Abstract
Purpose
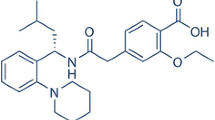
The objective of the present study was to develop HP-β-cyclodextrin inclusion complex-based rapid orally disintegrating tablets of pitavastatin calcium by unique sublimation technique for enhancing solubility and dissolution profile of anti-lipidemic class II drug.
Methods
The inclusion complex was prepared by a physical mixing method containing HP-β-cyclodextrin and pitavastatin calcium. The inclusion complex formation between the guest and host was confirmed by molecular docking studies. The developed inclusion complex was further characterized using differential scanning calorimetry (DSC), Fourier transform-infrared spectroscopy (FT-IR), powder X-ray diffraction study (PXRD), and scanning electron microscopy studies. Fast orally disintegrating tablets of pitavastatin calcium inclusion complex were developed by a unique sublimation technique employing full factorial design (24) using the Design Expert® software. Independent factors, namely, type of super disintegrants and sublimating agents at varying concentrations, were studied for effect on disintegration time, hardness of the tablet, and in vitro drug release.
Results
Molecular docking studies showed a score of −8.08. Optimized formulation containing 5% Ac-di-sol, 10% camphor showed hardness of about 3.37 ± 0.11 kg/cm2, least disintegration time 15.31 ± 0.32 s, and highest in vitro drug release of 99.11 ± 0.23% at the end of 15 min.
Conclusion
The employed complexation method enhanced solubility and increased in vitro dissolution of pitavastatin calcium. It was concluded that fast orally disintegrating tablets containing inclusion complex gave desired characteristics which provided rapid onset of action by fast disintegration and could improve patient compliance.














Similar content being viewed by others
References
Pereva S, Sarafska T, Bogdanova S. Spassov: Efficiency of “cyclodextrin-ibuprofen” inclusion complex formation. J Drug Delivery Sci Technol. 2016;35:34–9.
Aungst BJ. Novel formulation strategies for improving oral bioavailability of drugs with poor membrane permeation or presystemic metabolism. J Pharm Sci. 1993;82(10):979–87.
Uekama K, Hirayama F, Irie T. Cyclodextrin drug carrier systems. Chem Rev. 1998. https://doi.org/10.1021/cr970025p.
Rajewski R. Stella V: Pharmaceutical applications of cyclodextrins. 2. in vivo drug delivery. J Pharm Sci. 1996;85:1142–69.
Del Valle EM. Cyclodextrins and their uses: a review. Process Biochem. 2004;39:1033–46.
Suvarna V, Kajwe A, Manikanta M, Gurubasavaraj VP, Bharathkumar I, Sherje A. Inclusion complexes of nateglinide with HP–beta–CD and L-arginine for solubility and dissolution enhancement: preparation, characterization, and molecular docking study. J Pharm Innov. 2017;12:168–81.
Fu Y, Yang S, Jeong SH, Kimura S, Park K. Orally fast disintegrating tablets: developments, technologies, taste-masking and clinical studies. Crit Rev Ther Drug Carrier Syst. 2004. https://doi.org/10.1615/CritRevTherDrugCarrierSyst.v21.i6.10.
Mizumoto T, Masuda Y, Yamamoto T, Yonemochi E, Terada K. Formulation design of a novel fast-disintegrating tablet. Int J Pharm. 2005. https://doi.org/10.1016/j.ijpharm.2005.09.009.
Zade PS, Kawtikwar PS, Sakarkar DM. Formulation, evaluation and optimization of fast dissolving tablet containing tizanidine hydrochloride. Inter J Pharm Tech Res. 2009;1:34–42.
Parkash V, Maan S, Deepika, Yadav S, Hemlata, Jogpal V. Fast disintegrating tablets: opportunity in drug delivery system. J Adv Pharm Technol Res. 2011. https://doi.org/10.4103/2231-4040.90877.
Pitavastatin. Drugbank online. 2013. https://go.drugbank.com/drugs/DB08860. Accessed 16 Dec 2021.
Higuchi T, Connors KA. Phase solubility techniques. Adv Anal ChemInstrum. 1965;4:117–212.
Saokham P, Muankaew C, Jansook P, Loftsson T. Solubility of cyclodextrins and drug/cyclodextrin complexes. Molecules. 2018;23:1161.
De Miranda C, Azevedo Martins E, Veiga F, Humberto F. Cyclodextrins and ternary complexes: technology to improve solubility of poorly soluble drugs. Brazilian J Pharm Sci. 2011;47:665–81.
Loh GOK, Tan YTF, Peh KK. Enhancement of norfloxacin solubility via inclusion complexation with β-cyclodextrin and its derivative hydroxypropyl-β-cyclodextrin. Asian J Pharm Sci. 2016;11(4):536–46.
Loftsson T, Hreinsdóttir D, Másson M. Evaluation of cyclodextrinsolubilization of drugs. Int J Pharm. 2005. https://doi.org/10.1016/j.ijpharm.2005.05.042.
Vakani SS, Kajwe A, Suvarna V, et al. Influence of auxiliary agents on solubility and dissolution profile of repaglinide with hydroxypropyl-β-cyclodextrin: inclusion complex formation and its solid-state characterization. J Incl Phenom Macrocycl Chem. 2015. https://doi.org/10.1007/s10847-015-0559-y.
Sallam NM, Sanad RA, Kharshoom RM, Zeneldin MA. Development of salbutamol sulphate sublingual films in pullulan matrix for enhanced bioavailability & clinical efficacy. Curr Drug Deliv. 2017. https://doi.org/10.2174/1567201813666161024161308.
Rao NGR, Kulkarni U, Kumar KR. Comparative study on effect of different subliming agents on fast dissolving tablets of chlorthalidone by vacuum drying technique. Int J Pharmaceut Sci Res. 2010;1(8):78–87.
Jagdale SC, et al. Selection of superdisintegrant for Famotidine rapidly disintegrating tablets. J Chem Pharm Res. 2010;2(2):65–72.
Mostafa M, Gardouh AR, Abogresha NM, Gad S. Factorial design, formulation, in vitro and in vivo evaluation of rapid orally disintegrating tablets prepared by sublimation technique using captopril as a model drug. J Drug Delivery Sci Technol. 2020. https://doi.org/10.1016/j.jddst.2020.101635.
Mostafa HF, Ibrahim MA, Sakr A. Development and optimization of dextromethorphan hydrobromide oral disintegrating tablets: effect of formulation and process variables. Pharm Dev Technol. 2013;18(2):454–63.
Nazmi M, Islam SMA, Bhuiyan MA, Reza MS. Effect of superdisintegrant and their mode of incorporation on disintegration time and release profile of carbamazepine from immediate release tablet. J App Pharm Sci. 2013;3(05):80–4.
Acknowledgements
The authors are grateful to the management for offering the requisite technical help to accomplish this study. The authors would like to thank MSN Laboratories Pvt. Ltd, Telangana for providing a gift sample of drug.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Pimple, P., Singh, P., Prabhu, A. et al. Development and Optimization of HP-β-CD Inclusion Complex-Based Fast Orally Disintegrating Tablet of Pitavastatin Calcium. J Pharm Innov 17, 993–1010 (2022). https://doi.org/10.1007/s12247-022-09661-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12247-022-09661-x