Abstract
The Terminos Lagoon Flora and Fauna Protection Area is the largest coastal lagoon in the southern Gulf of Mexico and has the largest population of bottlenose dolphins, Tursiops truncatus, reported in the region. Photo identification studies have reported permanent and seasonal residence of dolphins; however, how dolphins coexist ecologically is unknown. In the present study, from 17 dead specimens stranded on the coast of Isla del Carmen (barrier island) between 2014 and 2016, stable isotopes in the liver, heart, and muscle, which represent different time scales, were analyzed to determine residence through δ13C values and niche width and trophic coexistence through δ15N values. Differences between sexes (δ13C liver: p = 0.009; heart: p = 0.01) and age classes (δ15N muscle: p = 0.02) were found, which indicated trophic sexual segregation and changes in resource use during growth, respectively. The lack of significant differences in δ13C values indicated isotopic equilibrium between the tissues, indicating the possible permanent residence of 12 dolphins in Terminos Lagoon as a coastal ecotype (− 16‰) and a second group (five dolphins) as an oceanic ecotype (− 18‰). In conclusion, most dolphins stranded in the Terminos Lagoon are potential permanent residents of this habitat, with evidence of resource partitioning as a coexistence strategy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
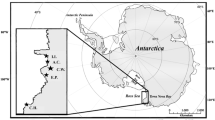
The Terminos Lagoon Flora and Fauna Protection Area is an aquatic coastal ecosystem located in the southern part of the Gulf of Mexico (Fig. 1). The lagoon is delimited by a barrier island: Carmen Island (30 km long and 2.5 km wide); thus, water exchanges with the sea take place through two inlets, one in the northeastern part and the other in the northwestern part (Villéger et al. 2010). Additionally, the lagoon has one of the largest populations of Tursiops truncatus in the gulf; the highest number of recaptures (residents) in the Yucatan Peninsula was recorded through photo identification (Delgado-Estrella 2015).
Within the lagoon, the places where dolphins are most frequently observed are Panlao, Puerto Pesquero, and areas close to river mouths (Martínez-Serrano et al. 2011), which are probably due to these areas containing favorable conditions as shelter sites for females with calves or multiple pods of dolphins. Dolphin movements are directly related to the availability of prey; thus, individuals may travel long distances in open water in search of food (these individuals are classified as the oceanic ecotype), whereas coastal ecotype individuals consume local prey populations in coastal ecosystems and travel short distances (Wells et al. 1980; Cockcroft and Ross 1990). These two ecotypes have been differentiated through morphological, ecological, and genetic differences and through stable isotope analysis (Wells and Scott 2009; Diaz-Gamboa et al. 2017).
In the Terminos Lagoon, studies of the abundance of dolphins indicate ranges between 800 and 1000 individuals per season. The peak abundance was recorded during the dry season (February to May), which was maintained until the rainy season (June to September) (Delgado-Estrella 2015; Fichez et al. 2017). Based on the results of the photo identification analysis, between 20 and 30% of the individuals were permanent residents of the lagoon, and these abundances are likely associated with the consumption of fishes from the families Ariidae (~ 50% between Ariopsis felis and Bagre marinus) and Sciaenidae (~ 40% among Cynoscion arenarius, C. nothus, and Micropogonias undulatus) according to stomach content analysis (Naranjo-Ruiz et al. 2019).
An ecosystem is made up of different microhabitats that generate specialized ecological niches for organisms. In odontocetes, interspecific specialization reflects differential uses of the niche (Guevara-Aguirre and Gallo-Reynoso 2016), whereas the coexistence of sympatric populations of the same species requires a certain degree of resource partitioning reflected in different feeding behaviors and intraspecific specializations (Loizaga de Castro et al. 2017); these ecological aspects have not been investigated in the T. truncatus population of the southern region of the Gulf of Mexico.
An excellent tool for investigating ecological aspects is stable isotopes, and the most frequently used are nitrogen (δ15N) and carbon (δ13C), which can be used to explore differences in the trophic ecology of sympatric populations (Post 2002; Fernández et al. 2011; Wilson et al. 2017) and to determine the origin of stranded carcasses (MacKenzie et al. 2011; Thomas et al. 2017). Isotopes can be assessed for predators assuming that the combination of both isotopes in predator tissues reflects assimilated food sources, with a difference known as isotopic fractionation (Hobson 1999; Fry 2006; Teixeira et al. 2022).
δ13C undergoes an approximate increase of 1 ‰ per trophic level, reflecting the primary production baseline in the food web and the productivity of the ecosystem being analyzed (Fry 2006). The most productive regions are located close to the coast and have high δ13C values, while the least productive areas are marine and offshore environments and are characterized by lower δ13C values (Graham et al. 2010). These differences have been recorded between the coastal and offshore ecotypes of dolphins, with variations between 1 and 2‰ in δ13C values (Michener and Kaufman 2007; Díaz-Gamboa et al. 2017).
For δ15N, an enrichment of ~ 3 to 4‰ is estimated for each trophic level, which indicates the trophic position since this isotope reflects the relative contributions of different trophic levels to the diet of animals, where inter- or intraspecific variations in the isotopic composition of organisms that are distributed in the same sites reflect a differential use of resources (Fry 2006).
Several authors mention that the liver has fast rates of isotopic incorporation, followed by the heart (days–week) and muscles (months) (MacNeil et al. 2005; Newsome et al. 2010; Vander-Zanden et al. 2015; Carter et al. 2019). Hence, the study of isotopes in more than one tissue explores different time scales and yields integrated information over different time periods or “isotopic clocks” (Carter et al. 2019). Moreover, if tissues with different incorporation rates exhibit isotopic equilibrium, that is, if they have the same isotopic signal, they may reflect a feeding site that does not change over time (Hobson 1999); this knowledge of dietary changes is ideal for use as a tool for monitoring potential residence.
It is known that different groups of dolphins use a particular habitat (δ13C) and food resources (δ15N) as a coexistence strategy. To investigate this phenomenon, organisms that stranded and died on the barrier island between the lagoon and the Gulf of Mexico from 2014 to 2016 were analyzed, the isotopic compositions of multiple tissues were analyzed, and the potential prey were analyzed for possible contributions to these tissues (Naranjo-Ruiz et al. 2019).
However, prior to the ecological analysis of a species and specifically when dealing with carcasses, it is necessary to assess the quality of the samples. Although the values of δ15N, in particular, are affected by the action of decomposer carcasses, no significant changes are detected in the first stages of decomposition until the corpse begins to swell (Payo-Payo et al. 2013; Keenan and DeBruyn 2019; Cloyed et al. 2022). Hence, up to that limit, tissues obtained from strandings can be used for isotope analysis.
This allows us to determine the potential residence represented by the low variability in δ13C among tissues and the coexistence strategy characterized by the high intraspecific variability in δ15N in one of the key species in the ecological balance of the Terminos Lagoon, the largest coastal natural protected area in the southern Gulf of México.
Materials and Methods
Study Area
Terminos Lagoon (Fig. 1), 18°20′—19°00′ N, 91°10′—92°00′ W, is a shallow estuarine system (1662 km2) with an average depth of 4 m. It is separated from the Gulf of Mexico by a barrier island (Isla del Carmen) that leaves two connections to the sea, each with a deep channel (Ramos-Miranda and Villalobos-Zapata 2015).
There are three distinct seasonal periods throughout the year: “dry” season from February to May and “rainy” season from June to September. These periods are separated by a period of winds from the northeast called “nortes” that extend through January (Fichez et al. 2017).
Stranding
Between 2014 and 2016, stranded bottlenose dolphins (T. truncatus) were examined in situ according to the protocol established by the government of Mexico for Strandings of Aquatic Mammals (DOF 2014): The geographical position, photographic evidence, and morphometric data were recorded. Samples of tissues, including muscle, liver, and heart, were frozen at − 15 °C. The maximum length of each dolphin was used to indicate its age class according to the criteria established for the Gulf of Mexico dolphins by Romero-Tenorio and Delgado-Estrella (2015).
Collection of Potential Prey Species
Between 2016 and 2017, 10-min intervals were deployed at a speed of 2 to 2.5 knots for each of the 17 sampling stations on a 5 m long shrimp trawl net (2.5 m opening, ¾″ mesh). The catch was classified by station and frozen in the Laboratory of Trophic Ecology of the EPOMEX Institute of the Universidad Autónoma de Campeche.
Twelve of the most abundant species of the Terminos Lagoon (Ramos-Miranda et al. 2005; Irola-Sansores et al. 2021) were selected from among the species captured; these included mollusks, crustaceans, and fishes from different trophic levels. The fishes were selected based on the best represented species in the stomach contents of dolphins in this region (Naranjo-Ruiz et al. 2019).
Isotope Analysis
Samples of the liver, muscle, and heart of T. truncatus and of the muscle of each prey species were dehydrated in an oven at a constant temperature of 55 °C for 48 h. Subsequently, each sample was macerated in an agate mortar to homogenize and reduce it and to be analyzed via a mass spectrometer of isotopic ratios coupled to an elemental analyzer of carbon and nitrogen (Laboratorio de Química Marina, Área de Espectrometría de Masas, CICIMAR, Instituto Politécnico Nacional).
Data Analysis
C:N Ratio and Mathematical Lipid Correction
The C:N ratio was used as an indicator of lipid influence in samples of dolphins. Some tissues can have large amounts of fat due to their metabolic nature. Tissues with C:N values higher than 3.5 were mathematically corrected to avoid the influence of lipids (depleted in δ13C) without damaging the δ15N composition (Post et al. 2007; Hardt et al. 2013 Giménez et al. 2017) in tissues of aquatic species using the equation proposed by Post et al. (2007):
Statistical Analysis
The stable isotope values of dolphins met the assumptions of normality (Shapiro-Wilk; δ15N W = 0.96, p = 0.22 and δ13C W = 0.97, p = 0.36) and homoscedasticity (Levene test: δ15N F = 0.97, p = 0.38 and δ13C F = 0.33, p = 0.71); therefore, parametric tests were performed with Statistica v7.0.6.1.0 (StatSoft, 2001). One-way ANOVA was used to determine whether the δ15N and δ13C values differed significantly among the tissue, sex, age class (adults, subadults, and calves), season and year of stranding (2014, 2015, and 2016), ecotype (coastal δ13C more than − 16‰, oceanic δ13C less than − 17‰), trophic position, and stranding area; where there were significant differences, the Tukey post hoc test was applied.
Isotopic Niche Width
To evaluate the coexistence, the stable Bayesian ellipse in R (SIBER) was used to determine the isotopic niche width and feeding strategy through ellipse width (lower variance: specialist vs. high variance: generalist), which is based on isotopic ratio measures calculated by a covariance matrix that defines the shape and area of the standard ellipse area corrected (SEAc) within a 95% confidence interval (Jackson et al. 2011). Values of SEAc ≤ 1 corresponded to predators with specialist habits, while values > 1 were considered generalists. Additionally, the overlap of the ellipses was measured to determine the possible differences between sexes, age classes, ecotypes, and stranding areas in terms of resource use and habitat; according to Langton (1982), values from 0 to 0.29 indicate low overlap, values from 0.30 to 0.59 correspond to a mean overlap, and values higher than 0.60 indicate high overlap between categories.
Use of Food Resources
Based on the analysis of the stomach contents of stranded dolphins (Naranjo-Ruiz et al. 2019) and the most abundant species in the Terminos Lagoon throughout the year (Irola-Sansores et al. 2021; Paz-Rios et al. 2022), stable isotope mixing model in R (SIMMR) was used to infer the relative contribution of potential prey species to consumers; in addition, the trophic discrimination factor (TDF), which is defined as the increase in the isotopic composition of the diet in the tissues of the predator (Fry 2006), was calculated by SIMMR to correct the isotopic values of the consumer (Parnell et al. 2013; Phillips et al. 2014). The proportion of each prey species was inferred from its isotopic signal under the assumption that the average isotopic signal represents 100% of the prey consumed.
Trophic Position
To determine if there are differences in the use of resources in the Terminos Lagoon, the trophic position (TP) of each dolphin was calculated according to the criteria and equation proposed by Post (2002):
where ʎ is the trophic position of the isotopic baseline, since Eucinostomus gula complies with the 3 criteria established by Post (2002): (1) is a very abundant species in the Terminos Lagoon in all seasons, (2) is part of the T. truncatus diet, and (3) has a trophic position of approximately 2 (Froese and Pauly 2018). Further, Δn is the value of fractionation in δ15N for muscle (3.4 Wilson et al. 2017), δ15Nconsumer is the δ15N value of T. truncatus, and δ15NBase is the baseline δ15N.
Results
Seventeen of the 35 dolphins stranded in the Terminos Lagoon between 2014 and 2016 were selected (44 samples: 15 from the liver, 16 from the heart, and 13 from the muscle) according to the degree of decomposition and the availability of tissues (Table 1). Six were female, ten were male, and the sex of one could not be determined. Six were calves, five were subadults, and six were adults. Six of the stranding events occurred during the dry season, three during the rainy season and eight during the nortes season. Two areas in Isla del Carmen were identified as the area of stranding, forming two groups according to the geographical area on the island: Boca del Carmen and the North Coast (Fig. 1). A total of 12 potential prey species (57 muscle samples) were analyzed: for the dry season, six prey species were analyzed (18 item samples); for the rainy season, seven prey species were analyzed (21 item samples); and for the nortes season, six prey species were analyzed (18 item samples) (Table 2).
C:N Ratio, Mathematical Lipid Correction, and Isotopic Composition
By calculating the C:N ratio, the δ13C data were adjusted for tissues that were not within the range proposed; the largest adjustment was for the liver (2.26‰); the adjustment was 0.83‰ for the heart and 0.29‰ for the muscle.
The isotopic composition, particularly δ15N, presented a constant decreasing trend among tissues (heart > liver > muscle; approximately 1‰). However, no significant differences were found for δ15N (F(2.41) = 2.87; p = 0.067) or δ13C (F(2.41) = 0.96; p = 0.39). The isotopic composition between the sexes indicated significant differences only for δ13C in the liver (F(1.12) = 9.64; p = 0.009) and heart (F(1.13) = 9.01; p = 0.01). Moreover, among the age classes, significant differences were found for δ15N only in muscle (F(2,10) = 5.22; p = 0.02), particularly between subadults and calves (MS = 0.70, df = 10.00).
There were no significant differences among seasons or years (p > 0.05). However, significant differences were found for δ13C in heart tissue (F(2.13) = 23.50; p = 0.00005), suggesting three groups of dolphins: Five were recognized as the oceanic ecotype (− 18.0 to − 17.0‰), five were recognized as the coastal ecotype (− 16.0‰), and a subgroup of seven dolphins was recognized as the “estuarine shoreline” ecotype (− 15.0‰). The heart tissue differed between the two areas where it was stranded: Boca del Carmen and the North Coast (δ15N F(1.14) = 5.78; p = 0.03).
Isotopic Niche Width
In general, the SEAc values were mostly for generalist eating habits (> 1), with the exception of the age class, where the values of the amplitude of the ellipse in muscle were from specialist feeding habits in subadults (SEAc = 0.61). The SIBER analysis showed that the ellipses had medium to low overlap (0–0.59) in the following categories: sex (Fig. 2), age class (Fig. 3), and ecotype (Fig. 4) as well as between the two stranding areas (Fig. 5), indicating the existence of different food groups in each category.
Trophic Position
A representative species in the Terminos Lagoon was E. gula, which had a δ15N value of 6.50‰, and its trophic position was used as the baseline (TP = 2.7). For dolphins, the trophic position varied between 4.2 and 5.6, with an average of 4.7 ± 0.3, and differed between the two stranding areas (ANOVA: F(1,18) = 7.4341, p = 0.01). The highest levels occurred in the North Coast, varying from 4.2 to 5.1, with an average of 4.5 ± 0.3; in contrast, in Boca del Carmen, the trophic position varied from 3.4 to 4.4, with an average of 4.1 ± 0.2.
Use of Food Resources
To detect seasonal differences in the proportions of prey consumed, the liver (Phillips et al. 2014; Vander-Zanden et al. 2015) and heart (because of the similar behavior of isotopic variation with the liver) are likely to be the most appropriate since they have a higher rate of isotopic turnover than muscle has.
The trophic discrimination factor (TDF) values were similar among tissues: 3.9 ± 0.1 for liver Δ15N and 1.1 ± 0.5 for Δ13C. For the heart, Δ15N was 3.8 ± 0.5, and Δ13C was 0.8 ± 0.3. For muscle, Δ15N was 3.9 ± 0.5, and Δ13C was 0.6 ± 0.0.
The prey contributions in muscle among the age classes were compared (Fig. 6). During the rainy season, the squid L. brevis made the greatest contribution to adults, subadults, and calves, with 37%, 37.7%, and 72.8%, respectively. In the nortes season, for adults, the greatest contribution was made by the blue crab C. similis (28.7%); for subadults and calves, the greatest contribution was made by the gafftopsail sea-catfish B. marinus (33.1% and 53.9%, respectively). During the dry season, the silver jenny mojarra E. argenteus had the greatest contribution to all age classes, with 33.4%, 28.8%, and 30.7% for adults, subadults, and juveniles, respectively.
The greatest contributions of prey to the heart tissue of dolphins (Fig. 7) stranded in Boca del Carmen during the rainy season were made by the mantis shrimp S. empusa (27.9%) and the squid L. brevis (22.5%). In the nortes and the dry seasons, the greatest contributions were made by the jenny mojarra E. gula (27.3%) and the silver jenny mojarra E. argenteus (57.8%), respectively. In the case of the North Coast, these dolphins received more specific contributions; for the rainy season, the major contribution was made by the squid L. brevis (59.3%); for the nortes season, the major contribution was made by the gafftopsail sea-catfish B. marinus (43.6%); and for the dry season, the major contribution was made by the silver jenny mojara E. argenteus (40.8%).
Discussion
Ecological studies of marine species that lack visible physical barriers are challenging (Hobson 1999; Fruet et al. 2017); the use of stable carbon and nitrogen isotopes can overcome this problem (Newsome et al. 2010). Intraspecific variations in stable isotopes are key in biological conservation because intraspecific and interindividual variations in stable isotopes reveal information about resource partitioning (Fernández et al. 2011; Parra et al. 2022) and residence (Hobson 2007).
C:N Ratio
Cetaceans are aquatic organisms that store large amounts of fat that help them maintain their temperature in the aquatic environment (Iverson 2009). A large amount of fat stored in tissues can be reflected by high C:N values (> 3.4). Previous studies have shown that the equation proposed by Post et al. (2007) is feasible for comparison of the results obtained from mathematical correction versus chemical lipid extraction, where the results are similar without damaging the nitrogen isotopic signal (Hardt et al. 2013; Giménez et al. 2017). In the present study, with dolphin tissues, the liver (a lipid reservoir: Reynolds and Rommel 2009) was shown to be a good fit after correction, so mathematical correction has been used as an effective way to avoid the influence of lipids.
Trophic Discrimination Factor
Discrimination or enrichment in isotopes is the result of assimilation and synthesis of the source: δ15N reflects the intake of protein in the diet and the excretion of endogenous nitrogen in the urine, and δ13C reflects the quality of the food (Post 2002) and is lost through respiration. In each case, the discrimination or enrichment in isotopes is measured as the difference between the isotopic value of the consumer’s body and the isotopic value of the diet.
The values obtained for the TDF of both isotopes did not differ among tissues, which allowed us to validate the isotopic equilibrium. T. truncatus in the Terminos Lagoon had TDF δ15N muscle values (Δ15N 3.9) similar to those found on the coasts of Florida (Δ15N 3.4; Wilson et al. 2017) and Galicia (Δ15N 3.3; Fernández et al. 2011); these similar values of Δ15N indicate that these coastal dolphins feed on prey of similar trophic levels in different parts of the world. Therefore, the TDF calculated in the present study is the first in the southeastern Gulf of Mexico and can be used as a reference point for future studies of dolphins in similar tropical coastal ecosystems; this approach could help to achieve a better fit in mixing models (Phillips et al. 2014) and in trophic position calculations.
Use of Multiple Tissues
Since the rate of isotopic turnover differs between tissues, the use of multiple tissues here provides more robust information on trophic dynamics at different time scales. Thus, the short-term differences are reflected in the values measured in the liver or the heart compared to the long-term differences shown by the muscle (MacNeil et al. 2005; Carter et al. 2019), which remain stable throughout the seasons. Several authors mention that isotope enrichment in consumers occurs during the assimilation of carbon and nitrogen from the diet; thus, the importance of knowledge enrichment among tissues allows us to provide the basis for a better understanding of dietary sources and trophic position (Chen et al. 2012).
In the present study, which included tissues from the dolphins T. truncatus, the enrichment of the heart above the other tissues was similar to the results obtained by Hobson et al. (1996), where the δ15N values obtained for the heart and liver of sea lions were the same at 16.1‰, while the muscle was less enriched at 15.4‰. Hussey et al. (2010) reported that the heart was more enriched in δ15N than other tissues, such as the liver and muscle. These differences can be related to the amino acid composition among tissues that can generate variations in isotopic composition (Newsome et al. 2010).
Isotopic Niche Width
In aquatic ecosystems, higher δ13C values indicate more coastal regions (> − 16‰) than oceanic regions (< − 18‰) (Graham et al. 2010; Newsome et al. 2010), which can be reflected in the isotopic composition of individuals (Barros et al. 2010; Díaz-Gamboa et al. 2017; Fernández et al. 2011; Wilson et al. 2017). In this study, the isotopic data of δ13C obtained from the tissues of dolphins functioned as a tool for inferring the unknown origin of stranded carcasses.
In this study, it was possible to statistically differentiate the three groups. The oceanic ecotype (− 18‰) presented the most negative values of δ13C, and the rest were considered coastal ecotypes (− 16‰); however, a subgroup of dolphins was named “coastal–shoreline” (− 15‰) due to its isotopic signal being even more enriched in δ13C than it was in other coastal individuals, and this subgroup was composed mainly of females. An important area for females in terms of breeding and feeding is the mouths of rivers (Secchi et al. 2016), such as the mouth of the Candelaria River and the Panlao Lagoon in the Terminos Lagoon (Delgado-Estrella 2002). These results are consistent with the sexual segregation behaviors observed (Morteo et al. 2014), indicating that this behavior occurs not only for genetic flow purposes but also in relation to food.
For the coastal ecotype, isotopic equilibrium indicates a home environment, verifying the potential residence and almost exclusive feeding site (Hobson 2007; Sinisalo et al. 2008; Torres-Rojas et al. 2014; Germain et al. 2011). Whereas those who presented slight variations in δ13C signals among tissues showed changes in feeding sites within the same lagoon, these changes occurred mainly in males, who are recognized for exhibiting broader movements in the ecosystems that they inhabit (Morteo et al. 2014). Hence, the Terminos Lagoon represents a valuable site for the feeding, sheltering, breeding, and growth of T. truncatus.
Trophic Position and Use of Food Resources
Each generalist population is composed of individual specialists who take advantage of a small portion of the niche (Bolnick et al. 2002); by analyzing the variations in the isotopic composition, mainly in δ15N, it can be inferred whether the eating habits have a broad or narrow spectrum (Bearhop et al. 2004; Jackson et al. 2011). In present study, it was determined that two groups of resident dolphins in a semienclosed body of water take advantage of the resources at different times and in different proportions. This finding supports niche theory, in which the distribution of resources is established as a coexistence strategy (Bearzi 2005; Loizaga de Castro et al. 2017).
The TP value 4.3 calculated in this study from the δ15N isotopic data coincided with those obtained by Naranjo-Ruiz et al. (2019), based on the analysis of the prey obtained from the stomach contents of T. truncatus in the Terminos Lagoon Flora and Fauna Protection Area. The trophic level obtained is that of a top predator (Cortés 1999).
The differences among age classes indicated changes in feeding throughout the growth of dolphins, as their energy demand varies across developmental stages (Secchi et al. 2016). These changes were reflected in the muscle, which is a tissue that undergoes greater turnover during the growth of the organisms (Newsome et al. 2010). Notably, the calves were above the level of adults and subadults; these values are common in calves because they were breastfeeding, which makes them become enriched with signals from their mothers (Fernández et al. 2011).
Subadults segregate themselves from herds and create associations with other juveniles (McHugh et al. 2011 and Morteo et al. 2014), stop obtaining food from their mothers (lactation), and practice hunting techniques and improvements, forcing them to reduce their dietary variety (Rossman et al. 2015). Knowledge of these supposed specializations is highly relevant because coastal dolphins become vulnerable populations, as they are subjected to habitat loss and competition for prey with humans, reducing their food source availability.
Differences in δ15N values in active tissues that reflect changes in short periods of time revealed that at least two subpopulations consumed the same prey species available but in different ways based on the prey species and their proportions in the tissues. Dolphins stranded in the North Coast area tended to take advantage of food resources in a more specific way, with contributions of specific prey species in the dolphin tissues each season, than did the dolphins of Boca del Carmen, which had similar contributions from various prey species throughout the seasons. These findings could be related to differences in prey availability and habitats in the Terminos Lagoon (Irola-Sansores et al. 2020; Paz-Rios et al. 2022). These variations in the proportions of the prey allow us to know how, despite having the same prey available, they distribute these resources at different times and in different proportions.
In the present study, it was found that most of the bottlenose dolphins that were stranded in the marine portion of Isla del Carmen were residents of the Terminos Lagoon. A subgroup of the coastal ecotype that mainly included the estuarine zones of the lagoon was identified. Resident dolphins presented sexual and trophic segregation, while by age class, the subadults exhibited specialist eating habits. Similarly, two groups of dolphin residents related to the stranding area in the island reflected a used different food resource in the lagoon. The importance of the Terminos Lagoon as a habitat where Tursiops truncatus plays an important role as a regulator of the ecosystem was verified, as it is a top predator that maintains balance in the functioning of the ecosystem.
References
Barros, B.N., P.H. Ostro, C.A. Stricker, and R.S. Wells. 2010. Stable isotopes differentiate bottlenose dolphins off west-central Florida. Marine Mammal Science 26 (2): 324–362. https://doi.org/10.1111/j.1748-7692.2009.00315.x.
Bearhop, S., C.E. Adams, S. Waldron, R.A. Fuller, and H. Macleod. 2004. Determining trophic niche width: A novel approach using stable isotope analysis. Journal of Animal Ecology 73 (5): 1007–1012. https://doi.org/10.1111/j.0021-8790.2004.00861.x.
Bearzi, M. 2005. Habitat partitioning by three species of dolphins in Santa Monica Bay, California. Bulletin, Southern California Academy of Sciences 104 (3): 113–124. https://doi.org/10.3160/0038-3872(2005)104[113:HPBTSO]2.0.CO;2.
Bolnick, D.I., L.H. Yang, J.A. Fordyce, J.M. Davis, and R. Svanbäck. 2002. Measuring individual-level resource specialization. Ecology 83 (10): 2936–2941. https://doi.org/10.1890/0012-9658(2002)083[2936:MILRS]2.0.CO;2.
Carter, W.A., U. Bauchinger, and S.R. McWilliams. 2019. The importance of isotopic turnover for understanding the aspects of animal ecology and nutrition. Diversity 11 (5): 84. https://doi.org/10.3390/d11050084.
Chen, G., H. Zhou, D. Ji, and B. Gu. 2012. Stable isotope enrichment in muscle, liver, and whole fish tissues of brown-marbled groupers (Eplinephelus fuscoguttatus) Ecological Processes. 1:7. https://doi.org/10.1186/2192-1709-1-7
Cloyed, C.S., C. Johnson, K.P. DaCosta, L.R. Clance, M.L. Russell, C. Díaz-Clark, E.E. Hieb, and R.H. Carmichael. 2022. Effects of tissue decomposition on stable isotope ratios and implications for use of stranded animals in research. Ecosphere. https://doi.org/10.1002/ecs2.4385.
Cockcroft, V.G., and G.J.B. Ross. 1990. Food and feeding of the Indian Ocean bottlenose dolphin off Southern Natal South Africa. 295–308. https://doi.org/10.1016/B978-0-12-440280-5.50019-6 In: Leatherwood, S., and R.R. Reeves. (Eds). The bottlenose dolphin. Academic Press. San Diego, California, E.E.U.U. 653.
Cortés, E. 1999. Standardized diet compositions and trophic levels of sharks. ICES Journal of Marine Science 56 (5): 707–717. https://doi.org/10.1006/jmsc.1999.0489.
Delgado-Estrella, A. 2002. Comparación de parámetros poblacionales de las toninas, Tursiops truncatus, en la región sureste del golfo de México (Estados de Tabasco, Campeche, Yucatán y Quintana Roo), 160. Tesis doctoral: Facultad de Ciencias, Universidad Nacional Autónoma de México.
Delgado-Estrella, A. 2015. Patrones de residencia y movimientos a largo plazo de las toninas Tursiops truncatus, en la región sureste del Golfo de México. Therya 6(2): 297–314. https://doi.org/10.12933/therya-15-265
Díaz-Gamboa, R.E., D. Gendron, and G. Busquets-Vass. 2017. Isotopic niche width differentiation between common bottlenose dolphin ecotypes and sperm whales in the Gulf of California. Marine Mammal Science 34 (2): 440–457. https://doi.org/10.1111/mms.12465.
DOF. 2014. Acuerdo mediante el cual se expide el Protocolo de atención para varamiento de mamíferos marinos. Available In: http://dof.gob.mx/nota_detalle.php?codigo=5348898&fecha=17/06/2014.
Fernández, R., S. García-Tiscar, M. Begoña-Santos, A. López, J.A. Martínez-Cedeira, J. Newton, and G.J. Pierce. 2011. Stable isotope analysis in two sympatric populations of bottlenose dolphins Tursiops truncatus: Evidence of resource partitioning? Marine Biology 158: 1043–1055. https://doi.org/10.1007/s00227-011-1629-3.
Fichez, R., D. Archundia, C. Grenz, P. Douillet, F. Gutiérrez-Mendieta, M. Origel-Moreno, L. Denis, A.C. Ruiz-Esparza, and J. Zavala-Hidalgo. 2017. Global climate change and local watershed management as potential drivers of salinity variation in a tropical coastal lagoon (Laguna de Términos, Mexico). Aquatic Sciences 79: 219–230. https://doi.org/10.1007/s00027-016-0492-1.
Froese, R., D. and Pauly. (Editors) (2018). FishBase. World Wide Web electronic publication. Disponible en: https://fishbase.mnhn.fr/search.php
Fruet, P.F., E.R. Secchi, J.C. Di-Tullio, P.C. Simões-Lopes, F. Daura-Jorge, A.P.B. Costa, E. Vermeulen, P.A.C. Flores, R.C. Genoves, P. Laporta, L.B. Beheregara, and L.M. Möller. 2017. Genetic divergence between two phenotypically distinct bottlenose dolphin ecotypes suggests separate evolutionary trajectories. Ecology and Evolution 7 (21): 9131–9143. https://doi.org/10.1002/ece3.3335.
Fry, B. 2006. Stable isotope ecology Springer-Verlag, New York, 308.
Germain, L.R., M.D. McCarthy, P.L. Koch, and J.T. Harvey. 2011. Stable carbon and nitrogen isotopes in multiple tissues of wild and captive harbor seals (Phoca vitulina) off the California coast. Marine Mammal Science 28 (3): 542–560. https://doi.org/10.1111/j.1748-7692.2011.00516.x.
Giménez, J., F. Ramirez, M.G. Forero, J. Almunia, R. Stephanis, and J. Navarro. 2017. Lipid effects on isotopic values in bottlenose dolphins ( Tursiops truncatus ) and their prey with implications or diet assessment. Marine Biology 164: 122. https://doi.org/10.1007/s00227-017-3154-5.
Graham, B.S., P.L. Koch, S.D. Newsome, K.W. McMahon, and D. Aurioles. 2010. Using isoscapes to trace the movements and foraging behavior of top predators in oceanic ecosystems. Chapter 14. In: West, J., B.G. Bowen, T. Dawson, and K. Tu. (eds). Isoscapes: understanding movement, pattern, and process on earth through isotope mapping, Springer, Dordrecht. https://doi.org/10.1007/978-90-481-3354-3_14
Guevara-Aguirre, D., and J.P. Gallo-Reynoso. 2016. Uso de hábitat de dos ecotipos de toninas (Tursiops truncatus) en el golfo de California, México. Revista Mexicana De Biodiversidad 87: 1045–1054. https://doi.org/10.1016/j.rmb.2016.07.015.
Hardt, F.A.S., M.J. Cremer, A.J. Tonello-Junior, A. Bellante, G. Buffa, G. Buscaino, S. Mazzola, A.S. Barreto, L.A. Martinelli, and G.M. Zuppi. 2013. Use of carbon and nitrogen stable isotopes to study the feeding ecology of small coastal cetacean populations in southern Brazil. Biota Neotropica 13(4). https://doi.org/10.1590/S1676-06032013000400009
Hobson, K.A. 1999. Tracing origins and migration of wildlife using stable isotopes: A review. Oecologia 120: 314–326. https://doi.org/10.1007/s004420050865.
Hobson, K.A., D.M. Schell, D. Renouf, and E. Noseworthy. 1996. Stable carbon and nitrogen isotopic fractionation between diet and tissues of captive seals: Implications for dietary reconstructions involving marine mammals. Canadian Journal of Fisheries and Aquatic Sciences 53 (3): 528–533. https://doi.org/10.1139/f95-209.
Hobson, K.A. 2007. Chapter 6: isotopic tracking of migrant wildlife. In: Michener, R., and K. Lajtha. (Eds). Stable isotopes in ecology and environmental science. Second edition.
Hussey, N.E., J. Brush, I.D. McCarthy, and A.T. Fisk. 2010. δ15N and δ13C diet–tissue discrimination factors for large sharks under semi-controlled conditions. Elsevier. Comparative Biochemistry and Physiology, Part A 155: 445–453. https://doi.org/10.1016/j.cbpa.2009.09.023.
Irola-Sansores, B.N., Y.E. Torres-Rojas, and A. Cuevas-Jiménez. 2021. Spatial-temporal behavior of benthic communities in the Terminos Lagoon, Mexico: Possible areas of protection and conservation. JAINA Costas y Mares Ante El Cambio Climático 3 (1): 55–76. https://doi.org/10.26359/52462.0421.
Iverson, S.J. 2009. Blubber. In Encyclopedia of marine mammals (second edition), ed. W.F. Perrin, B. Würsig, and J.G.M. Thewissen, 1295p. E.E.U.U: Academic Press.
Jackson, A.L., R. Inger, A.C. Parnell, and S. Bearhop. 2011. Comparing isotopic niche widths among and within communities: SIBER – Stable Isotope Bayesian Ellipses in R. Journal of Animal Ecology 80 (3): 595–602. https://doi.org/10.1111/j.1365-2656.2011.01806.x.
Keenan, S.W., and J.M. DeBruyn. 2019. Changes to vertebrate tissue stable isotope (δ15N) composition during decomposition. Nature. Scientific Reports 9: 9929. https://doi.org/10.1038/s41598-019-46368-5.
Langton, R.W. 1982. Diet overlap between Atlantic cod, Gadus morhua, silver hake, Merluccius bilinearis, and fifteen other northwestern Atlantic finfish. Fishery Bulletin- National Oceanic and Atmospheric Administration 80 (4): 745–759.
Loizaga de Castro, R., F. Saporiti, D.G. Vales, L. Cardona, and E.A. Crespo. 2017. Using stable isotopes to assess whether two sympatric dolphin species share trophic resources. Marine Mammal Science 33 (4): 1235–1244. https://doi.org/10.1111/mms.12428.
MacKenzie, K.M., M.R. Palmer, A. Moore, A.T. Ibbotson, W.R.C. Beaumont, D.J.S. Poulter, and C.N. Trueman. 2011. Locations of marine animals revealed by carbon isotopes. Scientific Reports. 1: 21. https://doi.org/10.1038/srep00021.
MacNeil, A.M., G.B. Skomal, and A.T. Fisk. 2005. Stable isotopes from multiple tissues reveal diet switching in sharks. Marine Ecology Progress Series 302: 199–206. Available In: https://www.int-res.com/abstracts/meps/v302/p199-206/N1 . https://doi.org/10.3354/meps302199
Martínez-Serrano, I.A., G. Heckel, and Y. Schramm. 2011. Distribution and home range of bottlenose dolphins (Tursiops truncatus) off Veracruz, Mexico. Ciencias Marinas (37): 379–392. Available In: http://www.scielo.org.mx/scielo.php?script=sci_arttext&pid=S0185-38802011000400001&lng=es&nrm=iso
McHugh, K.A., J.B. Allen, A.A. Barleycorn, and R.S. Wells. 2011. Natal philopatry, ranging behavior, and habitat selection of juvenile bottlenose dolphins in Sarasota Bay. Florida. Journal of Mammalogy 92 (6): 1298–1313. https://doi.org/10.1644/11-MAMM-A-026.1.
Michener, R.H., and L. Kaufman. 2007. Chapter 9: stable isotope ratios as tracers in marine food webs: an update. In: Michener, R., and K. Lajtha. (Eds). Stable isotopes in ecology and environmental science. Second edition.
Morteo, E., A. Rocha-Olivares, and L.G. Abarca-Arenas. 2014. Sexual segregation of coastal bottlenose dolphins (Tursiops truncatus) in the southwestern Gulf of Mexico. Aquatic Mammals 40 (4): 375–385. https://doi.org/10.1578/AM.40.4.2014.375.
Naranjo-Ruiz, K.L., A. Delgado-Estrella, M.R.K. y Morquecho-León, Y.E. Torres-Rojas. 2019. Determinación de peces presas consumidos por toninas (Tursiops truncatus) que vararon en la Isla del Carmen, Campeche. Revista Mexicana de Biodiversidad. IB-UNAM 9. https://doi.org/10.22201/ib.20078706e.2019.90.2513
Newsome, S.D., M.T. Clementz, and P.L. Koch. 2010. Using stable isotope biogeochemistry to study marine mammal ecology. Marine Mammal Science 26: 509–572. https://doi.org/10.1111/j.1748-7692.2009.00354.x.
Parnell, A.C., D.L. Phillips, S. Bearhop, B.X. Semmens, E.J. Ward, J.W. Moore, A.L. Jackson, J. Grey, D.J. Kelly, and R. Inger. 2013. Bayesian stable isotope mixing models. Environmetrics 24: 387–399. https://doi.org/10.1002/env.2221.
Parra, G.J., Z. Wojtkowiak, K.J. Peters, and D. Cagnazzi. 2022. Isotopic niche overlap between sympatric Australian snubfin and humpback dolphins. Ecology and Evolution. https://doi.org/10.1002/ece3.8937.
Payo-Payo, A., B. Ruiz, L. Cardona, and A. Borrell. 2013. Effect of tissue decomposition on stable isotope signatures of striped dolphins Stenella coeruleoalba and loggerhead sea turtles Caretta caretta. Aquatic Biology 18 (2): 141–147. https://doi.org/10.3354/ab00497.
Paz-Ríos, C.E., A. Sosa-López, Y.E. Torres-Rojas, and R.E. del Río-Rodríguez. 2022. Long-term multiscale analysis of temporal variability in the fish community in Terminos Lagoon. Estuarine, Coastal and Shelf Science 277: 108066. https://doi.org/10.1016/j.ecss.2022.108066
Phillips, D.L., R. Inger, S. Bearhop, A.L. Jackson, J.W. Moore, A.C. Parnell, B.X. Semmens, and E.J. Ward. 2014. Best practices for use of stable isotope mixing models in food-web studies. Canadian Journal of Zoology 92: 823–835. https://doi.org/10.1139/cjz-2014-0127.
Post, D.M. 2002. Using stable isotopes to estimate trophic position models methods and assumptions ecology. Ecology 83 (3): 703–718. https://doi.org/10.1890/0012-9658(2002)083[0703:USITET]2.0.CO;2.
Post, D.M., C.A. Layman, D.A. Arrington, G. Takimoto, J. Quattrochi, and C.G. Montaña. 2007. Getting to the fat of the matter: Models, methods and assumptions for dealing with lipids in stable isotope analyses. Oecologia 152 (1): 179–189. https://doi.org/10.1007/s00442-006-0630-x.
Ramos-Miranda, J., and G.J. Villalobos-Zapata. (Editors). 2015. Aspectos socioambientales de la región de la laguna de Términos, 210. Campeche: Universidad Autónoma de Campeche.
Ramos-Miranda, J., L. Quiniou, D. Flores-Hernandez, T. Do-Chi, L. Ayala-Perez, and A. Sosa-Lopez. 2005. Spatial and temporal changes in the nekton of the Terminos Lagoon, Campeche. Mexico. Journal of Fish Biology. 66: 513–530. https://doi.org/10.1111/j.1095-8649.2005.00619.x.
Reynolds, J.E., and S.A. Rommel. 2009. Anatomical dissection: Thorax and abdomen. In Encyclopedia of Marine Mammals (Second Edition), ed. W.F. Perrin, B. Würsig, and J.G.M. Thewissen, 1295p. E.E.U.U: Academic Press.
Romero-Tenorio, A., and A. Delgado-Estrella. 2015. Comparación del ritmo de crecimiento de las toninas (Tursiops truncatus) pertenecientes a tres localidades distintas. Therya 6 (2): 389–400. https://doi.org/10.12933/therya-15-263.
Rossman, S., Barros Berens-McCabe, and N. B., Gandhi, H., Ostrom. P. H., Sticker, C A., & Wells, R. S. 2015. Foraging habits in a generalist predator: Sex and age influence habitat selection and resource use among bottlenose dolphins (Tursiops truncatus). Marine Mammal Science 31 (1): 155–168. https://doi.org/10.1111/mms.12143.
Secchi, E.R., S. Botta, M.M. Wiegand, L. Azevedo-Lopez, P.F. Fruet, R.C. Genoves, and J.C. Di-Tullio. 2016. Long-term and gender-related variation in the feeding ecology of common bottlenose dolphins inhabiting a subtropical estuary and the adjacent marine coast in the western South Atlantic. Marine Biology Research. 13 (1): 121–134. https://doi.org/10.1080/17451000.2016.1213398.
Sinisalo, T., R.I. Jones, E. Helle, and E.T. Valtonen. 2008. Changes in diets of individual Baltic ringed seals (Phoca hispida botnica) during their breeding season inferred from stable isotope analysis of multiple tissues. Marine Mammal Science 24 (1): 159–170. https://doi.org/10.1111/j.1748-7692.2007.00170.x.
Teixeira, R.C., G.C. Troina, F.G. Daura-Jorge, P.C. Simõnes-Lopes, and S. Botta. 2022. A practical guide on stable isotope analysis for cetacean research. Marine Mammal Science 38 (3): 1200–1228. https://doi.org/10.1111/mms.12911.
Thomas, L., C.G. Booth, P.E. Rosel, A. Hohn, J. Litz, and L.H. Schwacke. 2017. Where were they from? Modelling the sorces stock of dolphins stranded after the Deepwater Hhorizon oil splill using genetic and stable isotope data. Endangered Species Research 33: 253–264. https://doi.org/10.3354/esr00754.
Torres-Rojas, Y.E., F.P. Osuna, A.H. Herrera, F.G. Magaña, S.A. García, H. Villalobos-Ortíz, and L. Sampson. 2014. Feeding grounds of juvenile scalloped hammerhead sharks (Sphyrna lewini) in the south-eastern Gulf of California. Hidrobiología 726 (1): 81–94. https://doi.org/10.1007/s10750-013-1753-9.
Vander-Zanden, M.J., M.K. Clayton, E.K. Moody, C.T. Solomon, and B.C. Weidel. 2015. Stable isotope turnover and half-life in animal tissues: A literature synthesis. PLoS ONE 10 (1): e0116182. https://doi.org/10.1371/journal.pone.0116182.
Villéger, S., J. Ramos-Miranda, D. Flores-Hernández, and D. Mouillot. 2010. Contrasting changes in taxonomic vs. functional diversity of tropical fish communities after habitat degradation. Ecological Applications 20 (6): 1512–1522.
Wells, R., and M.D. Scott. 2009. Common bottlenose dolphin Tursiops truncatus. 249–255. In: Perrin, W.F., B. Würsig. and J.G.M. Thewissen. (Eds). Encyclopedia of marine mammals. Second Edition. 1316.
Wells, R., A. Irvine, and M. Scott. 1980. The social ecology of inshore odontocetes. P. 263–317. In: Herman L. (Ed). Cetacean behavior: mechanisms and functions. USA. 463. https://doi.org/10.1086/412413
Wilson, R.M., R.B. Tyson, J.A. Nelson, B.C. Balmer, J.P. Chanton, and D.P. Nowacek. 2017. Niche differentiation and prey selectivity among common bottlenose dolphins (Tursiops truncatus) sighted in St. George Sound, Gulf of Mexico. Frontiers in Marine Science 4: 235. https://doi.org/10.3389/fmars.2017.00235.
Acknowledgements
N.R.K.L. acknowledges Consejo Nacional de Humanidades Ciencia y Tecnología (CONAHCYT) for Ph.D. student grants. D.E.A. acknowledges the attention of the strandings and the taking of samples of carcasses that were covered by the permits granted by Dirección General de Vida Silvestre: SGPA/DGVS/08666/14 and SGPA/DGVS/05358/16. T.R.Y.E. acknowledges CONAHCYT for financing the Project “Análisis de δ 13 C y δ 18 O en otolitos de peces marinos presente en el área de protección de flora y fauna Laguna de Términos, Campeche: Indicadores de Cambio Climático”.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Communicated by Steven Litvin
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Naranjo-Ruiz, K.L., Torres-Rojas, Y.E. & Delgado-Estrella, A. Potential Residence and Coexistence Strategy of Tursiops truncatus in a Coastal Lagoon in the Southern Gulf of Mexico: Ecological Inferences Using Stable Isotopes. Estuaries and Coasts (2024). https://doi.org/10.1007/s12237-024-01337-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12237-024-01337-8