Abstract
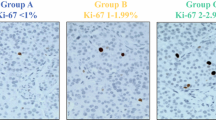
We investigated the expression patterns of Ki67 and p53 in metastatic pancreatic adenocarcinomas and analyzed their relationship with disease progression-free survival (PFS) and overall survival (OS) in the overall study population and in patients treated with a gemcitabine-containing chemotherapy versus FOLFIRINOX chemotherapy. Patients with histologically confirmed stage IV adenocarcinoma of the pancreas treated at AUBMC were included after obtaining institutional review board approval (IRB ID: IM.ST.05). The ROC was plotted to identify the threshold Ki-67, p53 and CA19–9 value for disease progression, the identified value was further used in Kaplan Meier curves to compare PFS for both groups (gemcitabine versus FOLFIRINOX). A value of p < 0.05 was considered significant in all analyses. On univariate analysis, patients who had a Ki-67 > 12.5% or a p53 > 15% had significantly shorter PFS (p = 0.034 and p = 0.016, respectively). This effect was restricted to Gemcitabine or gemcitabine-combination treated patients. A decrease in CA19–9 levels 6–8 weeks after chemotherapy of >58% had significantly longer PFS (p = 0.027). On multivariate analysis after controlling for grade, age and P53, Ki-67 remained significant, for every one unit increase in Ki-67 the progression risk increases by 1.017 times. Our study highlights the negative impact of high P53 expression and Ki67 proliferation index on PFS in patients with metastatic pancreatic cancer.






Similar content being viewed by others
References
Siegel R, Ma J, Zou Z, Jemal A (2014) Cancer statistics, 2014. CA Cancer J Clin 64(1):9–29. https://doi.org/10.3322/caac.21208
Saiki Y, Horii A (2014) Molecular pathology of pancreatic cancer. Pathol Int 64(1):10–19. https://doi.org/10.1111/pin.12114
Shimamura A, Fisher DE (1996) p53 in life and death. Clin Cancer Res 2(3):435–440
Striefler JK, Sinn M, Pelzer U, Juhling A, Wislocka L, Bahra M, Sinn BV, Denkert C, Dorken B, Oettle H, Riess H, Blaker H, Lohneis P (2016) P53 overexpression and Ki67-index are associated with outcome in ductal pancreatic adenocarcinoma with adjuvant gemcitabine treatment. Pathol Res Pract 212(8):726–734. https://doi.org/10.1016/j.prp.2016.06.001
Scholzen T, Gerdes J (2000) The Ki-67 protein: from the known and the unknown. J Cell Physiol 182(3):311–322. https://doi.org/10.1002/(SICI)1097-4652(200003)182:3<311::AID-JCP1>3.0.CO;2-9
Jalava P, Kuopio T, Juntti-Patinen L, Kotkansalo T, Kronqvist P, Collan Y (2006) Ki67 immunohistochemistry: a valuable marker in prognostication but with a risk of misclassification: proliferation subgroups formed based on Ki67 immunoreactivity and standardized mitotic index. Histopathology 48(6):674–682. https://doi.org/10.1111/j.1365-2559.2006.02402.x
Gentile V, Vicini P, Giacomelli L, Cardillo MR, Pierangeli A, Degener AM (2006) Detection of human papillomavirus DNA, p53 and ki67 expression in penile carcinomas. Int J Immunopathol Pharmacol 19(1):209–215
Lee CS (1996) Differences in cell proliferation and prognostic significance of proliferating cell nuclear antigen and Ki-67 antigen immunoreactivity in in situ and invasive carcinomas of the extrahepatic biliary tract. Cancer 78(9):1881–1887
Hruban RH, Iacobuzio-Donahue C, Wilentz RE, Goggins M, Kern SE (2001) Molecular pathology of pancreatic cancer. Cancer J 7(4):251–258
Wang B, Matsuoka S, Carpenter PB, Elledge SJ (2002) 53BP1, a mediator of the DNA damage checkpoint. Science 298(5597):1435–1438. https://doi.org/10.1126/science.1076182
Smith RA, Tang J, Tudur-Smith C, Neoptolemos JP, Ghaneh P (2011) Meta-analysis of immunohistochemical prognostic markers in resected pancreatic cancer. Br J Cancer 104(9):1440–1451. https://doi.org/10.1038/bjc.2011.110
Bold RJ, Hess KR, Pearson AS, Grau AM, Sinicrope FA, Jennings M, McConkey DJ, Bucana CD, Cleary KR, Hallin PA, Chiao PJ, Abbruzzese JL, Evans DB (1999) Prognostic factors in resectable pancreatic cancer: p53 and bcl-2. J Gastrointest Surg 3(3):263–277
Dergham ST, Dugan MC, Joshi US, Chen YC, Du W, Smith DW, Arlauskas P, Crissman JD, Vaitkevicius VK, Sarkar FH (1997) The clinical significance of p21(WAF1/CIP-1) and p53 expression in pancreatic adenocarcinoma. Cancer 80(3):372–381
Nio Y, Dong M, Uegaki K, Hirahara N, Minari Y, Sasaki S, Takamura M, Iguchi C, Tamura K (1999) Comparative significance of p53 and WAF/1-p21 expression on the efficacy of adjuvant chemotherapy for resectable invasive ductal carcinoma of the pancreas. Pancreas 18(2):117–126
Nio Y, Dong M, Iguchi C, Yamasawa K, Toga T, Itakura M, Tamura K (2001) Expression of Bcl-2 and p53 protein in resectable invasive ductal carcinoma of the pancreas: effects on clinical outcome and efficacy of adjuvant chemotherapy. J Surg Oncol 76(3):188–196
Makinen K, Hakala T, Lipponen P, Alhava E, Eskelinen M (1998) Clinical contribution of bcl-2, p53 and Ki-67 proteins in pancreatic ductal adenocarcinoma. Anticancer Res 18(1B):615–618
DiGiuseppe JA, Hruban RH, Goodman SN, Polak M, van den Berg FM, Allison DC, Cameron JL, Offerhaus GJ (1994) Overexpression of p53 protein in adenocarcinoma of the pancreas. Am J Clin Pathol 101(6):684–688
Ahrendt SA, Brown HM, Komorowski RA, Zhu YR, Wilson SD, Erickson BA, Ritch PS, Pitt HA, Demeure MJ (2000) p21WAF1 expression is associated with improved survival after adjuvant chemoradiation for pancreatic cancer. Surgery 128(4):520–530. https://doi.org/10.1067/msy.2000.108052
Vitellius C, Eymerit-Morin C, Luet D, Fizanne L, Foubert F, Bertrais S, Rousselet MC, Caroli-Bosc FX (2017) Relationship between the expression of O6-Methylguanine-DNA methyltransferase (MGMT) and p53, and the clinical response in metastatic pancreatic adenocarcinoma treated with FOLFIRINOX. Clin Drug Investig 37:669–677. https://doi.org/10.1007/s40261-017-0522-3
Galmarini CM, Clarke ML, Falette N, Puisieux A, Mackey JR, Dumontet C (2002) Expression of a non-functional p53 affects the sensitivity of cancer cells to gemcitabine. Int J Cancer 97(4):439–445
Fiorini C, Cordani M, Padroni C, Blandino G, Di Agostino S, Donadelli M (2015) Mutant p53 stimulates chemoresistance of pancreatic adenocarcinoma cells to gemcitabine. Biochim Biophys Acta 1853(1):89–100. https://doi.org/10.1016/j.bbamcr.2014.10.003
Venkatasubbarao K, Peterson L, Zhao S, Hill P, Cao L, Zhou Q, Nawrocki ST, Freeman JW (2013) Inhibiting signal transducer and activator of transcription-3 increases response to gemcitabine and delays progression of pancreatic cancer. Mol Cancer 12(1):104. https://doi.org/10.1186/1476-4598-12-104
Ko AH, Hwang J, Venook AP, Abbruzzese JL, Bergsland EK, Tempero MA (2005) Serum CA19-9 response as a surrogate for clinical outcome in patients receiving fixed-dose rate gemcitabine for advanced pancreatic cancer. Br J Cancer 93(2):195–199. https://doi.org/10.1038/sj.bjc.6602687
Halm U, Schumann T, Schiefke I, Witzigmann H, Mossner J, Keim V (2000) Decrease of CA 19-9 during chemotherapy with gemcitabine predicts survival time in patients with advanced pancreatic cancer. Br J Cancer 82(5):1013–1016. https://doi.org/10.1054/bjoc.1999.1035
Ziske C, Schlie C, Gorschluter M, Glasmacher A, Mey U, Strehl J, Sauerbruch T, Schmidt-Wolf IG (2003) Prognostic value of CA 19-9 levels in patients with inoperable adenocarcinoma of the pancreas treated with gemcitabine. Br J Cancer 89(8):1413–1417. https://doi.org/10.1038/sj.bjc.6601263
Saad ED, Machado MC, Wajsbrot D, Abramoff R, Hoff PM, Tabacof J, Katz A, Simon SD, Gansl RC (2002) Pretreatment CA 19-9 level as a prognostic factor in patients with advanced pancreatic cancer treated with gemcitabine. Int J Gastrointest Cancer 32(1):35–41. https://doi.org/10.1385/IJGC:32:1:35
Conroy T, Desseigne F, Ychou M, Bouché O, Guimbaud R, Bécouarn Y, Adenis A, Raoul J-L, Gourgou-Bourgade S, de la Fouchardière C, Bennouna J, Bachet J-B, Khemissa-Akouz F, Péré-Vergé D, Delbaldo C, Assenat E, Chauffert B, Michel P, Montoto-Grillot C, Ducreux M (2011) FOLFIRINOX versus gemcitabine for metastatic pancreatic Cancer. N Engl J Med 364(19):1817–1825. https://doi.org/10.1056/NEJMoa1011923
Acknowledgements
This work has been funded by the Seed grant from the American University of Beirut.
Funding
This study was funded by the Seed grant from the American University of Beirut.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of Interest
The authors report no direct conflicts of interest pertaining to the work. Other conflicts of interest not related to this work are as follows. ST reports receiving payment for development of educational presentations from Merck and travel support from MSD, Amgen and Roche. AS reports having received research grants, honorarium or educational support from Sanofi, Roche, Merck, Bristol Meyer, Amgen, Novartis, MSD and Piere Fabre. DM reports having received honoraria or travel support from Roche, Amgen, Pfizer, MSD and Novartis. MC reports having received research funding from GSK. AT reports having received research grant from Merck serono, travel support from MSK and Roche and consultancy from Pfizer, MSK, Lilly and Novartis. HA reports having received honoraria from Roche, Merck, MSD and AstraZeneca. For the remaining authors none were declared.
Rights and permissions
About this article
Cite this article
Temraz, S., Shamseddine, A., Mukherji, D. et al. Ki67 and P53 in Relation to Disease Progression in Metastatic Pancreatic Cancer: a Single Institution Analysis. Pathol. Oncol. Res. 25, 1059–1066 (2019). https://doi.org/10.1007/s12253-018-0464-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12253-018-0464-y