Abstract
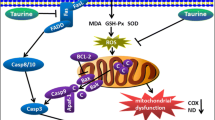
Alcoholic liver disease (ALD) caused by excessive alcohol consumption is associated with oxidative stress, mitochondrial dysfunction, and hepatocellular apoptosis. Cilostazol, a licensed clinical drug used to treat intermittent claudication, has been reported to act as a protective agent in a spectrum of diseases. However, little information regarding its role in ethanol-induced hepatocellular toxicity has been reported. In the current study, we investigated the protective effects and mechanisms of cilostazol on ethanol-induced hepatocytic injury. Rat primary hepatocytes were pretreated with cilostazol prior to ethanol treatment. MTT and LDH assay indicated that ethanol-induced cell death was ameliorated by cilostazol in a dose-dependent manner. Our results display that overproduction of intracellular reactive oxygen species (ROS) and 4-hydroxy-2-nonenal (4-HNE) induced by ethanol was attenuated by pretreatment with cilostazol. Furthermore, cilostazol significantly inhibited ethanol-induced generation of ROS in mitochondria. Importantly, it was shown that cilostazol could improve mitochondrial function in primary hepatocytes by restoring the levels of ATP and mitochondrial membrane potential (MMP). Additionally, cilostazol was found to reduce apoptosis induced by ethanol using a terminal deoxynucleotidyl transferase dUTP nick-end labeling (TUNEL) assay. Mechanistically, we found that cilostazol prevented mitochondrial pathway-mediated apoptotic signals by reversing the expression of Bax and Bcl2, the level of cleaved caspase-3, and attenuating cytochrome C release. These findings suggest the possibility of novel ALD therapies using cilostazol.









Similar content being viewed by others
References
Bakhautdin B, Das D, Mandal P, Roychowdhury S, Danner J, Bush K, Pollard K, Kaspar JW, Li W, Salomon RG, McMullen MR, Nagy LE (2014) Protective role of HO-1 and carbon monoxide in ethanol-induced cell death in hepatocytes and liver injury in mice. J Hepatol 61(5):1029–1037
Byrne CD, Targher G (2015) NAFLD: a multisystem disease. J Hepatol 62(1):S47–S64
Canbay A, Friedman S, Gores GJ (2004) Apoptosis: the nexus of liver injury and fibrosis. Hepatology 39:273–278
Choi JM, Shin HK, Kim KY, Lee JH, Hong KW (2002) Neuroprotective effect of cilostazol against focal cerebral ischemia via antiapoptotic action in rats. J Pharmacol Exp Ther 300:787–793
Cunningham CC, Bailey SM (2001) Ethanol consumption and liver mitochondria function. Biol Signals Recept 10(3–4):271–282
Dawson DL, Cutler BS, Meissner MH, Strandness DE Jr (1998) Cilostazol has beneficial effects in treatment of intermittent claudication: results from a multicenter, randomized, prospective, double-blind trial. Circulation 98:678–686
Gao B, Bataller R (2011) Alcoholic liver disease: pathogenesis and new therapeutic targets. Gastroenterology 141:1572–1585
Hoek JB, Cahill A, Pastorino JG (2002) Alcohol and mitochondria: a dysfunctional relationship. Gastroenterology 122:2049–2063
Kim KY, Shin HK, Choi JM, Hong KW (2002) Inhibition of LPS-induced apoptosis by cilostazol in human umbilical vein endothelial cells. J Pharmacol Exp Ther 300:709–715
Lim JH, Woo JS, Shin YW (2009) Cilostazol protects endothelial cells against lipopolysaccharide-induced apoptosis through ERK1/2- and P38 MAPK-dependent pathways. Korean J Intern Med 24:113–122
Luning Z, Qiang L, Sun B, Zhiying X, Zhiming G (2013) Cilostazol promotes mitochondrial biogenesis in human umbilical vein endothelial cells through activating the expression of PGC-1α. Biochem Biophys Res Commun 433:52–57
Lee JH, Park SY, Shin YW, Hong KW, Kim CD, Sung SM, Kim KY, Lee WS (2006) Neuroprotection by cilostazol, phosphodiesterase type 3 inhibitor, against apoptotic white matter changes, and cerebral hypo perfusion injury in rat after chronic cerebral hypoperfusion. Brain Res 1082:182–191
Nagy LE, de Silva SEF (1994) Adenosine A1 receptors mediate chronic ethanol-induced increases in receptor-stimulated cAMP in cultured hepatocytes. Biochem J 304:205–210
Sheng B, Gong K, Niu Y, Liu L, Yan Y, Lu G, Zhang L, Hu M, Zhao N, Zhang X, Tang P, Gong Y (2009) Inhibition of gamma-secretase activity reduces Abeta production, reduces oxidative stress, increases mitochondrial activity and leads to reduced vulnerability to apoptosis: implications for the treatment of Alzheimer’s disease. Free Radic Biol Med 46:1362–1375
Sheng B, Wang X, Su B, Lee HG, Casadesus G, Perry G, Zhu X (2012) Impaired mitochondrial biogenesis contributes to mitochondrial dysfunction in Alzheimer’s disease. J Neurochem 120(3):419–429
Stewart S, Jones D, Day CP (2001) Alcoholic liver disease: new insights into mechanisms and preventative strategies. Trends Mol Med 7:408–413
Sudo T, Tachibana K, Toga K, Tochizawa S, Inoue Y, Kimura Y, Hidaka H (2000) Potent effects of novel anti-platelet aggregatory cilostamide analogues on recombinant cyclic nucleotide phosphodiesterase isozyme activity. Biochem Pharmacol 59:347–356
Takagi T, Mishiro K, Shimazawa M, Yoshimura S, Iwama T, Hara H (2014) The phosphodiesterase III inhibitor cilostazol ameliorates ethanolinduced endothelial dysfunction. Curr Neurovasc Res 11(4):302–311
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interests.
Rights and permissions
About this article
Cite this article
Xie, X., Xu, X., Sun, C. et al. Protective effects of cilostazol on ethanol-induced damage in primary cultured hepatocytes. Cell Stress and Chaperones 23, 203–211 (2018). https://doi.org/10.1007/s12192-017-0828-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12192-017-0828-3