Abstract
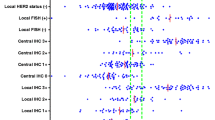
The correct identification of HER2-positive cases is a key point to provide the most appropriate therapy to breast cancer (BC) patients. We aimed at investigating the reproducibility and accuracy of HER2 expression by immunohistochemistry (IHC) in a selected series of 35 invasive BC cases across the pathological anatomy laboratories in Tuscany, Italy. Unstained sections of each BC case were sent to 12 participating laboratories. Pathologists were required to score according to the Food and Drug Administration (FDA) four-tier scoring system (0, 1+, 2+, 3+). Sixteen and nineteen cases were HER2 non-amplified and amplified respectively on fluorescence in situ hybridization. Among 192 readings of the 16 HER2 non-amplified samples, 153 (79.7 %) were coded as 0 or 1+, 39 (20.3 %) were 2+, and none was 3+ (false positive rate 0 %). Among 228 readings of the 19 HER2 amplified samples, 56 (24.6 %) were scored 0 or 1+, 79 (34.6 %) were 2+, and 93 (40.8 %) were 3+. The average sensitivity was 75.4 %, ranging between 47 % and 100 %, and the overall false negative rate was 24.6 %. Participation of pathological anatomy laboratories performing HER2 testing by IHC in external quality assurance programs should be made mandatory, as the system is able to identify laboratories with suboptimal performance that may need technical advice. Updated 2013 ASCO/CAP recommendations should be adopted as the widening of IHC 2+ “equivocal” category would improve overall accuracy of HER2 testing, as more cases would be classified in this category and, consequently, tested with an in situ hybridisation method.


Similar content being viewed by others
References
Popescu NC, King CR, Kraus MH (1989) Localization of the human erbB-2 gene on normal and rearranged chromosomes 17 to bands q12-21.32. Genomics 4:362–366
Perez EA, Cortes J, Gonzalez-Angulo AM, JMS et al (2014) HER2 testing: current status and future directions. Cancer Treat Rev 40:276–284
Hanna WM, Ruschoff J, Bilous M et al (2014) HER2 in situ hybridization in breast cancer: clinical implications of polysomy 17 and genetic heterogeneity. Mod Pathol 27:4–18
Wolff AC, Hammond EH, Schwartz JN et al (2007) American Society of Clinical Oncology/College of American Pathologists guideline recommendations for human epidermal growth factor receptor 2 testing in breast cancer. J Clin Oncol 25:118–145
Provenzano E, Johnson N (2009) Overview of recommendations of HER2 testing in breast cancer. Diagn Histopathol 15:478–484
Walker RA, Bartlett JM, Dowsett M et al (2008) HER2 testing in UK: further update to recommendations. J Clin Pathol 61:818–824
Wolff AC, Hammond EH, Hicks DG et al (2013) Recommendations for human epidermal growth factor receptor 2 testing in breast cancer: American Society of Clinical Oncology/College of American Pathologists clinical practice guideline update. J Clin Oncol 31:3997–4013
Hanna W, O’Malley FP, Barnes P et al (2007) Updated recommendations from the Canadian National Consensus Meeting on HER2/neu testing in breast cancer. Curr Oncol 4:149–153
Paik S, Bryant J, Tan-Chiu E et al (2002) Real-world performance of HER2 testing-National Surgical adjuvant Breast and Bowel Project Experience. J Natl Cancer Inst 94:852–854
Vogel CL, Bloom K, Burris H et al (2011) Discordance between central and local laboratory Her2 testing from a large HER2-negative population in VIRGO, a metastatic breast cancer registry. Cancer Res 71(24):188s
Reiner-Concin A (2008) External quality assurance in immunohistochemistry-is it the solution to a complex problem? Breast Care 3:78–79
Mirlacher M, Kasper M, Storz M et al (2004) Influence of slide aging on results of translational research studies using immunohistochemistry. Mod Pathol 17:1414–1420
Marchiò C, Lambros MB, Gugliotta P et al (2008) Does chromosome 17 centromere copy number predict polysomy in breast cancer? A fluorescence in situ hybridization and microarray-based CGH analysis. J Pathol 219:16–24
Vance GH, Barry TS, Bloom KJ et al (2009) Genetic heterogeneity in HER2 testing in breast cancer: panel summary and guidelines. Arch Pathol Lab Med 133:611–612
Vandem Bempt I, Van Loo P, Drijkoningen M et al (2008) Polysomy 17 in breast cancer: clinicopathologic significance and impact on HER-2 testing. J Clin Oncol 26:4869–4874
Cohen J (1968) Weighted kappa: nominal scale agreement with provision for scaled disagreement or partial credit. Psychol Bull 70:213–220
Landis JR, Koch GG (1977) The measurement of observer agreement for categorical data. Biometrics 33:159–174
Ellis IO, Dowsett M, Bartlett J et al (2000) Recommendations for HER2 testing in the UK. J Clin Pathol 53:890–892
von Wasielewski R, Krusche CA, Ruschoff J et al (2008) Implementation of external quality assurance trials for immunohistochemically determined breast cancer biomarkers in Germany. Breast Care 3:128–133
Schrohl AS, Pedersen HC, Jonsen SS et al (2011) Human epidermal growth factors receptors 2 (HER2) immunoreactivity: specificity of three pharmacodiagnostic antibodies. Histopathology 59:975–983
Leong TY-M, Cooper K, Leong AS-Y (2010) Immnunohistology - past, present and future. Adv Anat Pathol 17:404–418
Reiner-Concin A, Regitnig P, Dinges HP et al (2008) Practice of HER2 immunohistochemistry in breast carcinoma in Austria. Pathol Oncol Res 14:253–259
Bartlett JM, Going JJ, Mallon EA et al (2001) Evaluating HER2 amplification and overexpression in breast cancer. J Pathol 195:422–428
Rhodes A, Jasani B, Anderson E et al (2002) Evaluation of HER-2/neu immunohistochemical assay sensitivity and scoring on formalin-fixed and paraffin-processed cell lines and breast tumors: a comparative study involving results from laboratories in 21 countries. Am J Clin Pathol 118:408–417
Sauter G, Lee J, Bartlett JMS et al (2009) Guidelines for human epidermal growth factor receptor 2 testing: biologic and methodologic considerations. J Clin Oncol 27:1323–1333
Acknowledgments
We acknowledge the pathologists participating in the Tuscany Breast Cancer Study: P. Collecchi (AOU Pisana, Pisa), G. Giustarini (Nuovo Ospedale San Giuseppe-Empoli), L. Martini (Ospedale Versilia-Lido di Camaiore), T. Megha (AOU Senese-Siena), S. Tozzini (Ospedale Cosma e Damiano-Pistoia), C. Sabò (Ospedale Campo di Marte-Lucca), A. Carnevali (Ospedale San Donato-Arezzo), G. Tinacci (Ospedale Santa Maria Annunziata-Firenze), M. Stumpo (Ospedale Misericordia-Grosseto), M. Culli (Ospedale Civico-Carrara), V. Vezzosi (AOU Careggi-Florence), F. Zolfanelli (Ospedale San Giovanni di Dio-Florence).
We also thank Mrs C. Lucarelli and Mrs M. Pepi (Section of Pathological Anatomy-University of Florence) for technical skill.
Conflicts of Interest Statement
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Consortia
Corresponding author
Rights and permissions
About this article
Cite this article
Bianchi, S., Caini, S., Paglierani, M. et al. Accuracy and Reproducibility of HER2 Status in Breast Cancer Using Immunohistochemistry: A Quality Control Study in Tuscany Evaluating the Impact of Updated 2013 ASCO/CAP Recommendations. Pathol. Oncol. Res. 21, 477–485 (2015). https://doi.org/10.1007/s12253-014-9852-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12253-014-9852-0