Abstract
Objective
Although Parkinson’s disease (PD) is frequently accompanied by depression, brain perfusion deficits in PD with depression remain unclear. This study aimed to assess alterations in regional cerebral blood flow (rCBF) in depressed PD patients using 99mTc hexamethyl-propylene-amine-oxime single-photon emission computed tomography (SPECT).
Methods
Among 78 patients with PD, 35 patients were classified into the depressed PD group, while the rest (43 patients) was assigned to the nondepressed PD group based on the scores of the Geriatric Depressive Scale (GDS). All participants underwent brain SPECT imaging. The voxel-wise whole-brain analysis and region-of-interest (ROI) analysis of the limbic areas were conducted to compare rCBF between the depressed and nondepressed PD groups.
Results
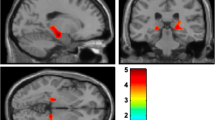
The depressed PD patients demonstrated higher GDS scores than nondepressed patients, whereas between-group differences in the PD severity and cognitive function were not significant. Perfusion in the left cuneus was increased, while that in the right superior temporal gyrus and right medial orbitofrontal cortex was reduced in the depressed PD patients as compared with nondepressed PD patients. In addition, the ROI analysis demonstrated rCBF decreases in the amygdala, anterior cingulate cortex, hippocampus, and parahippocampal gyrus in the depressed PD group. A positive correlation was found between the GDS scores and rCBF in the left cuneus cluster in the depressed PD patients.
Conclusion
This study identified the regional pattern of brain perfusion that distinguished depressed from nondepressed PD patients. Hyperperfusion in the occipital areas and hypoperfusion in the fronto-temporo-limbic regions may be potential imaging biomarkers for depression in PD.

Similar content being viewed by others
References
Shulman LM, Taback RL, Bean J, Weiner WJ. Comorbidity of the nonmotor symptoms of Parkinson’s disease. Mov Disord. 2001;16(3):507–10.
Reijnders JS, Ehrt U, Weber WE, Aarsland D, Leentjens AF. A systematic review of prevalence studies of depression in Parkinson’s disease. Mov Disord. 2008;23(2):183–9 (quiz 313).
Lieberman A. Depression in Parkinson’s disease—a review. Acta Neurol Scand. 2006;113(1):1–8.
Marsh L. Depression and Parkinson’s disease: current knowledge. Curr Neurol Neurosci Rep. 2013;13(12):409.
Palhagen SE, Ekberg S, Walinder J, Granerus AK, Granerus G. HMPAO SPECT in Parkinson’s disease (PD) with major depression (MD) before and after antidepressant treatment. J Neurol. 2009;256(9):1510–8.
Imamura K, Okayasu N, Nagatsu T. The relationship between depression and regional cerebral blood flow in Parkinson’s disease and the effect of selegiline treatment. Acta Neurol Scand. 2011;124(1):28–39.
Jeong HS, Chung YA. Contribution of neuroimaging in the diagnosis of brain disorders: recent findings and future applications. Int J Imag Syst Technol. 2016;26(2):124–35.
Hoehn MM, Yahr MD. Parkinsonism: onset, progression and mortality. Neurology. 1967;17(5):427–42.
Kang Y, Na DL, Hahn S. A validity study on the Korean Mini-Mental State Examination (K-MMSE) in dementia patients. J Korean Neurol Assoc. 1997;15(2):300–8.
Kim JM, Shin IS, Yoon JS, Lee HY. Comparison of diagnostic validities between MMSE-K and K-MMSE for screening of dementia. J Korean Neuropsychiatr Assoc. 2003;42(1):124–30.
Jung IK, Kwak DI, Joe SH, Lee HS. A study of standardization of Korean form of Geriatric Depression Scale (KGDS). J Korean Geriatr Psychiatry. 1997;1(1):61–72.
Brett M, Anton J-L, Valabregue R, Poline J-B. Region of interest analysis using the MarsBar toolbox for SPM 99. Neuroimage. 2002;16(2):S497.
Maldjian JA, Laurienti PJ, Kraft RA, Burdette JH. An automated method for neuroanatomic and cytoarchitectonic atlas-based interrogation of fMRI data sets. Neuroimage. 2003;19(3):1233–9.
Tzourio-Mazoyer N, Landeau B, Papathanassiou D, Crivello F, Etard O, Delcroix N, et al. Automated anatomical labeling of activations in SPM using a macroscopic anatomical parcellation of the MNI MRI single-subject brain. Neuroimage. 2002;15(1):273–89.
Mentis MJ, McIntosh AR, Perrine K, Dhawan V, Berlin B, Feigin A, et al. Relationships among the metabolic patterns that correlate with mnemonic, visuospatial, and mood symptoms in Parkinson’s disease. Am J Psychiatry. 2002;159(5):746–54.
Fujimoto T, Takeuchi K, Matsumoto T, Fujita S, Honda K, Higashi Y, et al. Metabolic changes in the brain of patients with late-onset major depression. Psychiatry Res. 2008;164(1):48–57.
Smith GS, Kramer E, Ma Y, Kingsley P, Dhawan V, Chaly T, et al. The functional neuroanatomy of geriatric depression. Int J Geriatr Psychiatry. 2009;24(8):798–808.
Hasler G, Fromm S, Alvarez RP, Luckenbaugh DA, Drevets WC, Grillon C. Cerebral blood flow in immediate and sustained anxiety. J Neurosci. 2007;27(23):6313–9.
Bhagwagar Z, Wylezinska M, Jezzard P, Evans J, Boorman E, Matthews PM, et al. Low GABA concentrations in occipital cortex and anterior cingulate cortex in medication-free, recovered depressed patients. Int J Neuropsychopharmacol. 2008;11(2):255–60.
Maciag D, Hughes J, O’Dwyer G, Pride Y, Stockmeier CA, Sanacora G, et al. Reduced density of calbindin immunoreactive GABAergic neurons in the occipital cortex in major depression: relevance to neuroimaging studies. Biol Psychiatry. 2010;67(5):465–70.
Roland PE, Friberg L. The effect of the GABA-A agonist THIP on regional cortical blood flow in humans. A new test of hemispheric dominance. J Cereb Blood Flow Metab. 1988;8(3):314–23.
Spanaki MV, Siegel H, Kopylev L, Fazilat S, Dean A, Liow K, et al. The effect of vigabatrin (gamma-vinyl GABA) on cerebral blood flow and metabolism. Neurology. 1999;53(7):1518–22.
Friberg TR, Borrero G. Diminished perception of ambient light: a symptom of clinical depression? J Affect Disord. 2000;61(1–2):113–8.
Coullaut-Valera Garcia J, Arbaiza Diaz del Rio I, Coullaut-Valera Garcia R, Ortiz T. Alterations of P300 wave in occipital lobe in depressive patients. Actas Esp Psiquiatr. 2007;35(4):243–8.
Normann C, Schmitz D, Furmaier A, Doing C, Bach M. Long-term plasticity of visually evoked potentials in humans is altered in major depression. Biol Psychiatry. 2007;62(5):373–80.
Fitzgerald PB, Laird AR, Maller J, Daskalakis ZJ. A meta-analytic study of changes in brain activation in depression. Hum Brain Mapp. 2008;29(6):683–95.
Rigucci S, Serafini G, Pompili M, Kotzalidis GD, Tatarelli R. Anatomical and functional correlates in major depressive disorder: the contribution of neuroimaging studies. World J Biol Psychiatry. 2010;11(2 Pt 2):165–80.
Takahashi T, Yucel M, Lorenzetti V, Walterfang M, Kawasaki Y, Whittle S, et al. An MRI study of the superior temporal subregions in patients with current and past major depression. Prog Neuropsychopharmacol Biol Psychiatry. 2010;34(1):98–103.
Allison T, Puce A, McCarthy G. Social perception from visual cues: role of the STS region. Trends Cogn Sci. 2000;4(7):267–78.
Gallagher HL, Frith CD. Functional imaging of ‘theory of mind’. Trends Cogn Sci. 2003;7(2):77–83.
Cullen KR, Gee DG, Klimes-Dougan B, Gabbay V, Hulvershorn L, Mueller BA, et al. A preliminary study of functional connectivity in comorbid adolescent depression. Neurosci Lett. 2009;460(3):227–31.
Mayberg HS, Starkstein SE, Sadzot B, Preziosi T, Andrezejewski PL, Dannals RF, et al. Selective hypometabolism in the inferior frontal lobe in depressed patients with Parkinson’s disease. Ann Neurol. 1990;28(1):57–64.
Dunnett SB, Robbins TW. The functional role of mesotelencephalic dopamine systems. Biol Rev Camb Philos Soc. 1992;67(4):491–518.
Roberts AC. The importance of serotonin for orbitofrontal function. Biol Psychiatry. 2011;69(12):1185–91.
Ring HA, Bench CJ, Trimble MR, Brooks DJ, Frackowiak RS, Dolan RJ. Depression in Parkinson’s disease. A positron emission study. Br J Psychiatry. 1994;165(3):333–9.
Matsui H, Nishinaka K, Oda M, Komatsu K, Kubori T, Udaka F. Minor depression and brain perfusion images in Parkinson’s disease. Mov Disord. 2006;21(8):1169–74.
Duhameau B, Ferre JC, Jannin P, Gauvrit JY, Verin M, Millet B, et al. Chronic and treatment-resistant depression: a study using arterial spin labeling perfusion MRI at 3 Tesla. Psychiatry Res. 2010;182(2):111–6.
Kohn Y, Freedman N, Lester H, Krausz Y, Chisin R, Lerer B, et al. 99mTc-HMPAO SPECT study of cerebral perfusion after treatment with medication and electroconvulsive therapy in major depression. J Nucl Med. 2007;48(8):1273–8.
Lui S, Parkes LM, Huang X, Zou K, Chan RC, Yang H, et al. Depressive disorders: focally altered cerebral perfusion measured with arterial spin-labeling MR imaging. Radiology. 2009;251(2):476–84.
Jarnum H, Eskildsen SF, Steffensen EG, Lundbye-Christensen S, Simonsen CW, Thomsen IS, et al. Longitudinal MRI study of cortical thickness, perfusion, and metabolite levels in major depressive disorder. Acta Psychiatr Scand. 2011;124(6):435–46.
Althaus A, Becker OA, Spottke A, Dengler R, Schneider F, Kloss M, et al. Frequency and treatment of depressive symptoms in a Parkinson’s disease registry. Parkinsonism Relat Disord. 2008;14(8):626–32.
van der Hoek TC, Bus BA, Matui P, van der Marck MA, Esselink RA, Tendolkar I. Prevalence of depression in Parkinson’s disease: effects of disease stage, motor subtype and gender. J Neurol Sci. 2011;310(1–2):220–4.
Grigoriadis S, Robinson GE. Gender issues in depression. Ann Clin Psychiatry. 2007;19(4):247–55.
Acknowledgments
This study was also supported by a grant of Ministry of Science, ICT and Future Planning, Republic of Korea (NRF-2015M3C7A1064832).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no financial conflicts of interest.
Additional information
Y.-D. Kim and H. S. Jeong contributed equally to this work.
Rights and permissions
About this article
Cite this article
Kim, YD., Jeong, H.S., Song, IU. et al. Brain perfusion alterations in depressed patients with Parkinson’s disease. Ann Nucl Med 30, 731–737 (2016). https://doi.org/10.1007/s12149-016-1119-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12149-016-1119-2