Abstract
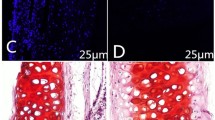
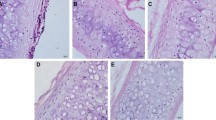
Forty tracheas were harvested from donor New Zealand rabbits. Thirty of the tracheas were randomly divided into four treatment groups corresponding to 4, 5, 6, or 7% NaClO4 and one untreated group (n = 6 each group). Scanning electron microscopy distinctly revealed the cilium of epithelial cells in the fresh trachea. The internal surface of the trachea was rough in the 4% treatment group and smooth in the 5% treatment group, whereas the matrix was fractured in the 6% treatment group and highly fractured in the 7% treatment group. We observed that the number of nuclei in the cells of the 4, 5, 6, and 7% treatment groups decreased compared to the cells of the untreated group (p < 0.05). Although there was a significant decrease in maximum tensile strength, tensile strain at fracture and the elastic modulus (p < 0.05) with increasing concentrations of NaClO4, the content of glycosaminoglycans (GAGs) did not significantly decline (p > 0.05) in the 5% treatment group. In addition, histopathological analysis showed that the fiber component and basement membrane of the matrix in the 5% treatment group were retained after optimal decellularization. Despite the preserved cartilage, in vitro immunohistochemical analysis revealed that the matrix did not show the presence of major histocompatibility complex (MHC) antigens. The remaining ten donor tracheas, which were divided into a positive control group and an optimal decellularized group, were used for allogeneic transplantation. Blood samples were taken regularly, and histologic examinations were performed at 30 days postimplantation, which showed no significant immune rejection. In conclusion, we surveyed the structural integrity through morphological observation and compared the biomechanical and immunogenic changes in the tracheal matrix under the different treatments. The optimal decellularized tracheal matrix with preserved cartilage, which was acquired via 5% NaClO4 treatment, exhibited structural integrity, antigen cell removal and immune privilege and would be suitable for use as a tissue-engineered trachea for in vivo transplantation in rabbit models.
Similar content being viewed by others
References
Baiguera, S., B. D’Innocenzo, and P. Macchiarini (2011) Current status of regenerative replacement of the airway. Expert. Rev. Respir. Med. 5: 487–494.
Haag, J. C., P. Jungebluth, and P. Macchiarini (2013) Tracheal replacement for primary tracheal cancer. Curr. Opin. Otolaryngol. Head Neck Surg. 21: 171–177.
Jungebluth, P., G. Moll, and S. Baiguera (2012) Tissue-engineered airway: A regenerative solution. Clin. Pharmacol. Ther. 91: 81–93.
Kojima, K. and C. A. Vacanti (2014) Tissue engineering in the trachea. Anat. Rec. 297: 44–50.
Jungebluth, P. and P. Macchiarini (2014) Airway transplantation. Thorac. Surg. Clin. 24: 97–106.
Ma, R., M. Li, and J. Luo (2013) Structural integrity, ECM components and immunogenicity of decellularized laryngeal scaffold with preserved cartilage. Biomat. 34: 1790–1798.
Kiselevsky, M. V., N. Y. Anisimova, and O. V. Lebedinskaya (2011) Optimization of a method for preparation and repopulation of the tracheal matrix for allogenic transplantation. Bull Exp. Biol. Med. 151: 107–113.
Kisilevski, M. V., A. Niu, and O. V. Lebedinskaia (2012) Heterotopic transplantation of non-immunogenic trachea populated with recipient bone marrow stromal cells. Morfologiia 141: 66–70.
Zang, M., Q. Zhang, and E. I. Chang (2012) Decellularized tracheal matrix scaffold for tissue engineering. Plast. Reconstr. Surg. 130: 532–540.
Luo, X., Y. Liu, and Z. Zhang (2013) Long-term functional reconstruction of segmental tracheal defect by pedicled tissueengineered trachea in rabbits. Biomat. 34: 3336–3344.
Kalathur, M., S. Baiguera, and P. Macchiarini (2010) Translating tissue-engineered tracheal replacement from bench to bedside. Cell Mol. Life Sci. 67: 4185–4196.
Baiguera, S., P. Jungebluth, and A. Burns (2010) Tissue engineered human tracheas for in vivo implantation. Biomat.31: 8931–8938.
Jungebluth, P., T. Go, and A. Asnaghi (2009) Structural and morphologic evaluation of a novel detergent-enzymatic tissue-engineered tracheal tubular matrix. J. Thorac. Cardiovasc. Surg. 138: 586–593.
Macchiarini, P., P. Jungebluth, and T. Go (2008) Clinical transplantation of a tissue-engineered airway. Lancet 372: 2023–2030.
Jungebluth, P., A. Bader, and S. Baiguera (2012) The concept of in vivo airway tissue engineering. Biomat. 33: 4319–4326.
Baiguera, S., A. Gonfiotti, and M. Jaus (2011) Development of bioengineered human larynx. Biomat. 32: 4433–4442.
Partington, L., N. J. Mordan, and C. Mason (2013) Biochemical changes caused by decellularization may compromise mechanical integrity of tracheal scaffolds. Acta Biomater. 9: 5251–5261.
Hinderer, S., M. Schesny, and A. Bayrak (2012) Engineering of fibrillar decorin matrices for a tissue-engineered trachea. Biomat. 33: 5259–5266.
Vogel, G. (2013) Trachea transplants test the limits. Sci. 19: 340: 266–268.
Haykal, S., J. P. Soleas, and M. Salna (2012) Evaluation of the structural integrity and extracellular matrix components of tracheal allografts following cyclical decellularization techniques: Comparison of three protocols. Tissue Eng. Part C Methods18: 614–623.
Bhosale, A. M. and J. B. Richardson (2008) Articular cartilage: Structure, injuries and review of management. Br. Med. Bull. 87: 77–95.
van Kasteren, S. I., H. Overkleeft, and H. Ovaa (2014) Chemical biology of antigen presentation by MHC molecules. Curr. Opin. Immunol. 26: 21–31.
Alonso Arias, R., A. López-Vázquez, and C. López-Larrea (2012)Immunology and the challenge of transplantation. Adv. Exp. Med. Biol. 741: 27–43.
Figueiredo, C., D. Wedekind, and T. Müller (2013) MHC universal cells survive in an allogeneic environment after incompatible transplantation. Biomed. Res. Int. 2013: 7960–6.
Wang, E. C., E. J. Damrose, and A. H. Mendelsohn (2006) Distribution of class I and II human leukocyte antigens in the larynx. Otolaryngol. Head Neck Surg. 134: 280–287.
Huey, D. J., J. Sanchez-Adams, and V. P. Willard (2012) Immunogenicity of bovine and leporine articular chondrocytes and meniscus cells. Tissue Eng. Part A 18: 568–575.
Sykes, M. (2010) Immune evasion by chimeric trachea. N. Engl. J. Med. 362: 172–174.
Bujia, J., E. Wilmes, and C. Hammer (1991) Class II antigenicity of human cartilage: Relevance to the use of homologous cartilage graft for reconstructive surgery. Ann. Plast. Surg. 26: 541–543.
Langer, F. and A. E. Gross (1974) Immunogenicity of allograft articular cartilage. J. Bone Joint Surg. Am. 56: 297–304.
Delaere, P., J. Vranckx, and G. Verleden (2010) Tracheal allotransplantation after withdrawal of immunosuppressive therapy. N. Engl. J. Med. 362: 138–145.
Crapo, P. M., T. W. Gilbert, and S. F. Badylak (2011) An overview of tissue and whole organ decellularization processes. Biomat. 32: 3233–3243.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pan, S., Sun, F., Shi, H. et al. Evaluation of an immune-privileged scaffold for In vivo implantation of tissue-engineered trachea. Biotechnol Bioproc E 19, 925–934 (2014). https://doi.org/10.1007/s12257-014-0150-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-014-0150-1