Abstract
Purpose
Brain metastases (BM) occur in 15–35% of patients with metastatic breast cancer, conferring poor prognosis and impairing quality of life. Clinical scores have been developed to classify patients according to their prognosis. We aimed to check the utility of the Breast Graded Prognostic Assessment (B-GPA) and its modified version (mB-GPA) and compare them in routine clinical practice.
Methods
This is an ambispective study including all patients with breast cancer BM treated in a single cancer comprehensive center. We analyzed the overall survival (OS) from BM diagnosis until death. The Kaplan–Meier method and Cox proportional hazard regression model were used in the analyses. ROC curves were performed to compare both scores.
Results
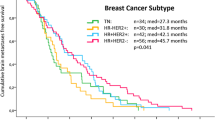
We included 169 patients; median age was 50 years. HER2-positive and triple negative patients were 33.7% and 20.7%, respectively. At the last follow-up, 90% of the patients had died. Median OS was 12 months (95% confidence interval 8.0–16.0 months). OS was worse in patients with > 3 BM and in patients with triple negative subtype.
Conclusions
In our series, we confirm that B-GPA and mB-GPA scores correlated with prognosis. ROC curves showed that B-GPA and mB-GPA have similar prognostic capabilities, slightly in favor of mB-GPA.

Similar content being viewed by others
Data availability
Database encompassing all patient data is saved in the Hospital’s hardware.
Code availability
IBM SPSS version 18 and R software version 3.3.3.
References
Zimm S, Wampler GL, Stablein D, et al. Intracerebral metastases in solid-tumor patients: natural history and results of treatment. Cancer. 1981;48:384–94.
Fokstuen T, Wilking N, Rutqvist LE, et al. Radiation therapy in the management of brain metastases from breast cancer. Breast Cancer Res Treat. 2000;62:211–6.
Griguolo G, Jacot W, Kantelhardt E, et al. External validation of Modified Breast Graded Prognostic Assessment for breast cancer patients with brain metastases: a multicentric European experience. Breast. 2018;37:36–41.
Sperduto PW, Kased N, Roberge D, et al. Summary report on the graded prognostic assessment: an accurate and facile diagnosis-specific tool to estimate survival for patients with brain metastases. J Clin Oncol. 2012;30:419–25.
Subbiah IM, Lei X, Weinberg JS, et al. Validation and development of a modified breast graded prognostic assessment as a tool for survival in patients with breast cancer and brain metastases. J Clin Oncol. 2015;33:2239–45.
Martin AM, Cagney DN, Catalano PJ, et al. Brain metastases in newly diagnosed breast cancer: a population-based study. JAMA Oncol. 2017;3:1069–77.
Lin NU, Claus E, Sohl J, et al. Sites of distant recurrence and clinical outcomes in patients with metastatic triple-negative breast cancer: high incidence of central nervous system metastases. Cancer. 2008;113:2638–45.
Piccart-Gebhart MJ, Procter M, Leyland-Jones B, et al. Trastuzumab after adjuvant chemotherapy in HER2-positive breast cancer. N Engl J Med. 2005;353:1659–72.
Lin NU, Amiri-Kordestani L, Palmieri D, et al. CNS metastases in breast cancer: old challenge, new frontiers. Clin Cancer Res. 2013;19:6404–18.
Perez EA, Romond EH, Suman VJ, et al. Trastuzumab plus adjuvant chemotherapy for human epidermal growth factor receptor 2—positive breast cancer: planned joint analysis of overall survival from NSABP B-31 and NCCTG N9831. J Clin Oncol. 2014;32:3744–52.
Kast K, Schoffer O, Link T, et al. Trastuzumab and survival of patients with metastatic breast cancer. Arch Gynecol Obstet. 2017;296:303–12.
Pernas S, Tolaney SM. HER2-positive breast cancer: new therapeutic frontiers and overcoming resistance. Ther Adv Med Oncol. 2019;19(11):1758835919833519.
Murthy RK, Loi S, Okines A, et al. Tucatinib, trastuzumab, and capecitabine for HER2-positive metastatic breast cancer. N Engl J Med. 2019;382(7):597–609.
Achrol AS, Rennert RC, Anders C, et al. Brain metastases. Nat Rev Dis Primers. 2019;5(1):5.
Moja L, Brambilla C, Compagnoni A, Pistotti V. Trastuzumab containing regimens for early breast cancer. Cochrane Database Syst Rev. 2006;2012(4):C006243.
Mulvenna P, Nankivell M, Barton R, et al. Dexamethasone and supportive care with or without whole brain radiotherapy in treating patients with non-small cell lung cancer with brain metastases unsuitable for resection or stereotactic radiotherapy (QUARTZ): results from a phase 3, non-inferiority, randomised trial. Lancet. 2016;388:2004–14.
Trovo M, Furlan C, Polesel J, et al. Radical radiation therapy for oligometastatic breast cancer: results of a prospective phase II trial. Radiother Oncol. 2018;126:177–80.
Muacevic A, Kreth FW, Tonn JC, Wowra B. Stereotactic radiosurgery for multiple brain metastases from breast carcinoma: feasibility and outcome of a local treatment concept. Cancer. 2004;100:1705–11.
Huang Z, Sun B, Shen G, et al. Brain metastasis reirradiation in patients with advanced breast cancer. J Radiat Res. 2017;58:142–8.
Yomo S, Hayashi M. The efficacy and limitations of stereotactic radiosurgery as a salvage treatment after failed whole brain radiotherapy for brain metastases. J Neurooncol. 2013;113:459–65.
Soon YY, Tham IWK, Lim KH, et al. Surgery or radiosurgery plus whole brain radiotherapy versus surgery or radiosurgery alone for brain metastases. Cochrane Database Syst Rev. 2014;2014(3):CD09454.
Niwinska A, Murawska M, Pogoda K. Breast cancer brain metastases: differences in survival depending on biological subtype, RPA RTOG prognostic class and systemic treatment after whole-brain radiotherapy (WBRT). Ann Oncol. 2010;21:942–8.
Sperduto PW, Berkey B, Gaspar LE, et al. A new prognostic index and comparison to three other indices for patients with brain metastases: an analysis of 1,960 patients in the RTOG database. Int J Radiat Oncol Biol Phys. 2008;70:510–4.
Griguolo G, Dieci MV, Giarratano T, et al. Beyond breast specific—Graded Prognostic Assessment in patients with brain metastases from breast cancer: treatment impact on outcome. J Neurooncol. 2017;131:369–76.
Sperduto PW, Mesko S, Li J, et al. Beyond an updated graded Prognostic Assessment (Breast GPA): a prognostic index and trends in treatment and survival in breast cancer brain metastases from 1985 to today. Int J Radiat Oncol Biol Phys. 2020;107(2):334–43.
Sperduto PW, Kased N, Roberge D, et al. Effect of tumor subtype on survival and the graded prognostic assessment for patients with breast cancer and brain metastases. Int J Radiat Oncol Biol Phys. 2012;82:2111–7.
Laakmann E, Riecke K, Goy Y, et al. Comparison of nine prognostic scores in patients with brain metastases of breast cancer receiving radiotherapy of the brain. J Cancer Res Clin Oncol. 2016;142:325–32.
Tai CH, Wu CC, Hwang ME, et al. Single institution validation of a modified graded prognostic assessment of patients with breast cancer brain metastases. CNS Oncol. 2018;7:25–34.
Zhuang Q, Wong RX, Lian WX, et al. Validation of modified Breast Graded Prognostic Assessment for breast cancer patients with brain metastases: extra-cranial disease progression is an independent risk factor. Ann Palliat Med. 2019;8:390–400.
Finn RS, Martin M, Rugo HS, et al. Palbociclib and letrozole in advanced breast cancer. N Engl J Med. 2016;375:1925–36.
Goetz MP, Toi M, Campone M, et al. MONARCH 3: abemaciclib as initial therapy for advanced breast cancer. J Clin Oncol. 2017;35:3638–46.
Hortobagyi GN. Ribociclib for the first-line treatment of advanced hormone receptor-positive breast cancer: a review of subgroup analyses from the MONALEESA-2 trial. Breast Cancer Res. 2018;20(1):123.
Turner NC, Slamon DJ, Ro J, et al. Overall survival with palbociclib and fulvestrant in advanced breast cancer. N Engl J Med. 2018;379:1926–36.
Gianni L, Dafni U, Gelber RD, et al. HERA 4-year follow-up. Lancet Oncol. 2011;12:236–44.
Cameron D, Piccart-Gebhart MJ, Gelber RD, et al. 11 years’ follow-up of trastuzumab after adjuvant chemotherapy in HER2-positive early breast cancer: final analysis of the HERceptin Adjuvant (HERA) trial. Lancet. 2017;389:1195–205.
Prat A, Pineda E, Adamo B, et al. Clinical implications of the intrinsic molecular subtypes of breast cancer. Breast. 2015;24:S26–35.
Von Minckwitz G, Huang CS, Mano MS, et al. Trastuzumab emtansine for residual invasive HER2-positive breast cancer. N Engl J Med. 2019;380:617–28.
Petrelli F, Ghidini M, Lonati V, et al. The efficacy of lapatinib and capecitabine in HER-2 positive breast cancer with brain metastases: a systematic review and pooled analysis. Eur J Cancer. 2017;84:141–8.
Bachelot T, Romieu G, Campone M, et al. Lapatinib plus capecitabine in patients with previously untreated brain metastases from HER2-positive metastatic breast cancer (LANDSCAPE): a single-group phase 2 study. Lancet Oncol. 2013;14:64–71.
Lin NU, Borges V, Anders C, et al. Intracranial efficacy and survival with tucatinib plus trastuzumab and capecitabine for previously treated HER2-positive breast cancer with brain metastases in the HER2CLIMB trial. J Clin Oncol. 2020;38(23):2610–9.
Cardoso F, Senkus E, Costa A, et al. 4th ESO-ESMO international consensus guidelines for advanced breast cancer (ABC 4) †. Immune-related Pathol Response Criteria. 2018;29:1634–57.
Funding
This research received no external funding.
Author information
Authors and Affiliations
Contributions
AS, JL, MG, SR, MB and CF conceived and planned the hypothesis. MG, MS, RV and SP improved the overview of the study. AS, JL, SR, MB and CF planned the study protocol and collected the data VN, AS and CF analyzed the data and presented the results. CF took the lead in writing the manuscript. All authors provided critical feedback and helped shape the research, analysis and writing of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
MG declares consulting/advisory fees from Pfizer, Eisai, Genomic-Health, Agendia, Daiichi-Sankyo, Novartis, Roche Pharma and Kern and travel/accommodation grants from Daiichi-Sankyo, Kern, Novartis and Pfizer. SP declares consulting/advisory fees from Astra-Zeneca, Daiichi-Sankyo, Novartis, Polyphor and Roche and travel/accommodation grants from Novartis. CFZ declares facilities to congress attendance from Pfizer. RVV declares professional fees from Novartis, Pfizer and Roche. RVF declares professional fees from Novartis, Gilead, Eisai, Esteve and Takeda. The rest of the authors have no conflicts of interest to declare.
Ethical approval
The protocol and the informed consent were approved by the Ethics Committee of University Hospital of Bellvitge (PR189/20). (Appendix 1in ESM).
Consent to participate
Patients included prospectively signed the informed consent approved by ethics committee (Appendix 2in ESM).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Fabregat-Franco, C., Stradella, A., Navarro, V. et al. Validation and comparison of Breast Graded Prognostic Assessment scores in patients with breast cancer and brain metastases. Clin Transl Oncol 23, 1761–1768 (2021). https://doi.org/10.1007/s12094-021-02577-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-021-02577-x