorylation, and protein kinase C δ mediates the activation. J Biol Chem 278:2824–2828. https://doi.org/10.1074/jbc.M211523200
Zuk PA, Zhu M, Mizuno H, Huang J, Futrell JW, Katz AJ, Benhaim P, Lorenz HP, Hedrick MH (2001) Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng 7:211–228. https://doi.org/10.1089/107632701300062859
Muscari C, Bonafè F, Fiumana E, Oranges CM, Pinto V, Caldarera CM, Guarnieri C, Morselli PG (2013) Comparison between stem cells harvested from wet and dry lipoaspirates. Connect Tissue Res 54:34–40. https://doi.org/10.3109/03008207.2012.717130
Ayestaray B, Georgescu D, Baron M, Bekara F (2013) Vacuum-assisted adipose tissue suction technique (VAST) to optimize fat harvesting. Aesthet Plast Surg 37:816–821. https://doi.org/10.1007/s00266-013-0118-0
Mojallal A, Auxenfans C, Lequeux C, Braye F, Damour O (2008) Influence of negative pressure when harvesting adipose tissue on cell yield of the stromal-vascular fraction. Biomed Mater Eng 18:193–197
Tambasco D, Arena V, Grussu F, Cervelli D (2013) Adipocyte damage in relation to different pressures generated during manual lipoaspiration with a syringe. Plast Reconstr Surg 131: 645e-6e. https://doi.org/10.1097/PRS.0b013e3182827760
Castro-Govea Y, De La Garza-Pineda O, Lara-Arias J, Chacón-Martínez H, Mecott-Rivera G, Salazar-Lozano A, Valdes-Flores E (2012) Cell-assisted lipotransfer for the treatment of Parry-Romberg syndrome. Arch Plast Surg 39:659–662. https://doi.org/10.5999/aps.2012.39.6.659
Bourin P, Bunnell BA, Casteilla L, Dominici M, Katz AJ, March KL, Redl H, Rubin JP, Yoshimura K, Gimble JM (2013) Stromal cells from the adipose tissue-derived stromal vascular fraction and culture expanded adipose tissue-derived stromal/stem cells: a joint statement of the International Federation for Adipose Therapeutics and Science (IFATS) and the International Society for Cellular Therapy (ISCT). Cytotherapy 15:641–648. https://doi.org/10.1016/j.jcyt.2013.02.006
Funding
This work was supported by the National Nature and Science Foundation, China (81372065 and 81871563), the Major Project of Guangzhou Municipal Science and Technology Bureau (201300000091 and 201508020253), the Fundamental Research Funds for the Central Universities (21619350), and the Foster Research Fund of The First Affiliated Hospital of Jinan University (2017306).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethics Approval and Consent to Participate
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent for participation in the study or use of their tissue was obtained from all participants. This article does not contain any studies with animals performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Huang, HL., Liao, X., Liu, HW. et al. Development and Evaluation of the Airtight, Minimal-Invasive, and Fast Device Harvesting Adipose Tissue for Autologous Fat Grafting. Indian J Surg 82, 545–550 (2020). https://doi.org/10.1007/s12262-019-02036-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12262-019-02036-6