Abstract
Objectives
To describe continuous glucose monitoring (CGM) derived glycemic variables, and study their association with HbA1c and socio-economic factors in young people with Type 1 diabetes mellitus (T1DM).
Methods
Ninety-two participants [age 15.7 ± 5.0 y (mean ± SD), HbA1c 8.0 ± 1.5% (mean ± SD)] wore a professional CGM sensor for 14 d.
Results
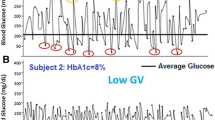
Median (IQR) time in range (TIR) was 41 (18)%. Participants spent 41 ± 20% of their day in hyperglycemia (>180 mg/dl), and 14 (13)% in hypoglycemia (<70 mg/dl). High glycemic variability (percent CV >36%) was seen in 92% participants. Older age at diagnosis was associated with higher TIR (β = 0.267, p = 0.01), lower time above range (TAR) (β = -0.352, p <0.001), but higher time below range (TBR) (β = 0.274, p = 0.006). The use of NPH vs. glargine basal insulin was associated with higher TBR (β = -0.262, p = 0.009) but lower TAR (β = 0.202, p = 0.041). HbA1c showed negative correlation with TIR (r = -0.449, p <0.001) and TBR (r = -0.466, p <0.001) and positive correlation with TAR (r = 0.580, p <0.001) and mean glucose (r = 0.589, p <0.001).
Conclusions
These data demonstrate wide gaps between the recommended vs. real world glycemic variables in patients with T1DM in this region on multiple daily insulin injections. CGM identifies glycemic variability and complements HbA1c in improving glycemic control.
Similar content being viewed by others
References
Diabetes Control and Complications Trial Research Group; Nathan DM, Genuth S, Lachin J, et al. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. New Engl J Med. 1993;329:977–86.
Nevo-Shenker M, Shalitin S. The impact of hypo- and hyperglycemia on cognition and brain development in young children with type 1 diabetes. Horm Res Paediatr. 2021;94:115–23.
Battelino T, Danne T, Bergenstal RM, et al. Clinical targets for continuous glucose monitoring data interpretation: recommendations from the international consensus on time in range. Diabetes Care. 2019;42:1593–603.
Battelino T, Alexander CM, Amiel SA, et al. Continuous glucose monitoring and metrics for clinical trials: an international consensus statement. Lancet Diabetes Endocrinol. 2023;11:42–57.
Tandon A, Bhowmik E, Ali Z, et al. Basic carbohydrate counting and glycemia in young people with type 1 diabetes in India: a randomized controlled trial. Nutrition. 2024;119:112318.
Wani R. Socioeconomic status scales-modified Kuppuswamy and Udai Pareekh’s scale updated for 2019. J Family Med Prim Care. 2019;8:1846.
Biester T, Grimsmann JM, Heidtmann B, et al. Intermittently scanned glucose values for continuous monitoring: cross-sectional analysis of glycemic control and hypoglycemia in 1809 children and adolescents with type 1 diabetes. Diabetes Technol Ther. 2021;23:160–7.
Cherubini V, Bonfanti R, Casertano A, et al. Time in range in children with type 1 diabetes using treatment strategies based on nonautomated insulin delivery systems in the real world. Diabetes Technol Ther. 2020;22:509–15.
Campbell FM, Murphy NP, Stewart C, Biester T, Kordonouri O. Outcomes of using flash glucose monitoring technology by children and young people with type 1 diabetes in a single arm study. Pediatr Diabetes. 2018;19:1294–301.
Laffel LM, Kanapka LG, Beck RW, et al. Effect of continuous glucose monitoring on glycemic control in adolescents and young adults with type 1 diabetes. JAMA. 2020;323:2388.
Karges B, Schwandt A, Heidtmann B, et al. Association of insulin pump therapy vs insulin injection therapy with severe hypoglycemia, ketoacidosis, and glycemic control among children, adolescents, and young adults with type 1 diabetes. JAMA. 2017;318:1358.
Elhabashy SA, Sakr EM, Salah NY. The efficacy of insulin degludec and insulin glargine over NPH insulin among toddlers and preschoolers with type 1 diabetes using glycemic variability and time in range. Eur J Pediatr. 2023;182:1857–68.
Danne T, Philotheou A, Goldman D, et al. A randomized trial comparing the rate of hypoglycemia-assessed using continuous glucose monitoring-in 125 preschool children with type 1 diabetes treated with insulin glargine or NPH insulin (the PRESCHOOL study). Pediatr Diabetes. 2013;14:593–601.
Valenzano M, Cibrario Bertolotti I, Valenzano A, Grassi G. Time in range–A1c hemoglobin relationship in continuous glucose monitoring of type 1 diabetes: a real-world study. BMJ Open Diabetes Res Care. 2021;9:e001045.
Helleputte S, De Backer T, Calders P, Pauwels B, Shadid S, Lapauw B. The added and interpretative value of CGM-derived parameters in type 1 diabetes depends on the level of glycemic control. Endocr Pract. 2021;27:44–50.
Babaya N, Noso S, Hiromine Y, et al. Relationship of continuous glucose monitoring-related metrics with HbA1c and residual β-cell function in Japanese patients with type 1 diabetes. Sci Rep. 2021;11:4006.
Vigersky RA, McMahon C. The relationship of hemoglobin A1c to time-in-range in patients with diabetes. Diabetes Technol Ther. 2019;21:81–5.
Marsters BL, Boucher SE, Galland BC, et al. Cutaneous adverse events in a randomized controlled trial of flash glucose monitoring among youth with type 1 diabetes mellitus. Pediatr Diabetes. 2020;21:1516–24.
Funding
This work was supported by a grant from the Endocrine Society of India (ESI Trainee Grant 2020).
Author information
Authors and Affiliations
Contributions
AT: Conceptualization, methodology, formal analysis, investigation, resources, data curation, writing- original draft, review & editing and funding acquisition; EB: Investigation, resources and data curation; ZA: Methodology, formal analysis, investigation and data curation; ST: Formal analysis, investigation and data curation; ABK: Formal analysis; PD, SS: Conceptualization, methodology, investigation and resources; VB: Conceptualization, methodology, formal analysis, investigation, resources, data curation, writing- original draft, review & editing, supervision, project administration and funding acquisition. VB will act as guarantor for this manuscript.
Corresponding author
Ethics declarations
Informed Consent
Informed written consent was taken from all adult patients or consent from the parents and assent from the child if below 18 y was taken.
Conflict of Interest
None.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tandon, A., Bhowmik, E., Ali, Z. et al. Beyond HbA1c: Identifying Gaps in Glycemic Control Among Children and Young People with Type 1 Diabetes Using Continuous Glucose Monitoring. Indian J Pediatr (2024). https://doi.org/10.1007/s12098-024-05112-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12098-024-05112-2