Abstract
Objective
This study aimed to compare the clinical outcomes of patients who received radioactive iodine (RAI) ablation after undergoing thyroidectomy for intermediate-to-high-risk differentiated thyroid carcinoma (DTC) according to the American Thyroid Association (ATA) criteria.
Methods
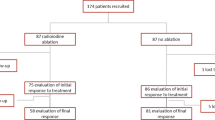
We retrospectively examined patients who underwent RAI ablation for DTC after surgical resection without macroscopic residual lesions or metastatic lesions between December 2011 and August 2016. Among 147 patients who underwent RAI ablation, those whose initial pathological stages or RAI ablation results were unknown and whose distant metastases were confirmed during RAI ablation were excluded. Low-dose therapy was defined as administration of 1110 MBq of 131iodine (131I), while high-dose therapy referred to administration of 2960–3700 MBq of 131I. We defined initial success of RAI ablation as a serum thyroglobulin concentration of < 2.0 ng/mL without thyroid-stimulating hormone stimulation and disappearance of 131I uptake in the thyroid bed on 131I scintigraphy 6–12 months after RAI ablation. RAI ablation success rates were compared between the low-dose and high-dose groups using Fisher’s exact test, and inverse probability of treatment weighting (IPTW) analysis was performed for adjusting potential biases.
Results
Among the 119 patients examined in this study (39 men and 80 women), 79 were classified as having intermediate risk, while 40 were classified as having high risk based on the ATA guideline. Initial RAI ablation success was achieved in 50/68 (73.5%) patients from the low-dose group and in 36/51 patients (70.6%) from the high-dose group (p = 0.84). Moreover, IPTW analysis showed no significant difference between the low-dose and high-dose groups. However, the success rate tended to be superior in high-risk patients who received high-dose therapy (86.2%) than in those who received low-dose therapy (72.7%) (p = 0.37).
Conclusion
There was no significant difference in the RAI ablation success rate between the low-dose and high-dose groups involving patients with intermediate-to-high-risk DTC. However, high-dose RAI ablation may be recommended in high-risk patients.

Similar content being viewed by others
References
Vigneri R, Malandrino P, Vigneri P. The changing epidemiology of thyroid cancer: why is incidence increasing? Curr Opin Oncol. 2015;27(1):1–7.
Hori M, Matsuda T, Shibata A, Katanoda K, Sobue T, Nishimoto H, et al. Cancer incidence and incidence rates in Japan in 2009: a study of 32 population-based cancer registries for the Monitoring of Cancer Incidence in Japan (MCIJ) project. Jpn J Clin Oncol. 2015;45(9):884–91.
Ruel E, Thomas S, Dinan M, Perkins JM, Roman SA, Sosa JA. Adjuvant radioactive iodine therapy is associated with improved survival for patients with intermediate-risk papillary thyroid cancer. J Clin Endocrinol Metab. 2015;100(4):1529–36.
Mallick U, Harmer C, Yap B, Wadsley J, Clarke S, Moss L, et al. Ablation with low-dose radioiodine and thyrotropin alfa in thyroid cancer. N Engl J Med. 2012;366(18):1674–85.
Schlumberger M, Catargi B, Borget I, Deandreis D, Zerdoud S, Bridji B, et al. Strategies of radioiodine ablation in patients with low-risk thyroid cancer. N Engl J Med. 2012;366(18):1663–73.
Castagna MG, Cevenini G, Theodoropoulou A, Maino F, Memmo S, Claudia C, et al. Post-surgical thyroid ablation with low or high radioiodine activities results in similar outcomes in intermediate risk differentiated thyroid cancer patients. Eur J Endocrinol. 2013;169(1):23–9.
Han JM, Kim WG, Kim TY, Joen MJ, Ryu JS, Song DE, et al. Effects of low-dose and high-dose postoperative radioiodine therapy on the clinical outcome in patients with small differentiated thyroid cancer having microscopic extrathyroidal extension. Thyroid. 2014;24(5):820–5.
Shengguang Y, Ji-Eun C, Lijuan HL. I-131 for Remnant ablation in differentiated thyroid cancer after thyroidectomy: a meta-analysis of randomized controlled evidence. Med Sci Monit. 2016;13(22):2439–50.
Du P, Jiao X, Zhou Y, Li Y, Kang S, Zhang D, et al. Low versus high radioiodine activity to ablate the thyroid after thyroidectomy for cancer: a meta-analysis of randomized controlled trials. Endocrine. 2015;48(1):96–105.
Sawka AM, Carty SE, Haugen BR, Hennessey JV, Kopp PA, Pearce EN, et al. American thyroid association guidelines and statements: past, present, and future. Thyroid. 2018;28(6):692–706.
Higashi T, Nishii R, Yamada S, Nakamoto Y, Ishizu K, Kawase S, et al. Delayed initial radioactive iodine therapy resulted in poor survival in patients with metastatic differentiated thyroid carcinoma: a retrospective statistical analysis of 198 cases. J Nucl Med. 2011;52(5):683–9.
Outpatient therapy with 131I (1,110 MBq) for residual thyroid destruction. 3rd version. http://jsnm.sakura.ne.jp/wp_jsnm/wp-content/themes/theme_jsnm/doc/i_131niyorugairai_youkou_3_20130710.pdf. Accessed 10 July 2013.
R Development Core Team. R: a language and environment for statistical computing. R Foundation for Statistical Computing; 2012.
Tamilia M, Al-Kahtani N, Rochon L, Hier MP, Payne RJ, Holcroft CA, et al. Serum thyroglobulin predicts thyroid remnant ablation failure with 30 mCi iodine-131 treatment in patients with papillary thyroid carcinoma. Nucl Med Commun. 2011;32(3):212–20.
Bernier MO, Morel O, Rodien P, Muratet JP, Giraud P, Rohmer V, et al. Prognostic value of an increase in the serum thyroglobulin level at the time of the first ablative radioiodine treatment in patients with differentiated thyroid cancer. Eur J Nucl Med Mol Imaging. 2005;32(12):1418–21.
Kawabe J, Higashiyama S, Kotani K, Yoshida A, Onoda N, Shiomi S. Evaluation of ablation of thyroid remnants with 1,850 MBq iodine-131 in 67 patients with thyroid cancer. Q J Nucl Med Mol Imaging. 2019;63(1):68–75.
Watanabe K, Uchiyama M, Fukuda K. The outcome of I-131 ablation therapy for intermediate and high-risk differentiated thyroid cancer using a strict definition of successful ablation. Jpn J Radiol. 2017;35(9):505–10.
Hugo J, Robenshtok E, Grewal R, Larson S, Tuttle RM. Recombinant human thyroid stimulating hormone-assisted radioactive iodine remnant ablation in thyroid cancer patients at intermediate to high risk of recurrence. Thyroid. 2012;22(10):1007–15.
Molinaro E, Giani C, Agate L, Biagini A, Pieruzzi L, Bianchi F, et al. Patients with differentiated thyroid cancer who underwent radioiodine thyroid remnant ablation with low-activity 131I after either recombinant human TSH or thyroid hormone therapy withdrawal showed the same outcome after a 10-year follow-up. J Clin Endocrinol Metab. 2013;98(7):2693–700.
Lee M, Lee YK, Joen TJ, Chang HS, Kim BW, Lee YS, et al. Low iodine diet for one week is sufficient for adequate preparation of high dose radioactive iodine ablation therapy of differentiated thyroid cancer patients in iodine-rich areas. Thyroid. 2014;24(8):1289–96.
Pitoia F, Marlowe RJ, Abelleira E, Faure EN, Bueno F, Schwarzstein D, et al. Radioiodine thyroid remnant ablation after recombinant human thyrotropin or thyroid hormone withdrawal in patients with high-risk differentiated thyroid cancer. J Thyroid Res. 2012;2012:481568.
Acknowledgements
Takashi Mizowaki has honoraria from Janssen Pharmaceutical K.K. and research funding from Varian Medical Systems, Inc. and Mitsubishi Heavy Industries, LTD.
Funding
There was no funding for this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The other authors do not have any conflicts of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Iizuka, Y., Katagiri, T., Ogura, K. et al. Comparison between the different doses of radioactive iodine ablation prescribed in patients with intermediate-to-high-risk differentiated thyroid cancer. Ann Nucl Med 33, 495–501 (2019). https://doi.org/10.1007/s12149-019-01357-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12149-019-01357-6