Abstract
Wrack accumulations are a source of nutrients on sandy beaches and host invertebrate species, increasing local biodiversity. The distribution of these important accumulations varies in space and time depending on the environmental conditions and the composition of species that are in the wrack. Species composition and spatiotemporal variability in wrack accumulations remain poorly understood in some regions. In this study, we evaluated the spatiotemporal variability in wrack biomass and diversity on four sandy beaches in northern Portugal over the course of 1 year. The results revealed that both the diversity and biomass of wrack varied temporally depending on the beach and tidal level where it was collected. Wrack biomass was about 20 to 35 times greater at higher than at the low tide level and was more abundant (87%) and twice as diverse at the two northernmost beaches of the study, probably due to a greater proximity to rocky shores. Large brown temperate and boreal macroalgae species such as fucoids (Fucus spp., Cystoseira spp., and Ascophyllum nodosum) and the kelp Saccorhiza polyschides were the most abundant taxa in the wrack. Fucoids were in the wrack throughout the year, while kelps predominated in the warmer months, generating 8% more biomass than that produced by fucoids throughout the year. However, since large brown macroalgae species are reducing their size and recruitment due to global warming in the north of the Iberian Peninsula, the biomass and diversity of wrack in northern Portugal could be limited in the future, compromising crucial functions of wrack in ecosystems.
Similar content being viewed by others
Introduction
Sandy beaches occupy dynamic interfaces between marine and terrestrial ecosystems. In contrast to sheltered coastal habitats, exposed sandy beaches lack large primary producers such as macroalgae or seagrasses. Instead, benthic microflora and, in certain cases, surf-zone phytoplankton are the only primary producers that can inhabit exposed sandy beaches, resulting in a low primary production ecosystem (McLachlan and Brown 2006). Nevertheless, large amounts of detached macroalgae and seagrass from other systems are washed away by waves and currents and accumulated on exposed sandy beaches. These accumulations, known as wrack, are an important source of nutrients and natural fertilizer for sandy beaches and nearby ecosystems (Cardona and García 2008; Joyce et al. 2021).
Wrack breaks up over time into small fragments due to waves and climatic conditions, or by the action of detritivores. These small fragments are remineralized by bacteria-producing organic carbon and nutrients that, over time, can accumulate within the beach, dissolve in the water column, or be transported to the atmosphere or nearshore terrestrial ecosystems (Hanisak 1993; Piriz et al. 2003). The transport of wrack to terrestrial ecosystems involves a significant marine subsidy of material, energy, and organisms (Polis and Hurd 1996) and could have a fertilization effect in the environment, increasing herbivory of terrestrial macrophytes and causing cascade effects along the food chain in the structure of the ecological communities (Piovia-Scott et al. 2013). Wrack is also an important hotspot of biodiversity because many organisms use wrack as a refuge from predators or feed directly on it (Lenanton et al. 1982; Robertson and Lenanton 1984; Rossi and Underwood 2002; Lastra et al. 2014). These organisms attract secondary consumers as arthropods, polychaetes, birds, or fishes, forming a bottom-up food web largely based on the wrack (Dugan et al. 2003; Hubbard and Dugan 2003; McLachlan and Brown 2006; Schlacher et al. 2017). In turn, important vectors to nearshore terrestrial ecosystems such as dipterans and spiders use the wrack as a habitat and refuge (Olabarria et al. 2007), which can be captured by omnivores such as mice or shorebirds (Obrist et al. 2020; Davidson et al. 2021). This fact affects the dynamics of the food web and is especially important when terrestrial productivity is low, for example during drought years (Anderson and Polis 1998). Despite the importance of wrack accumulations for marine biodiversity and ecosystem functioning, wrack dynamics is currently poorly studied. Moreover, wrack is often considered a social problem for local authorities, which remove it from touristic areas, limiting the well performance of coastal ecosystems (Mossbauer et al. 2012).
Wrack is generally deposited in a strip or patches that run parallel to the shoreline, marking the high and low tide lines. This deposition is also capable of trapping drifting sand and forming dunal coastal ecosystems (Hemminga and Nieuwenhuize 1990; Kirkman and Kendrick 1997). The frequency of wrack accumulation on the beaches varies temporally depending on several environmental and biological factors. For example, the senescence and detachment of some marine macroalgae and seagrasses may occur when they reach their maximum size in summer or in the first strong winter storms (Mann 1973; Seymour et al. 1989). Other factors such as wind or tide (Krumhansl and Scheibling 2012; Jiménez et al. 2017), the morphological characteristics of species (i.e., buoyancy) (Ochieng and Erftemeijer 1999; Gómez et al. 2013), and the structure of the shoreline (i.e., proximity to donor habitat, slope, substrate or wave exposure) (Orr et al. 2005; Barreiro et al. 2011; Wickham et al. 2020) can also influence the amount of biomass and the species composition of the wrack. Therefore, studying the spatiotemporal variability of the wrack is essential to know which species and what amount of biomass are expected to reach a region.
Species composition in the wrack is determined by the structure of nearby ecosystem assemblages. On the northwestern coast of Portugal, cold and nutrient-rich seawater allows the coexistence of boreal and temperate macroalgae species (Rubal et al. 2011; Veiga et al. 2013; Gaspar et al. 2019). The summer upwelling of this area is responsible for the reemergence of cold and nutrient-rich deep water to the surface, reducing coastal water temperatures (Lemos and Pires 2004). Consequently, the north coast of Portugal is the southern limit of distribution of several species of boreal macroalgae (e.g., Ascophyllum nodosum and Himanthalia elongata) (Araújo et al. 2009). However, climate change is causing the reduction or disappearance of boreal and temperate macroalgae on the Atlantic coasts of the Iberian Peninsula, while subtropical species are increasing (Lima et al. 2007; Piñeiro-Corbeira et al. 2016; Gaspar et al. 2019). Anthropogenic activities are also favoring the expansion of non-native macroalga species such as Sargassum muticum in northern Portugal, which is less sensitive to disturbance than indigenous canopy-forming species (Rubal et al. 2020).
Guerrero-Meseguer et al. 2020 provided a baseline study on the species composition and abundance of wrack in northern Portugal on two different dates. Other studies in Portugal have reflected the species composition of wrack in estuaries affected by eutrophication (Marques et al. 2003) or have estimated spatiotemporal variability of wrack biomass to assess its influence on macrofauna communities (Gonçalves et al. 2009; Gonçalves and Marques 2011; Bessa et al. 2014). However, none of these studies has described the taxa which composed the wrack or the differences in wrack composition between tidal levels. Due to the wide tidal amplitude in northern Portugal, wrack can accumulate at different tidal levels, resulting in different fates and ecological functions. The wrack that accumulates at a high tide level could serve as a refuge for wildlife, as well as a source of food and nutrients for many animals and adjacent terrestrial habitats. Instead, the wrack that accumulates at low tide level could be washed away by the waves and returned to the sea, providing these ecosystem properties in the subtidal. Thus, knowing the annual spatiotemporal variability of the species that compose the wrack at different tidal levels on sandy beaches of northern Portugal is essential to understand the role of these beaches in the remineralization of nutrients for nearby ecosystems (marine and terrestrial) or which areas and seasons could be more productive and richer in associated biodiversity. This study aims to describe the spatiotemporal variability of the biomass and diversity of wrack at two different tidal levels on four sandy beaches in northern Portugal. This information is key to exploring changes in the biomass and composition of wrack under future global warming scenarios.
Methods
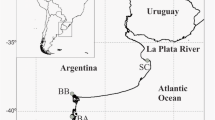
To study the spatiotemporal variability of the northern Portuguese wrack, the wrack was collected from four sandy beaches in northern Portugal (Âncora, Cabedelo, Paramos, and São Pedro de Maceda; Fig. 1) on twelve randomly chosen dates from May 2019 to May 2020 (Supplementary Table S1). These beaches were selected because they were longer than 100 m and had the same orientation (west), slope (with a drop of between −300 and −500 cm from the backshore to the seashore), and granulometry (medium and coarse sand; Guerrero-Meseguer et al. 2020). In addition, the beaches had similar tidal characteristics (semidiurnal and mesotidal, with tidal ranges between 2 and 3.5 m) and exposure to the open sea (Dias et al. 2002), were free of permanent human constructions, and, to the best of our knowledge, were not subjected to algae removal activities.
Wrack collections were made at low tide at two tidal levels (high tide level and low tide level). The high tide level corresponded to the area that extended from the high tide line to the end of the backshore and the low tide level comprised the area from the high tide line to the seashore. Wrack samples were randomly collected at each tidal level, beach, and date by using a 100 × 100 cm quadrat (n = 10) along a 100 m transect parallel to the shoreline. The wrack from each quadrat was collected in labelled plastic bags and transported to the laboratory, where it was cleaned and identified. When macrophytes were degraded, they were identified at the lowest possible taxonomic level (mainly genus). In the cases in which it was not possible to separate the taxa on the same day of the sampling, the collected samples were frozen until their identification. Once each taxon was identified, it was immediately dried at 60 °C until reaching a constant weight to quantify the biomass and diversity of taxa per quadrat. Depending on the taxon and the amount of each taxon per sample, the drying time varied from 1 to 3 days.
Data Analyses
Wrack data was analyzed using univariate and multivariate techniques to test whether the response variables examined differed between tidal levels and beaches at different sampling dates. Univariate analyses of variance (ANOVA) were used to explore significant differences in wrack biomass and diversity. These analyses were based on a three-way model with Beach as an orthogonal fixed factor with four levels (i.e., Âncora, Cabedelo, Paramos, and São Pedro de Maceda), Tidal level as an orthogonal fixed factor with two levels (high and low), and Date as an orthogonal random factor with twelve levels (May 2019, June, July, July, August, September, October, November, December, January, February, March, and May 2020) and ten replicates (i.e., each quadrat). Cochran’s C tests were previously done to test for homogeneity of variances and, when tests were significant (p < 0.05), data were transformed to remove heterogeneity. When the transformation was not possible, the untransformed data was analyzed and the results were considered robust with significant p < 0.01, to compensate for the increased probability of type I error (Underwood 1997). Whenever ANOVA showed significant differences, a Student–Newman–Keuls (SNK) post hoc test was done to explore the differences between all pairs of levels of the factor of interest.
Permutational multivariate analyses of variance (PERMANOVA) (Anderson 2001) based on Bray–Curtis untransformed dissimilarities were used to explore differences in the multivariate data from the wrack assemblage. The PERMANOVA design was as mentioned for the ANOVA. When PERMANOVA showed significant differences (p < 0.05), a pairwise comparison was done to explore significant differences between all pairs of levels of the selected factor. To test whether the differences among tidal levels, beaches, and dates were due to different multivariate dispersion among groups rather than centroid location, the PERMDISP procedure was done (Anderson 2006). The multivariate pattern was illustrated by non-metric multidimensional scaling (nMDS) ordination based on the centroids of the tidal level-beach interaction. The SIMPER procedure (Clarke 1993) was used to determine the percentage contribution (δi%) of the different taxa from the wrack to the Bray–Curtis dissimilarity among tidal level, beaches, and dates (δi). A wrack taxon was considered important if its contribution to the total percentage of dissimilarity was ≥ 1%.
Results
Wrack Diversity
The results of the ANOVA analyses showed a triple interaction for the beach, tidal level, and date factors, indicating a high temporal variability in wrack diversity depending on the tidal level and beach (Table 1).
Temporal Variability in Wrack Diversity Between Tidal Levels
Wrack diversity at the high tide level was significantly higher than at the low tide level on most dates and beaches, especially in summer (Fig. 2). In contrast, wrack at the low tide level showed low taxon diversity and varied slightly among beaches over time (Fig. 2). Only in Paramos and São Pedro de Maceda, the wrack diversity at the low tide level exceeded that of the high tide level on some dates (Fig. 2C, D).
Temporal Variability in Wrack Diversity Among Beaches
Due to the low wrack diversity which showed the beaches at the low tide level (Fig. 2), post hoc analyses were focused on the results obtained at the high tide level to assess the temporal variability among the beaches.
Âncora was the beach that reached the highest values of wrack diversity over the study period (3.1 ± 0.4 taxa m2), followed by Cabedelo (2.0 ± 0.1 taxa m2), São Pedro de Maceda (1.6 ± 0.2 taxa m2), and Paramos (1.5 ± 0.1 taxa m2). Wrack diversity from Âncora was similar to or greater than that at Cabedelo on most dates (Fig. 2A, B; Supplementary Fig. S1). In addition, the wrack from the southernmost beaches of the study area (Paramos and São Pedro de Maceda) was generally less diverse than that from the northernmost beaches (Âncora and Cabedelo; Fig. 2; Supplementary Fig. S1).
Wrack Biomass
The ANOVA analyses showed a triple interaction for the beach, tidal level, and date factors, which indicated a high spatiotemporal variability in the biomass of the wrack depending on the tidal level and beach (Table 1).
Temporal Variability in Wrack Biomass Between Tidal Levels
Differences in wrack biomass between tidal levels followed different patterns depending on the beach. In summer, the wrack biomass at the high tide level of Âncora was greater than at the low tide level, but it was similar in the rest of the year (Fig. 3A). The wrack biomass at the high tide level of Cabedelo was greater than at the low tide level for most of the year (Fig. 3B). However, Paramos and São Pedro de Maceda showed similar wrack biomass at both tidal levels on most dates, with the exception of Paramos in February (Fig. 3C, D).
Temporal Variability in Wrack Biomass Among Beaches
Wrack biomass at the high tide level was greater than or equal to that obtained at the low tide level on most beaches and dates (Fig. 3). Consequently, only the results of post hoc analyses obtained at the high tide level were considered to study temporal variability of wrack biomass among the beaches.
Total wrack biomass increased from south to north on the studied beaches, varying from 49.94 g DW m2 in São Pedro de Maceda to 980.7 g DW m2 in Âncora. On most dates, the wrack biomass of Âncora was similar to that obtained at the other beaches, but in August, this beach reached four times more biomass than the others (Fig. 3A; Supplementary Fig. S1). Cabedelo reached greater or similar values of wrack biomass than Paramos and São Pedro de Maceda on most dates. Paramos and São Pedro de Maceda had very similar wrack biomass throughout the study period (Fig. 3B–D; Supplementary Fig. S1).
Spatio-Temporal Variability in the Wrack Assemblage
The results of the PERMANOVA analysis showed a significant interaction among the beach, tidal level, and date factors for the wrack assemblage structure (Table 2). PERMDISP analysis indicated that data dispersion could contribute to these differences (F95, 864 = 14.24; p = 0.001).
Temporal Variability in the Wrack Assemblage Between Tidal Levels
All the beaches, except São Pedro de Maceda, showed significant differences in the structure of wrack assemblage between tidal levels (Table 3). This fact was especially notorious on the beaches further north of the study area (Âncora and Cabedelo, Fig. 4).
Depending on the date and the beach, different taxa were largely responsible for the differences found between tidal levels. Differences between tidal levels on the structure of wrack assemblage of Âncora and Cabedelo were due to a greater biomass at the high tide level than at the low tide level of the taxa Saccorhiza polyschides, Ulva spp., Ceramium spp., Fucus spp., and Ascophyllum nodosum (Supplementary Table S1). In Paramos, the differences between both tidal levels on the structure of wrack assemblage were mainly due to a greater biomass of Sargassum muticum at the high tide level than at the low tide level in the warmer months, while in the colder months, the differences were due to a greater biomass of Fucus spp. at the high tide level than at the low tide level (Supplementary Table S1).
Temporal Variability in the Wrack Assemblage Among Beaches
To study the differences in the structure of the wrack assemblage among the beaches, the results have been focused on the high tide level, due to its greater biomass. The wrack assemblage structure was very variable among beaches and dates but showed a certain seasonal pattern, revealing the main differences among beaches from May to August, from September to October, and from November to May (Supplementary Table S2).
From May to August, the wrack assemblage of the beaches further the north of the study area was different from that of the beaches further the south due to a greater biomass of Saccorhiza polychides in Âncora and of Fucus spp. in Cabedelo (Fig. 5A, B, Supplementary Table S2). Beaches further south showed similar structure of wrack assemblage from May to July, but, in August, the wrack assemblage of Paramos showed a significantly greater biomass of Sargassum muticum than that of São Pedro de Maceda (Fig. 5C, D, Supplementary Table S2).
From September to October, the structure of the wrack assemblage of the beaches further the north of the study area also was significantly different from that of the beaches further the south. These differences were mainly due to a greater biomass of the taxa Saccorhiza polychides and Fucus spp. in Âncora and Cabedelo (Fig. 5, Supplementary Table S2). In addition, in September, the wrack assemblage structure of Âncora showed a higher biomass of the taxa Ascophyllum nodosum than Paramos and São Pedro de Maceda (Fig. 5A, Supplementary Table S2) and Cabedelo showed the greatest values of biomass of Cystoseira spp. (Fig. 5B, Supplementary Table S2). The wrack assemblage structure of Paramos and São Pedro de Maceda was significantly variable during this time interval, showing a greater biomass of Fucus spp. in Paramos and of the taxa Saccorrhiza polychides and Polysiphonia spp. in São Pedro de Maceda (Fig. 5C, D, Supplementary Table S2).
From November to May, the wrack assemblage structure was highly variable among beaches. These differences were mainly due to a greater biomass of the taxa Fucus spp., Ascophyllum nodosum, and Cystoseira spp. in Cabedelo than on the other beaches (Fig. 5B, Supplementary Table S2).
Discussion
Wrack accumulations from the sandy beaches of northern Portugal showed high spatiotemporal variability in terms of biomass and diversity. The diversity, biomass, and assemblage structure of the wrack of northern Portugal varied throughout the year, depending on the tidal level and the beach where it was collected. However, in general, wrack showed greater biomass at the higher than at the low tide level, especially on the beaches that were located further north of the study area and during the summer months. These differences were mainly due to a greater biomass of the taxa Saccorhiza polyschides in Âncora and Fucus spp. in Cabedelo. The results of this study imply that, in northern Portugal, wrack accumulation is highly variable over time and large fucoid species are the main contributors in terms of wrack biomass.
Wrack data obtained in this study shared the high spatiotemporal variability found in other studies focused on wrack accumulation (e.g., Dugan et al. 2003; Orr et al. 2005; Barreiro et al. 2011; Gómez et al. 2013). However, it should be noted that, within this variability, certain spatial and seasonal patterns emerged in the diversity and biomass of the wrack of northern Portugal. Coinciding with other studies carried out in this area (Gonçalves et al. 2009; Bessa et al. 2014), the beaches located further north showed a greater biomass of wrack (87%) than the beaches located further south. The wrack taxa that contributed the most to these differences in biomass were Saccorhiza polyschides in the warmer months and Fucus spp. throughout the year. In addition, other boreal and temperate fucoids such as Ascophyllum nodosum, Halidrys siliquosa, Himanthalia elongata, and Cystoseira spp. were also abundant in the wrack of the beaches located further north of the study area, especially after summer. These beaches (i.e., Âncora and Cabedelo) were closer to rocky platforms than the beaches located further the south in the study area (i.e., Paramos and São Pedro de Maceda), which could explain their greater abundance of canopy-forming algae such as Fucus spp., Cystoseira spp. and the kelp Saccorhiza polyschides. The differences in wrack biomass found here among beaches could mean a lower supply of nutrients in the beaches that were located further south in northern Portugal. Therefore, future studies should determine the consequences of lower wrack accumulation on the southernmost beaches of northern Portugal to assess possible effects on the functioning of the food chain on sandy beaches and nearby ecosystems.
The upwelling of cold, nutrient-rich seawater that occurs in the northern Iberian Peninsula in the summer provides favorable conditions for the settlement of boreal species such as Ascophyllum nodosum, Halidrys siliquosa, and Himanthalia elongata (Hoarau et al. 2007; Maggs et al. 2008; Araújo et al. 2009; Assis et al. 2018). Thus, the composition of wrack assemblages in this study coincides with the most common macroalgae existing on the rocky coast of northern Portugal (Araújo et al. 2009; Rubal et al. 2011; Veiga et al. 2013). Other studies carried out near estuarine areas in the northwest of the Iberian Peninsula also found a large amount of fucoid macroalgae taxa such as Fucus spp., Ascophyllum nosodum, Cystoseira spp., and Sargassum muticum but showed a greater biomass of the taxa Ulva spp. and Gracilaria gracilis than on the sandy beaches (Barreiro et al. 2011; Gómez et al. 2013). These data confirm that the diversity of taxa in the wrack depends on the environment and the types of species that surround the ecosystems where they are deposited.
The high tide level accumulated 20 to 35 times more wrack biomass than the low tide level during the study period. Wrack taxa, which have buoyant structures are more likely to be resuspended by the tide than other sheetlike and thin macroalgae (Orr et al. 2005; Gómez et al. 2013). Considering that buoyant wrack taxa such as Fucus spp., Ascophylum nodosum and Sargassum muticum were very frequent at the high tide level, we could assume that the individuals that remained at the low tide level were dragged out to sea due to the high tidal force of the study area, which could explain its lower accumulation at the low tide level. Moreover, at the low tide level, a greater quantity of laminar fragments of macroalgae such as Ulva spp. and seagrasses such as Zostera noltei was found, which are more likely to accumulate at the low tide level of sandy beaches due to the lack of buoyant structures. These results suggest that the buoyancy ability of the macroalgae may be responsible of their deposition at different tidal levels, but more research is needed to verify the influence of species morphology and tidal force on the structure of the wrack assemblage of northern Portugal.
Life history can also influence the detachment of some seaweed species, such as the case of the kelp Saccorhiza polyschides in this study. Kelps tend to senesce in mass during the summer, when they reach their maximum biomass and are dislodged by the force of waves (Fernández 2011). This fact explains that the greatest contribution of Saccorhiza polyschides biomass to the total wrack biomass in this study occurred during the warmer months. Saccorhiza polyschides reaches very large sizes compared to the other macroalgae that formed the wrack accumulations in northern Portugal and, consequently, had more influence than other wrack taxa on the spatiotemporal patterns detected in wrack biomass in this study. In fact, the biomass contribution of Saccorhiza polyschides in northern Portugal during the warmer months was greater (47%) than the reached by Fucus spp. throughout the year (39%). Since the taxa Fucus spp. and Saccorhiza polyschides dominated the wrack collected in this study, the conservation of macroalgae canopy species and kelp forests in northern Portugal is crucial to prevent limitations on the entry of organic matter into the sandy beaches and to maintain the coastal biodiversity of the area and therefore, the trophic chain of nearby ecosystems.
The European Atlantic coast is a global warming hotspot, as sea surface temperatures have risen between 0.3 and 0.8 °C per decade since mid-century, especially in the southernmost region (OSPAR 2010). This increase in temperature seems to be weakening the magnitude and frequency of summer upwelling in the north of the Iberian Peninsula (Lemos and Pires 2004). Upwelling weakness is expected to increase in the long term (Sydeman et al. 2014) and, therefore, lower nutrient availability could be accompanied by increased seawater temperatures. Since the cold and nutrient enrichment derived from upwelling is responsible for the survival of boreal species in northern Portugal, the performance of these species will be negatively affected in a global warming scenario, decreasing their abundance in the wrack. Kelps and canopy-forming macroalgae species on the Iberian Peninsula coasts are in regression due to climate change and other environmental stressors (Casado-Amezúa et al. 2019). In the northern Iberian Peninsula, the kelp Saccorhiza polychisdes have been reduced to small patches and isolated individuals due to low recruitment after long warm summer periods (Fernández 2011). On the other hand, although fucoids can inhabit warm areas, a decrease in the percentage of reproductive structures and in the size of the species has been observed for Fucus serratus in marginal localities in the south of the Iberian Peninsula (Viejo et al. 2011) and for Fucus spiralis on the Moroccan coast (Gayral 1958). Kelps and fucoids were the main contributors to wrack accumulation on the studied sandy beaches but, in a future global warming scenario, their contribution to wrack biomass in northern Portugal could be severely limited.
This study is an example of the large spatial and temporal variability of wrack accumulations in terms of biomass and species composition on sandy beaches. This study shows for the first time the temporal and spatial variability of the different taxa that make up the wrack of northern Portugal over a year. Wrack is responsible for maintaining the flow of nutrients and is a biodiversity hotspot on sandy beaches. Therefore, special attention is required for the conservation of kelp and large brown macroalgae species in northern Portugal in order to maintain the ecosystem services provided by the wrack on sandy beaches in the future. The data used for this study are available in the database of the Global Biodiversity Information Facility (GBIF) (Guerrero-Meseguer et al., 2022).
Data Availability
This information and its reference have been added in the text: The data used for this study are available in the database of the Global Biodiversity Information Facility (GBIF) (Guerrero-Meseguer et al., 2022).
References
Anderson, Marti J. 2001. Permutation tests for univariate or multivariate analysis of variance and regression. Canadian Journal of Fisheries and Aquatic Sciences 58: 626–639.
Anderson, Marti J. 2006. Distance-based tests for homogeneity of multivariate dispersions. Biometrics 62: 245–253. https://doi.org/10.1111/j.1541-0420.2005.00440.x.
Anderson, Wendy B., and Gary A. Polis. 1998. Marine subsidies of island communities in the Gulf of California: Evidence from stable carbon and nitrogen isotopes. Oikos 81: 75. https://doi.org/10.2307/3546469.
Araújo, Rita, Ignacio Bárbara, Marta Tibaldo, Estibaliz Berecibar, Pilar Díaz. Tapia, Rui Pereira, Rui Santos, and Isabel Sousa Pinto. 2009. Checklist of benthic marine algae and cyanobacteria of northern Portugal. Botanica Marina 52: 24–46. https://doi.org/10.1515/BOT.2009.026.
Assis, Jorge, Miguel B. Araújo, and Ester A. Serrão. 2018. Projected climate changes threaten ancient refugia of kelp forests in the North Atlantic. Global Change Biology 24: e55–e66. https://doi.org/10.1111/gcb.13818.
Barreiro, F., M. Gómez, M. Lastra, J. López, and R. De La Huz. 2011. Annual cycle of wrack supply to sandy beaches: Effect of the physical environment. Marine Ecology Progress Series 433: 65–74. https://doi.org/10.3354/meps09130.
Bessa, F., A. Baeta, and J.C. Marques. 2014. Niche segregation amongst sympatric species at exposed sandy shores with contrasting wrack availabilities illustrated by stable isotopic analysis. Ecological Indicators 36. Elsevier Ltd: 694–702. https://doi.org/10.1016/j.ecolind.2013.09.026.
Cardona, Luis, and Marta García. 2008. Beach-cast seagrass material fertilizes the foredune vegetation of Mediterranean coastal dunes. Acta Oecologica 34: 97–103. https://doi.org/10.1016/j.actao.2008.04.003.
Casado-Amezúa, P., R. Araújo, I. Bárbara, R. Bermejo, Borja, I. Díez, C. Fernández, et al. 2019. Distributional shifts of canopy-forming seaweeds from the Atlantic coast of Southern Europe. Biodiversity and Conservation 28. Springer Netherlands: 1151–1172. https://doi.org/10.1007/s10531-019-01716-9.
Clarke, K.R. 1993. Non-parametric multivariate analyses of changes in community structure. Austral Ecology 18: 117–143. https://doi.org/10.1111/j.1442-9993.1993.tb00438.x.
Davidson, Katie H., Brian M. Starzomski, Rana El-Sabaawi, Morgan D. Hocking, John D. Reynolds, Sara B. Wickham, and Chris T. Darimont. 2021. Marine subsidy promotes spatial and dietary niche variation in an omnivore, the Keen’s mouse (Peromyscus keeni). Ecology and Evolution 11: 17700–17722. https://doi.org/10.1002/ece3.8225.
Dias, J.M.A., R. Gonzalez, C. Garcia, and V. Diaz-del-Rio. 2002. Sediment distribution patterns on the Galicia-Minho continental shelf. Progress in Oceanography 52: 215–231. https://doi.org/10.1016/S0079-6611(02)00007-1.
Dugan, Jenifer E., David M. Hubbard, Michael D. McCrary, and Mark O. Pierson. 2003. The response of macrofauna communities and shorebirds to macrophyte wrack subsidies on exposed sandy beaches of southern California. Estuarine, Coastal and Shelf Science 58: 25–40. https://doi.org/10.1016/S0272-7714(03)00045-3.
Fernández, C. 2011. The retreat of large brown seaweeds on the north coast of Spain: The case of Saccorhiza polyschides. European Journal of Phycology 46: 352–360. https://doi.org/10.1080/09670262.2011.617840.
Gaspar, Rui, Leonel Pereira, and Isabel Sousa-Pinto. 2019. The seaweed resources of Portugal. Botanica Marina 62: 499–525. https://doi.org/10.1515/bot-2019-0012.
Gayral, P. 1958. Algues de la Côte Atlantique Marocaine. La nature. Rabat, Société des sciences naturelles et physiques du Maroc.
Gómez, M., F. Barreiro, J. López, M. Lastra, and R. de la Huz. 2013. Deposition patterns of algal wrack species on estuarine beaches. Aquatic Botany 105. Elsevier B.V.: 25–33. https://doi.org/10.1016/j.aquabot.2012.12.001.
Gonçalves, Sílvia. C., Pedro M. Anastácio, Miguel A. Pardal, Patrícia G. Cardoso, Susana M. Ferreira, and João. C. Marques. 2009. Sandy beach macrofaunal communities on the western coast of Portugal - Is there a steady structure under similar exposed conditions? Estuarine, Coastal and Shelf Science 81: 555–568. https://doi.org/10.1016/j.ecss.2008.12.004.
Gonçalves, S.C., and J.C. Marques. 2011. The effects of season and wrack subsidy on the community functioning of exposed sandy beaches. Estuarine, Coastal and Shelf Science 95. Elsevier Ltd: 165–177. https://doi.org/10.1016/j.ecss.2011.08.036.
Guerrero-Meseguer, L. et al. 2022. Annual wrack biomass of the northern Portugal sandy beaches. GBIF.org. https://doi.org/10.14284/562.
Guerrero-Meseguer, L., P. Veiga and M. Rubal. 2022. CIIMAR - Interdisciplinary Center of Marine and Environmental Research: Portugal: Annual wrack biomass of the northern Portugal sandy beaches. https://dx.doi.org/10.14284/562.
Guerrero-Meseguer, Laura, Puri Veiga, and Marcos Rubal. 2020. Spatio-temporal variability of anthropogenic and natural wrack accumulations along the driftline: Marine litter overcomes wrack in the northern sandy beaches of Portugal. Journal of Marine Science and Engineering 8: 1–15. https://doi.org/10.3390/jmse8120966.
Hanisak, M. Denni. 1993. Nitrogen release from decomposing seaweeds: Species and temperature effects. Journal of Applied Phycology 5: 175–181. https://doi.org/10.1007/BF00004014.
Hemminga, M.A., and J. Nieuwenhuize. 1990. Seagrass wrack-induced dune formation on a tropical coast (Banc d’Arguin, Mauritania). Estuarine, Coastal and Shelf Science 31: 499–502. https://doi.org/10.1016/0272-7714(90)90040-X.
Hoarau, G., J.A. Coyer, J.H. Veldsink, W.T. Stam, and J.L. Olsen. 2007. Glacial refugia and recolonization pathways in the brown seaweed Fucus serratus. Molecular Ecology 16: 3606–3616. https://doi.org/10.1111/j.1365-294X.2007.03408.x.
Hubbard, David M., and Jenifer E. Dugan. 2003. Shorebird use of an exposed sandy beach in southern California. Estuarine, Coastal and Shelf Science 58: 41–54. https://doi.org/10.1016/S0272-7714(03)00048-9.
Jiménez, Maria A., Rafel Beltran, Anna Traveset, Maria Ll Calleja, Antonio Delgado-Huertas, and Núria. Marbà. 2017. Aeolian transport of seagrass (Posidonia oceanica) beach-cast to terrestrial systems. Estuarine, Coastal and Shelf Science 196: 31–44. https://doi.org/10.1016/j.ecss.2017.06.035.
Joyce, Matthew A., Sinead M. Crotty, Christine Angelini, Orlando Cordero, Ortals Davide De. Battisti, and John N. Griffin. 2021. Wrack enhancement of post-hurricane vegetation and geomorphological recovery in a coastal dune. PLoS ONE 17 (8): e0273258.
Kirkman, Hugh, and Gary A. Kendrick. 1997. Ecological significance and commercial harvesting of drifting and beach-cast macro-algae and seagrasses in Australia: A review. Journal of Applied Phycology 9: 311–326. https://doi.org/10.1023/A:1007965506873.
Krumhansl, K.A., and R.E. Scheibling. 2012. Production and fate of kelp detritus. Marine Ecology Progress Series 467: 281–302. https://doi.org/10.3354/meps09940.
Lastra, M., I.F. Rodil, A. Sánchez-Mata, M. García-Gallego, and J. Mora. 2014. Fate and processing of macroalgal wrack subsidies in beaches of Deception Island, Antarctic Peninsula. Journal of Sea Research 88. Elsevier B.V.: 1–10. https://doi.org/10.1016/j.seares.2013.12.011.
Lemos, Ricardo T., and Henrique O. Pires. 2004. The upwelling regime off the West Portuguese Coast, 1941–2000. International Journal of Climatology 24: 511–524. https://doi.org/10.1002/joc.1009.
Lenanton, R.C.J., A.I. Robertson, and J.A. Hansen. 1982. Nearshore accumulations of detached macrophytes as nursery areas for fish. Marine Ecology Progress Series 9: 51–57. https://doi.org/10.3354/meps009051.
Lima, Fernando P., Pedro A. Ribeiro, Nuno Queiroz, Stephen J. Hawkins, and Antóio M. Santos. 2007. Do distributional shifts of northern and southern species of algae match the warming pattern? Global Change Biology 13: 2592–2604. https://doi.org/10.1111/j.1365-2486.2007.01451.x.
Maggs, Christine A., Rita Castilho, David Foltz, Christy Henzler, Marc Taimour Jolly, John Kelly, Jeanine Olsen, et al. 2008. Evaluating signatures of glacial refugia for North Atlantic benthic marine taxa. Ecology 89: S108–S122. https://doi.org/10.1890/08-0257.1.
Mann, K.H. 1973. Seaweeds: Their productivity and strategy for growth. The role of large marine algae in coastal productivity is far more important than has been suspected. Science 182: 975–981. https://doi.org/10.1126/science.182.4116.975.
Marques, João. C., Søren. N. Nielsen, Miguel A. Pardal, and Sven E. Jørgensen. 2003. Impact of eutrophication and river management within a framework of ecosystem theories. Ecological Modelling 166: 147–168. https://doi.org/10.1016/S0304-3800(03)00134-0.
McLachlan, A.C., and A. Brown. 2006. The ecology of sandy shores. Amsterdam: Elsevier.
Mossbauer, Matthias, Inga Haller, Sven Dahlke, and Gerald Schernewski. 2012. Management of stranded eelgrass and macroalgae along the German Baltic coastline. Ocean & Coastal Management 57: 1–9. https://doi.org/10.1016/j.ocecoaman.2011.10.012.
Obrist, D.S., P.J. Hanly, J.C. Kennedy, O.T. Fitzpatrick, S.B. Wickham, C.M. Ernst, W. Nijland. et al. 2020. Marine subsidies mediate patterns in avian island biogeography. Proceedings of the Royal Society B: Biological Sciences 287. https://doi.org/10.1098/rspb.2020.0108.
Ochieng, Caroline A., and Paul L.A.. Erftemeijer. 1999. Accumulation of seagrass beach cast along the Kenyan coast: A quantitative assessment. Aquatic Botany 65: 221–238. https://doi.org/10.1016/S0304-3770(99)00042-X.
Olabarria, C., M. Lastra, and J. Garrido. 2007. Succession of macrofauna on macroalgal wrack of an exposed sandy beach: Effects of patch size and site. Marine Environmental Research 63: 19–40. https://doi.org/10.1016/j.marenvres.2006.06.001.
Orr, Michael, Martin Zimmer, Dennis E. Jelinski, and Malte Mews. 2005. Wrack deposition on different beach types: Spatial and temporal variation in the pattern of subsidy. Ecology 86: 1496–1507. https://doi.org/10.1890/04-1486.
OSPAR. 2010. Quality status report 2010. London: OSPAR Commission.
Piñeiro-Corbeira, Cristina, Rodolfo Barreiro, and Javier Cremades. 2016. Decadal changes in the distribution of common intertidal seaweeds in Galicia (NW Iberia). Marine Environmental Research 113: 106–115. https://doi.org/10.1016/j.marenvres.2015.11.012.
Piovia-Scott, Jonah, David A. Spiller, Gaku Takimoto, Louie H. Yang, Amber N. Wright, and Thomas W. Schoener. 2013. The effect of chronic seaweed subsidies on herbivory: Plant-mediated fertilization pathway overshadows lizard-mediated predator pathways. Oecologia 172: 1129–1135. https://doi.org/10.1007/s00442-012-2560-0.
Piriz, M.L., M.C. Eyras, and C.M. Rostagno. 2003. Changes in biomass and botanical composition of beach-cast seaweeds in a. Journal of Applied Phycology 15: 67–74.
Polis, Gary A., and Stephen D. Hurd. 1996. Linking marine and terrestrial food webs: Allochthonous input from the ocean supports high secondary productivity on small islands and coastal land communities. The American Naturalist 147: 396–423. https://doi.org/10.1086/285858.
Robertson, A.I., and R.C.J. Lenanton. 1984. Fish community structure and food chain dynamics in the surf-zone of sandy beaches: The role of detached macrophyte detritus. Journal of Experimental Marine Biology and Ecology 84: 265–283. https://doi.org/10.1016/0022-0981(84)90185-0.
Rossi, F., and A.J. Underwood. 2002. Small-scale disturbance and increased nutrients as influences on intertidal macrobenthic assemblages: Experimental burial of wrack in different intertidal environments. Marine Ecology Progress Series 241: 29–39. https://doi.org/10.3354/meps241029.
Rubal, M., P. Veiga, R. Vieira, and I. Sousa-Pinto. 2011. Seasonal patterns of tidepool macroalgal assemblages in the North of Portugal. Consistence between species and functional group approaches. Journal of Sea Research 66: 187–194. https://doi.org/10.1016/j.seares.2011.07.003.
Rubal, Marcos, Catarina A. Torres, and Puri Veiga. 2020. Low diversity of intertidal canopy-forming macroalgae at urbanized areas along the north Portuguese coast. Diversity 12: 211. https://doi.org/10.3390/d12060211.
Schlacher, Thomas A., Briony M. Hutton, Ben L. Gilby, Nicholaus Porch, Grainne S. Maguire, Brooke Maslo, Rod M. Connolly, Andrew D. Olds, and Michael A. Weston. 2017. Algal subsidies enhance invertebrate prey for threatened shorebirds: A novel conservation tool on ocean beaches? Estuarine, Coastal and Shelf Science 191: 28–38. https://doi.org/10.1016/j.ecss.2017.04.004.
Seymour, R.J., M.J. Tegner, P.K. Dayton, and P.E. Parnell. 1989. Storm wave induced mortality of giant kelp, Macrocystis pyrifera, in Southern California. Estuarine, Coastal and Shelf Science 28: 277–292. https://doi.org/10.1016/0272-7714(89)90018-8.
Sydeman, W.J., M. García-Reyes, D.S. Schoeman, R.R. Rykaczewski, S.A. Thompson, B.A. Black, and S.J. Bograd. 2014. Climate change and wind intensification in coastal upwelling ecosystems. Science 345: 77–80. https://doi.org/10.1126/science.1251635.
Underwood, A.J. 1997. Experiments in ecology: Their logical design and interpretation using analysis of variance. Cambridge: Cambridge University Press.
Veiga, P., M. Rubal, R. Vieira, F. Arenas, and I. Sousa-Pinto. 2013. Spatial variability in intertidal macroalgal assemblages on the North Portuguese coast: Consistence between species and functional group approaches. Helgoland Marine Research 67: 191–201. https://doi.org/10.1007/s10152-012-0315-2.
Viejo, Rosa M., Brezo Martínez, Julio Arrontes, Carmen Astudillo, and Laura Hernández. 2011. Reproductive patterns in central and marginal populations of a large brown seaweed: Drastic changes at the southern range limit. Ecography 34: 75–84. https://doi.org/10.1111/j.1600-0587.2010.06365.x.
Wickham, S.B., N. Shackelford, C.T. Darimont, W. Nijland, L.Y. Reshitnyk, J.D. Reynolds, and B.M. Starzomski. 2020. Sea wrack delivery and accumulation on islands: Factors that mediate marine nutrient permeability. Marine Ecology Progress Series 635: 37–54. https://doi.org/10.3354/meps13197.
Funding
Open Access funding provided thanks to the CRUE-CSIC agreement with Springer Nature. This research was developed under Project No. 029818, co-financed by COMPETE 2020, Portugal 2020, and the European Union through the ERDF, and by FCT through national funds. This study was partially funded by the FCT Strategic Funding UID/Multi/04423/2019. Laura Guerrero-Meseguer was funded by a grant from the Department of European Funds, University and Culture of the Government of the Balearic Islands, and Puri Veiga was hired through the Regulamento do Emprego Científico e Tecnológico—RJEC from the Portuguese Foundation for Science and Technology (FCT) program (CEECIND/03893/2018).
Author information
Authors and Affiliations
Contributions
MR contributed to the conception and design of the study. LG-M, PV, and MR recollected the data. LG-M, PV, and MR performed the statistical analysis. LG-M wrote the first draft of the manuscript. All authors contributed to the manuscript revision, read, and approved the submitted version.
Corresponding author
Additional information
Communicated by R. Scott Warren.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Guerrero-Meseguer, L., Veiga, P. & Rubal, M. Spatio-Temporal Variability of Wrack Along the Northern Portuguese Sandy Beaches. Estuaries and Coasts 46, 818–828 (2023). https://doi.org/10.1007/s12237-023-01176-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12237-023-01176-z