Abstract
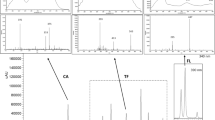
QuEChERS methodology is a new alternative for polyphenol analysis in foods and beverages. This extractive and clean up method includes several steps that should be optimized to accomplish a fast and efficient extraction. In this work, chemometrics tools were applied to optimize QuEChERS parameters for polyphenol extraction from beers. By means of D-optimal screening design, the most influential extraction parameters were defined, i.e., acetonitrile volume, acidity, PSA, and C18 amount. These parameters were optimized applying a central composite design with desirability function, establishing the following optimal conditions: 2.5 mL of acetonitrile as extraction volume, 0.5% v/v of formic acid for sample acidification, 40 mg PSA for d-SPE step, and 175 mg of C18. Method validation was carried out according to International Conference on Harmonization recommendations. Data calibration curves (0.10–10.00 mg L−1) fitted a linear regression model with determination coefficients (R2) ≥ 0.992. Repeatability (relative standard deviation, RSD) and intermediate precision (RSD) showed values ≤ 4.81% (n = 6) and ≤ 6.71% (n = 3), respectively. Recovery (n = 3) at three levels ranged from 93.98 to 119.92% (RDS ≤ 4.40%) and quantification limits ranged from 0.009 to 0.118 μg mL−1. Applying the optimized and validated method, 10 beer samples were analyzed. The principal phenolic acids found were t-ferulic acid, caffeic acid, p-coumaric acid, and p-hydroxybenzoic acid. Individually, t-ferulic acid showed the highest concentration in all samples presenting a content ranged from 0.01 ± 0.01 to 2.25 ± 0.02 μg mL−1. The proposed methodology proved to be fast, reliable, and efficient for the determination of polyphenols in beer.






Similar content being viewed by others
References
Alonso García A, Cancho Grande B, Simal Gándara J (2004) Development of a rapid method based on solid-phase extraction and liquid chromatography with ultraviolet absorbance detection for the determination of polyphenols in alcohol-free beers. J Chromatogr A 1054:175–180. https://doi.org/10.1016/j.chroma.2004.07.092
Anastassiades M, Lehotay SJ, Štajnbaher D, Schenck FJ (2003) Fast and easy multiresidue method employing acetonitrile extraction/partitioning and “dispersive solid-phase extraction” for the determination of pesticide residues in produce. J AOAC Int 86:412–431
Beltrán JL, Sanli N, Fonrodona G, Barrón D, Özkan G, Barbosa J (2003) Spectrophotometric, potentiometric and chromatographic pKa values of polyphenolic acids in water and acetonitrile–water media. Anal Chim Acta 484:253–264. https://doi.org/10.1016/S0003-2670(03)00334-9
Bourdat-Deschamps M, Leang S, Bernet N, Daudin J-J, Nélieu S (2014) Multi-residue analysis of pharmaceuticals in aqueous environmental samples by online solid-phase extraction–ultra-high-performance liquid chromatography-tandem mass spectrometry: optimisation and matrix effects reduction by quick, easy, cheap, effective, rugged and safe extraction. J Chromatogr A 1349:11–23. https://doi.org/10.1016/j.chroma.2014.05.006
Burin VM, Ferreira-Lima NE, Panceri CP, Bordignon-Luiz MT (2014) Bioactive compounds and antioxidant activity of Vitis vinifera and Vitis labrusca grapes: evaluation of different extraction methods. Microchem J 114:155–163. https://doi.org/10.1016/j.microc.2013.12.014
Callemien D, Collin S (2010) Structure, organoleptic properties, quantification methods, and stability of phenolic compounds in beer-a review. Food Rev Int 26:1–84
Campos-Requena V, Rivas B, Pérez M, Wilhelm M (2014) Application of design of experiments, response surface methodology and partial least squares regression on nanocomposites synthesis. Polym Bull 71:1961–1982. https://doi.org/10.1007/s00289-014-1166-6
Campos-Requena VH, Rivas BL, Pérez MA, Garrido-Miranda KA, Pereira ED (2015) Polymer/clay nanocomposite films as active packaging material: modeling of antimicrobial release. Eur Polym J 71:461–475. https://doi.org/10.1016/j.eurpolymj.2015.08.018
Carlson R, Nordahl Å, Barth T, Myklebust R (1991) An approach to evaluating screening experiments when several responses are measured. Chemom Intell Lab Syst 12:237–255. https://doi.org/10.1016/0169-7439(92)80004-N
Chiva-Blanch G, Urpi-Sarda M, Rotchés-Ribalta M, Zamora-Ros R, Llorach R, Lamuela-Raventós RM, Estruch R, Andrés-Lacueva C (2011) Determination of resveratrol and piceid in beer matrices by solid-phase extraction and liquid chromatography–tandem mass spectrometry. J Chromatogr A 1218:698–705. https://doi.org/10.1016/j.chroma.2010.12.012
Dejaegher B, Vander Heyden Y (2011) Experimental designs and their recent advances in set-up, data interpretation, and analytical applications. J Pharm Biomed Anal 56:141–158. https://doi.org/10.1016/j.jpba.2011.04.023
Delgado-Zamarreño MM, Pérez-Martín L, Bustamante-Rangel M, Carabias-Martínez R (2012) A modified QuEChERS method as sample treatment before the determination of isoflavones in foods by ultra-performance liquid chromatography–triple quadrupole mass spectrometry. Talanta 100:320–328. https://doi.org/10.1016/j.talanta.2012.07.070
Dvořáková M, Hulín P, Karabín M, Dostálek P (2007) Determination of polyphenols in beer by an effective method based on solid-phase extraction and high performance liquid chromatography with diode-array detection. Czech J Food Sci 25:182–188
Eriksson L, Johansson E, Wikström C (1998) Mixture design—design generation, PLS analysis, and model usage. Chemom Intell Lab Syst 43:1–24. https://doi.org/10.1016/S0169-7439(98)00126-9
Eriksson L, Johansson E, Kettaneh-Wold N, Wikstrom C, Wold S (2008) Design of experimental: principles and applications, 3rd edn. MKS Umetrics, Malmo
Fernandes PJ, Barros N, Câmara JS (2013) A survey of the occurrence of ochratoxin A in Madeira wines based on a modified QuEChERS extraction procedure combined with liquid chromatography–triple quadrupole tandem mass spectrometry. Food Res Int 54:293–301. https://doi.org/10.1016/j.foodres.2013.07.020
Fernández de Córdova ML, Medina AR (2014) Chapter 29 - analytical methods for determination of polyphenols in beer. In: Preedy V (ed) Processing and impact on antioxidants in beverages. Academic Press, San Diego, pp 289–299. https://doi.org/10.1016/B978-0-12-404738-9.00029-5
Ferreira SLC, Bruns RE, da Silva EGP, dos Santos WNL, Quintella CM, David JM, de Andrade JB, Breitkreitz MC, Jardim ICSF, Neto BB (2007) Statistical designs and response surface techniques for the optimization of chromatographic systems. J Chromatogr A 1158:2–14. https://doi.org/10.1016/j.chroma.2007.03.051
Floridi S, Montanari L, Marconi O, Fantozzi P (2003) Determination of free phenolic acids in wort and beer by coulometric array detection. J Agric Food Chem 51:1548–1554. https://doi.org/10.1021/jf0260040
Fontana AR, Bottini R (2014) High-throughput method based on quick, easy, cheap, effective, rugged and safe followed by liquid chromatography-multi-wavelength detection for the quantification of multiclass polyphenols in wines. J Chromatogr A 1342:44–53. https://doi.org/10.1016/j.chroma.2014.03.044
Herrero A, Ortiz MC, Sarabia LA (2013) D-optimal experimental design coupled with parallel factor analysis 2 decomposition a useful tool in the determination of triazines in oranges by programmed temperature vaporization–gas chromatography–mass spectrometry when using dispersive-solid phase extraction. J Chromatogr A 1288:111–126. https://doi.org/10.1016/j.chroma.2013.02.088
International Conference on Harmonisation (ICH) (2005) Harmonised tripartite guideline: validation of analytical procedures: text and methodology Q2 (R1)
Lehotay SJ, Son KA, Kwon H, Koesukwiwat U, Fu W, Mastovska K, Hoh E, Leepipatpiboon N (2010) Comparison of QuEChERS sample preparation methods for the analysis of pesticide residues in fruits and vegetables. J Chromatogr A 1217:2548–2560. https://doi.org/10.1016/j.chroma.2010.01.044
Mitić SS, Paunović DĐ, Pavlović AN, Tošić SB, Stojković MB, Mitić MN (2013) Phenolic profiles and total antioxidant capacity of marketed beers in Serbia. Int J Food Prop 17:908–922. https://doi.org/10.1080/10942912.2012.680223
Molina-García L, Ruiz-Medina A, Fernández-de Córdova ML (2011) A novel multicommuted fluorimetric optosensor for determination of resveratrol in beer. Talanta 83:850–856. https://doi.org/10.1016/j.talanta.2010.10.033
Pandey KB, Rizvi SI (2009) Plant polyphenols as dietary antioxidants in human health and disease. Oxidative Med Cell Longev 2:270–278
Piazzon A, Forte M, Nardini M (2010) Characterization of phenolics content and antioxidant activity of different beer types. J Agric Food Chem 58:10677–10683. https://doi.org/10.1021/jf101975q
Quifer-Rada P, Vallverdú-Queralt A, Martínez-Huélamo M, Chiva-Blanch G, Jáuregui O, Estruch R, Lamuela-Raventós R (2015) A comprehensive characterisation of beer polyphenols by high resolution mass spectrometry (LC–ESI-LTQ-Orbitrap-MS). Food Chem 169:336–343. https://doi.org/10.1016/j.foodchem.2014.07.154
Silva CL, Haesen N, Câmara JS (2012) A new and improved strategy combining a dispersive-solid phase extraction-based multiclass method with ultra high pressure liquid chromatography for analysis of low molecular weight polyphenols in vegetables. J Chromatogr A 1260:154–163. https://doi.org/10.1016/j.chroma.2012.08.082
Vinson JA, Mandarano M, Hirst M, Trevithick JR, Bose P (2003) Phenol antioxidant quantity and quality in foods: beers and the effect of two types of beer on an animal model of atherosclerosis. J Agric Food Chem 51:5528–5533. https://doi.org/10.1021/jf034189k
Zhao H, Chen W, Lu J, Zhao M (2010) Phenolic profiles and antioxidant activities of commercial beers. Food Chem 119:1150–1158. https://doi.org/10.1016/j.foodchem.2009.08.028
Acknowledgments
This work is part of Oscar Galarce-Bustos thesis to obtain the degree of Doctor in Science and Analytical Technology from the University of Concepcion, Chile. Authors want to thank to the National Commission of Scientific and Technological Research (CONICYT) of the Chilean Government for the doctoral scholarship granted.
Funding
This study was funded by the National Fund for Scientific and Technological Development (FONDECYT) project No. 1171857, by the National Fund for Scientific and Technological Equipment (FONDEQUIP) project No. 130209, and by the University of Concepcion.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Oscar Galarce-Bustos declares that he has no conflict of interest. Lissette Novoa declares that she has no conflict of interest. Jessy Pavon declares that she has no conflict of interest. Karem Henríquez-Aedo declares that she has no conflict of interest. Mario Aranda declares that he has no conflict of interest.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed Consent
Not applicable.
Rights and permissions
About this article
Cite this article
Galarce-Bustos, O., Novoa, L., Pavon-Perez, J. et al. Chemometric Optimization of QuEChERS Extraction Method for Polyphenol Determination in Beers by Liquid Chromatography with Ultraviolet Detection. Food Anal. Methods 12, 448–457 (2019). https://doi.org/10.1007/s12161-018-1376-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12161-018-1376-x