Abstract
Background and aims
As the prevalence of nonalcoholic fatty liver disease (NAFLD) is approximately 30% in the general population, it is important to develop a non-invasive biomarker for the diagnosis of nonalcoholic steatohepatitis (NASH). This prospective cross-sectional study aimed to develop a scoring system for NASH diagnosis through multiparametric magnetic resonance (MR) and clinical indicators.
Methods
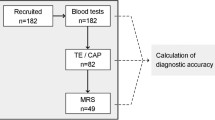
Medical history, laboratory tests, and MR parameters of patients with NAFLD were assessed. A scoring system was developed using a logistic regression model. In total, 127 patients (58 with nonalcoholic fatty liver [NAFL] and 69 with NASH) were enrolled. After evaluating 23 clinical characteristics of the patients (4 categorical and 19 numeric variables) for the NASH diagnostic model, an equation for MR elastography (MRE)-based NASH score was obtained using 3 demographic factors, 2 laboratory variables, and MRE.
Results
The MRE-based NASH score showed a satisfactory accuracy for NASH diagnosis (c-statistics, 0.841; 95% CI 0.772–0.910). At a cut-off MRE-based NASH score of 0.68 for NASH diagnosis, its sensitivity was 0.68 and specificity was 0.91. When an MRE-based NASH score of 0.37 was used as a cut-off for NASH exclusion, the sensitivity was 0.91 and specificity was 0.55. Overall, 35% (44/127) of patients were in the gray zone (between 0.37 and 0.68). Internal validation via bootstrapping also indicated the satisfactory accuracy of NASH diagnosis (optimism-corrected statistics, 0.811).
Conclusion
MRE-based NASH score is a useful and accurate non-invasive biomarker for diagnosis of NASH in patients with NAFLD.
Graphical abstract



Similar content being viewed by others
Availability of data and material
Not applicable.
Code availability
Not applicable.
Abbreviations
- ALT:
-
Alanine aminotransferase
- AST:
-
Aspartate transaminase
- AUC:
-
Area under the ROC curve
- BMI:
-
Body Mass Index
- CAP:
-
Controlled attenuation parameter
- CK18:
-
Cytokeratin 18
- FAST:
-
FibroScan-AST
- FLASH:
-
Fast low-angle shot
- IFG:
-
Impaired fasting glucose
- kPa:
-
Kilopascal
- MR:
-
Magnetic resonance
- MRE:
-
Magnetic resonance elastography
- MRI:
-
Magnetic resonance imaging
- MRS:
-
Magnetic resonance spectroscopy
- NAFLD:
-
Nonalcoholic fatty liver disease
- NASH:
-
Nonalcoholic steatohepatitis
- PDFF:
-
Proton density fat fraction
- PT:
-
Prothrombin time
- ROI:
-
Regions-of-interest
- TE:
-
Transient elastography
References
Chalasani N, Younossi Z, Lavine JE, Charlton M, Cusi K, Rinella M, et al. The diagnosis and management of nonalcoholic fatty liver disease: practice guidance from the American Association for the Study of Liver Diseases. Hepatology 2018;67:328–357. https://doi.org/10.1002/hep.29367
Rinella ME. Nonalcoholic fatty liver disease: a systematic review. JAMA 2015;313:2263–2273. https://doi.org/10.1001/jama.2015.5370
Younossi ZM, Koenig AB, Abdelatif D, Fazel Y, Henry L, Wymer M. Global epidemiology of nonalcoholic fatty liver disease—meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology 2016;64:73–84. https://doi.org/10.1002/hep.28431
Thampanitchawong P, Piratvisuth T. Liver biopsy: complications and risk factors. World J Gastroenterol 1999;5:301–304. https://doi.org/10.3748/wjg.v5.i4.301
Wong VW, Adams LA, de Lédinghen V, Wong GL, Sookoian S. Noninvasive biomarkers in NAFLD and NASH—current progress and future promise. Nat Rev Gastroenterol Hepatol 2018;15:461–478. https://doi.org/10.1038/s41575-018-0014-9
Kim TH, Lee Y, Lee YS, Gim JA, Ko E, Yim SY, et al. Circulating miRNA is a useful diagnostic biomarker for nonalcoholic steatohepatitis in nonalcoholic fatty liver disease. Sci Rep 2021;11:14639. https://doi.org/10.1038/s41598-021-94115-6
Amacher DE. Progress in the search for circulating biomarkers of nonalcoholic fatty liver disease. Biomarkers 2014;19:541–552. https://doi.org/10.3109/1354750x.2014.958535
Lee YS, Yoo YJ, Jung YK, Kim JH, Seo YS, Yim HJ, et al. Multiparametric MR is a valuable modality for evaluating disease severity of nonalcoholic fatty liver disease. Clin Transl Gastroenterol 2020;11:e00157. https://doi.org/10.14309/ctg.0000000000000157
Younossi ZM, Jarrar M, Nugent C, Randhawa M, Afendy M, Stepanova M, et al. A novel diagnostic biomarker panel for obesity-related nonalcoholic steatohepatitis (NASH). Obes Surg 2008;18:1430–1437. https://doi.org/10.1007/s11695-008-9506-y
Newsome PN, Sasso M, Deeks JJ, Paredes A, Boursier J, Chan WK, et al. FibroScan-AST (FAST) score for the non-invasive identification of patients with non-alcoholic steatohepatitis with significant activity and fibrosis: a prospective derivation and global validation study. Lancet Gastroenterol Hepatol 2020;5:362–373. https://doi.org/10.1016/s2468-1253(19)30383-8
Yoo JJ, Kim W, Kim MY, Jun DW, Kim SG, Yeon JE, et al. Recent research trends and updates on nonalcoholic fatty liver disease. Clin Mol Hepatol 2019;25:1–11. https://doi.org/10.3350/cmh.2018.0037
Park CC, Nguyen P, Hernandez C, Bettencourt R, Ramirez K, Fortney L, et al. Magnetic resonance elastography vs transient elastography in detection of fibrosis and noninvasive measurement of steatosis in patients with biopsy-proven nonalcoholic fatty liver disease. Gastroenterology 2017;152:598–607. https://doi.org/10.1053/j.gastro.2016.10.026 (e592)
Kim JW, Lee YS, Park YS, Kim BH, Lee SY, Yeon JE, et al. Multiparametric MR Index for the diagnosis of non-alcoholic steatohepatitis in patients with non-alcoholic fatty liver disease. Sci Rep 2020;10:2671. https://doi.org/10.1038/s41598-020-59601-3
American Diabetes Association. 2. Classification and diagnosis of diabetes: standards of medical care in diabetes—2021. Diabetes Care 2021;44(Suppl 1):S15–S33. https://doi.org/10.2337/dc21-S002
Kleiner DE, Brunt EM, Van Natta M, Behling C, Contos MJ, Cummings OW, et al. Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology 2005;41:1313–1321. https://doi.org/10.1002/hep.20701
Grimm A, Meyer H, Nickel MD, Nittka M, Raithel E, Chaudry O, et al. Evaluation of 2-point, 3-point, and 6-point Dixon magnetic resonance imaging with flexible echo timing for muscle fat quantification. Eur J Radiol 2018;103:57–64. https://doi.org/10.1016/j.ejrad.2018.04.011
Taylor AJ, Salerno M, Dharmakumar R, Jerosch-Herold M. T1 mapping: basic techniques and clinical applications. JACC Cardiovasc Imaging 2016;9:67–81. https://doi.org/10.1016/j.jcmg.2015.11.005
Chang Y, Kim JI, Lee B, Kim SG, Jung MJ, Kim YS, et al. Clinical application of ultrasonography-guided percutaneous liver biopsy and its safety over 18 years. Clin Mol Hepatol 2020;26:318–327. https://doi.org/10.3350/cmh.2019.0019n
Lee YH, Bang H, Park YM, Bae JC, Lee BW, Kang ES, et al. Non-laboratory-based self-assessment screening score for non-alcoholic fatty liver disease: development, validation and comparison with other scores. PLoS ONE 2014;9: e107584. https://doi.org/10.1371/journal.pone.0107584
Loomba R, Abraham M, Unalp A, Wilson L, Lavine J, Doo E, et al. Association between diabetes, family history of diabetes, and risk of nonalcoholic steatohepatitis and fibrosis. Hepatology 2012;56:943–951. https://doi.org/10.1002/hep.25772
Gastaldelli A, Cusi K. From NASH to diabetes and from diabetes to NASH: mechanisms and treatment options. JHEP Rep 2019;1:312–328. https://doi.org/10.1016/j.jhepr.2019.07.002
Frith J, Day CP, Henderson E, Burt AD, Newton JL. Non-alcoholic fatty liver disease in older people. Gerontology 2009;55:607–613. https://doi.org/10.1159/000235677
Ogrodnik M, Miwa S, Tchkonia T, Tiniakos D, Wilson CL, Lahat A, et al. Cellular senescence drives age-dependent hepatic steatosis. Nat Commun 2017;8:15691. https://doi.org/10.1038/ncomms15691
Loomis AK, Kabadi S, Preiss D, Hyde C, Bonato V, St Louis M, et al. Body Mass Index and risk of nonalcoholic fatty liver disease: two electronic health record prospective studies. J Clin Endocrinol Metab 2016;101:945–952. https://doi.org/10.1210/jc.2015-3444
Hagström H, Nasr P, Ekstedt M, Hammar U, Stål P, Hultcrantz R, et al. Risk for development of severe liver disease in lean patients with nonalcoholic fatty liver disease: a long-term follow-up study. Hepatol Commun 2018;2:48–57. https://doi.org/10.1002/hep4.1124
Younes R, Bugianesi E. NASH in lean individuals. Semin Liver Dis 2019;39:86–95. https://doi.org/10.1055/s-0038-1677517
Koo BK, Kim D, Joo SK, Kim JH, Chang MS, Kim BG, et al. Sarcopenia is an independent risk factor for non-alcoholic steatohepatitis and significant fibrosis. J Hepatol 2017;66:123–131. https://doi.org/10.1016/j.jhep.2016.08.019
Moya D, Baker SS, Liu W, Garrick M, Kozielski R, Baker RD, et al. Novel pathway for iron deficiency in pediatric non-alcoholic steatohepatitis. Clin Nutr 2015;34:549–556. https://doi.org/10.1016/j.clnu.2014.06.011
Panke CL, Tovo CV, Villela-Nogueira CA, Cravo CM, Ferreira FC, Rezende GFM, et al. Evaluation of thrombocytopenia in patients with non-alcoholic fatty liver disease without cirrhosis. Ann Hepatol 2020;19:88–91. https://doi.org/10.1016/j.aohep.2019.05.011
Yin Z, Murphy MC, Li J, Glaser KJ, Mauer AS, Mounajjed T, et al. Prediction of nonalcoholic fatty liver disease (NAFLD) activity score (NAS) with multiparametric hepatic magnetic resonance imaging and elastography. Eur Radiol 2019;29:5823–5831. https://doi.org/10.1007/s00330-019-06076-0
Pavlides M, Banerjee R, Sellwood J, Kelly CJ, Robson MD, Booth JC, et al. Multiparametric magnetic resonance imaging predicts clinical outcomes in patients with chronic liver disease. J Hepatol 2016;64:308–315. https://doi.org/10.1016/j.jhep.2015.10.009
Kim DW, Kim SY, Yoon HM, Kim KW, Byun JH. Comparison of technical failure of MR elastography for measuring liver stiffness between gradient-recalled echo and spin-echo echo-planar imaging: a systematic review and meta-analysis. J Magn Reson Imaging 2020;51:1086–1102. https://doi.org/10.1002/jmri.26918
Hu W, Yang H, Xu H, Mao Y. Radiomics based on artificial intelligence in liver diseases: where we are? Gastroenterol Rep (Oxf) 2020;8:90–97. https://doi.org/10.1093/gastro/goaa011
Jeong WK, Jamshidi N, Felker ER, Raman SS, Lu DS. Radiomics and radiogenomics of primary liver cancers. Clin Mol Hepatol 2019;25:21–29. https://doi.org/10.3350/cmh.2018.1007
Ni M, Wang L, Yu H, Wen X, Yang Y, Liu G, et al. Radiomics approaches for predicting liver fibrosis with nonenhanced T1-weighted imaging: comparison of different radiomics models. J Magn Reson Imaging 2021;53:1080–1089. https://doi.org/10.1002/jmri.27391
Funding
This study was supported by a National Research Foundation of Korea grant from the Korean government (the Ministry of Education, Science and Technology 2021R1C1C1009445 and 2018R1A2B2006183).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
All authors declare no potential conflicts of interest.
Ethical approval
This study had been approved by the institutional review board from Korea University Guro Hospital (2016GR0302).
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lee, YS., Lee, J.E., Yi, HS. et al. MRE-based NASH score for diagnosis of nonalcoholic steatohepatitis in patients with nonalcoholic fatty liver disease. Hepatol Int 16, 316–324 (2022). https://doi.org/10.1007/s12072-022-10300-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12072-022-10300-3