Abstract
The treatment of children with Obstructive Sleep Apnea Syndrome (OSAS) remains a point of debate among otorhinolaryngologists worldwide. This study aims to comparatively assess the clinical outcomes of adenotonsillectomy (ATE) and watchful waiting in children with OSAS. We searched the databases of PubMed, Scopus, and Cochrane Central Register of Controlled Trials (CENTRAL), until the 1st of October 2023. Comparative trials assessing the efficacy of adenotonsillectomy and watchful waiting in children with OSAS were considered. The primary outcome measure was the apnea-hypopnea index (AHI), and secondary outcomes included quality of life as measured by OSA-18 score, and mean SpO2 levels. A subgroup analysis evaluating the changes in AHI depending on the severity of the initial disease was also executed. We conducted a random-effects pairwise meta-analysis of change scores to include randomized and non-randomized controlled trials. The quality assessment was carried out by using the Cochrane risk of bias tool for randomized and the ROBINS-I tool for non-randomized trials, respectively. Two randomized and five non-randomized trials were included. There was a statistically significant difference regarding AHI in favor of the ATE group compared to the watchful waiting group (Standardized mean difference [SMD] was − 0.60, 95%CI −0.79 to −0.41, p < 0.001). Likewise, a statistically significant decrease in change scores for OSA-18 between ATE and watchful waiting was noted (SMD was − 0.79, 95%CI −0.97 to −0.61, p < 0.001). On the other hand, there was no significant difference when ATE and watchful waiting groups were compared for SpO2 levels between each other (SMD was 0.52, 95%CI −1.53 to 2.56, p < 0.62). In the subgroup analysis assessing mild OSAS, there was a significant difference in favor of ATE compared to watchful waiting (SMD was −0.91, 95%CI −1.35 to −0.47, p < 0.0001). For mild to moderate OSAS, similar results were noted favoring ATE as well (SMD was − 0.53, 95%CI −0.87 to −0.19, p < 0.003). This study provides evidence of moderate strength supporting the superiority of ATE over the watchful waiting approach in terms of AHI and OSA-18. This also appears to be the case for AHI in children with mild and mild to moderate OSAS.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Obstructive Sleep-Disordered Breathing (OSDB) is an umbrella term encompassing medical conditions, in which either partial or complete cessation of breathing occurs several times throughout the night, leading to poor concentration, daytime sleepiness, and chronic fatigue that negatively affects daily activities and quality of life [1, 2]. From an epidemiological point of view, OSDB occurs in 1 to 5% of children and its severity varies from simple snoring to Obstructive Sleep Apnea Syndrome (OSAS) [3, 4]. Although it can occur at any age, its prevalence is higher in children between two and six years of age. On the other hand, no significant differences in prevalence between males and females have been documented [3, 5, 6].
Pediatric OSAS is characterized by intermittent partial or complete collapse of the upper airway during sleep, resulting in reduction (hypopnea) or complete cessation (apnea) of airflow leading to arousal and hypoxia [7]. Although apnea is generally defined as the complete cessation of oronasal airflow for at least 10 s, in the pediatric population, any respiratory pause is considered apnea, regardless of the duration [8].
More specifically, OSAS is a multifactorial condition that might be linked to craniofacial anomalies, obesity, hypothyroidism, neuromuscular disorders, asthma, allergic rhinitis, and Down syndrome [9, 10]. In children, adenotonsillar hypertrophy is the most common cause of OSAS [11]. There is a significant correlation between tonsillar volume and OSBD severity [12].
Without timely diagnosis and intervention, pediatric OSAS can lead to serious clinical sequelae, including behavioral abnormalities, neurocognitive impairment, learning disabilities, cardiovascular and pulmonary hypertension, endocrine metabolic disorders, maxillofacial dysplasia (adenoid faces), growth development restriction, and an increase in the risk of cardiovascular events in adulthood [10, 13].
Clinical evaluation of pediatric OSAS consists of careful history taking, clinical examination, and eventually endoscopic evaluation. History taking and clinical examination have been reported to have positive predictive value for diagnosis of OSAS of 65% and 46%, respectively [14]. However, although history taking and physical examination are crucial in establishing OSDB diagnosis, the latter is normally confirmed by PSG [15]. Presently, PSG represents the gold standard for the diagnosis of pediatric OSAS [8]. According to the American Academy of Otolaryngology-Head and Neck Surgery Foundation (AAO-HNSF) guidelines, PSG should be performed prior to tonsillectomy in children affected by OSBD aged 2 to 18 years [16].
Pediatric OSAS sometimes, but not always, results in dips in hemoglobin saturation [17]. Hence, for the initial diagnosis of OSBD and OSAS, nocturnal pulse oximetry can also serve as a simple diagnostic tool, given its high positive predictive value (97%), its easy accessibility and applicability, as well as its relatively low cost. Furthermore, this method could be potentially useful for the selection of the most PSG-deserving children, even if this measurement is specific and not sensitive enough [17, 18].
In sleep study monitoring, the frequency of apneas and hypopneas per hour of sleep (apnea–hypopnea index [AHI]) is the key measure to define and stratify the severity of OSAS [19]. In children, the detection of a single apnea or hypopnea episode per hour is considered pathological (AHI > 1). Three degrees of OSAS severity have been identified in relation to the AHI in the pediatric population: mild AHI 1–4, moderate AHI 5–9, and severe AHI ≥ 10 [8].
Adenotonsillectomy (ATE) is currently one of the first-line treatments for pediatric OSAS. Its clinical effect is particularly significant in children with moderate or severe OSAS [20]. However limited evidence currently exists about the benefit of ATE for children with mild OSAS [21].
The aim of the present systematic review and meta-analysis is to compare the clinical efficacy of ATE with a watchful waiting strategy in the pediatric population with OSAS. In addition, we sought to investigate separately the cases of mild or mild to moderate OSAS in children, as this remains the main area of controversy.
Methods
The present systematic review with meta-analysis was registered with PROSPERO (CRD42022320656). We also used the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) [22].
Eligibility Criteria
Both randomized and non-randomized trials comparing watchful waiting with ATE exclusively in pediatric patients with OSAS were included in the systematic review. Of note, all three degrees of OSAS were considered in the analysis.
Literature Search
Two investigators (A.T. and S.A.) performed a thorough literature search in a blinded manner to identify published and unpublished studies comparing adenotonsillectomy with watchful waiting in children with OSAS. The databases of PubMed, Scopus, and Cochrane Central Register of Controlled Trials (CENTRAL) were assessed, until the 1st of October 2023 for trials comparatively assessing the efficacy of adenotonsillectomy and watchful waiting in children with OSAS. Of note, no language restrictions were applied. Moreover, we also searched the registries of ClinicalTrials.gov, International Standard Randomized Controlled Trial Number (ISRCTN), and Australian New Zealand Clinical Trials Registry (ANZCTR) for completed unpublished trials up to the same date. Furthermore, reference lists from relevant articles were manually searched to identify additional studies. The final search was performed using Ovid Search Tool (Ovid R by Wolters Kluwer) with the cooperation of a licensed librarian. The search strategy is presented in Supplemental File 1.
Selection of Studies
Two independent review authors (A.T. and S.A.) screened the titles and abstracts of articles retrieved through the systematic literature search to identify potentially relevant records independently. Following de-duplication, the titles and abstracts of the remaining papers were screened for eligibility. The full text of the remainder of the articles was assessed against our inclusion criteria. Any discrepancies between the two investigators were discussed and resolved through consensus.
Extraction of Data
Data extraction was independently conducted by two reviewers (A.T. and S.A.). From each eligible study, we collected the year of publication, country, study design, follow-up period, and intervention groups. We also extracted data about characteristics such as age, sex, inclusion and exclusion criteria, treatment protocols, and outcomes measured.
Outcome Assessment
The primary endpoint of this systematic review was the AHI since it is the main parameter of PSG used to determine OSAS and is considered the baseline indicator of OSAS. OSA-18 score and mean SpO2 levels were considered secondary outcomes. Further outcomes were not able to be synthesized in the present study because of a lack of available data.
Quality Assessment
Two researchers (A.T. and S.A.) conducted the quality appraisal of the randomized and non-randomized trials included in the analysis, using the Cochrane Collaboration’s ‘risk of bias’ tool [23] and ROBINS-I tool [24], respectively (Tables 1 and 2).
Statistical Analysis
For the change score pair-wise meta-analysis, the Review Manager (RevMan) Software (Version 5.3) [25] was utilized, analyzing changes from baseline during the follow-up. The outcomes were continuous in nature and we executed a random-effects quantitative synthesis implementing the effect size of standardized mean difference (SMD) and calculated 95% confidence intervals (CIs) [26]. In the present review, a p-value of less than 0.05 indicated statistical significance.
The statistical heterogeneity was quantified using the I-squared measure and a p-value of less than 0.05 indicated statistical significance [27]. Heterogeneity was assessed as per Cochran’s Q test. As such, a I2 value between 0% and 40% indicated that heterogeneity might not have been important, a I2 value between 30 and 60% represented moderate heterogeneity, an I2 value between 50 and 90% showed substantial heterogeneity, and a I2 value between 75 and 100% represented considerable heterogeneity [28]. Of note, the presence of small study effects could not be assessed by means of funnel plots due to the limited number of included studies [29].
Aiming to further evaluate the effect of ATE and watchful waiting on OSAS in children depending on the severity of the initial disease, we divided the study population based on the mean AHI baseline measurements in mild OSAS, mild to moderate OSAS, and moderate or severe OSAS, and we conducted a subgroup meta-analysis, evaluating uniquely the change-score of AHI in those patients.
Clinical Interpretation of the Results
For continuous data, Cohen’s rule of thumb was followed for the interpretation of effect sizes and the classification was as follows [30]:
-
− SMD < 0.4: small effect;
-
− 0.4 ≤ SMD < 0.7: moderate effect;
-
− 0.7 ≤ SMD: large effect.
Results
Literature Search
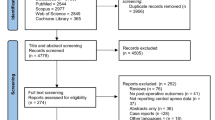
268 articles were identified during the literature search and underwent deduplication using the Ovid deduplication tool (Fig. 1). After deduplication, the remaining 173 articles underwent title review by two independent reviewers (A.T. and S.A.), of which only 89 needed abstract review due to lack of relevance to our research questions to our study. Having read the abstracts, 21 studies were thoroughly analyzed and 7 were found eligible for inclusion in the meta-analysis.
Study Characteristics
Five non-randomized and two randomized trials qualified for systematic reviewing. The enrolled studies were published between 2010 and 2020, with a total of 884 patients being eligible for qualitative synthesis. Two trials were conducted in US [31, 32], one in Spain [33], one in Sweden [34], one in Canada [35], one in Israel [36] and one in Taiwan [37]. Of note, the age of participants ranged between 1 and 16 years. Three papers reported on additional findings of the Marcus et al. trial [38,39,40] and were included in this review as part of this study. The follow-up period, namely the amount of time between the first and the second PSG, varied from 6 to 39 months, depending on the protocol of each study. Three studies assessed children with mild OSAS [31, 32, 37] two studies with mild to moderate OSAS [33, 34] and two studies with moderate or severe OSAS [35, 36] (Tables 1 and 2).
Synthesis of the Results
Apnea-Hypopnea Index (AHI)
AHI was assessed by data extracted from 2 randomized [34, 35] and 4 non-randomized trials [31, 33, 36, 37]. There was a statistically significant difference in favor of the ATE group compared to the watchful waiting group (SMD = −0.60, 95%CI −0.79 to −0.41, p < 0.0001). Of note, no important heterogeneity was detected (I2 = 19%; p = 0.29) (Fig. 2).
OSA-18
OSA-18 score was assessed by data extracted from 2 randomized [34, 35] and 1 non-randomized trial [32]. ATE was statistically superior over watchful waiting (SMD = −0.79, 95%CI −0.97 to −0.61, p < 0.00001). No important heterogeneity was found (I2 = 0%; p = 0.73) (Fig. 3).
Mean SpO2
The changes in mean Sp02 measurements during polysomnography were assessed from 1 randomized [34] and 3 non-randomized trials [33, 36, 37]. The difference in favor of ATE was not found to be statistically important (SMD = 0.52, 95%CI −1.53 to 2.56, p < 0.62). It should be noted that substantial heterogeneity was detected (I2 = 97%, p < 0.00001) (Fig. 4).
Subgroup Analysis
Mild pediatric OSAS was assessed based on AHI change-score by data extracted from 2 non-randomized trials [31, 37]. There was a significant difference in favor of ATE (SMD = −0.91, 95%CI −1.35 to −0.47, p < 0.0001). No important heterogeneity was spotted (I2 = 0%; p = 0.40) (Fig. 5).
Mild to moderate pediatric OSAS was assessed based on AHI change-score by data extracted from 1 randomized and 1 non-randomized trial [33, 34]. There was also noted a significant difference in favor of AHI (SMD = −0.53, 95%CI −0.87 to −0.19, p < 0.003). No important heterogeneity was found (I2 = 26%; p = 0.24) (Fig. 6).
Forest plot for the assessment of AHI in children with mild to moderate OSAS. Two different subgroups are considered. Vertical line demonstrates no difference between the two comparison groups. SMD = standardized mean difference, IV = inverse variance, SD = standard deviation, CI = confidence interval
Moderate and severe pediatric OSAS was assessed based on AHI change-score by data extracted from 1 randomized and 1 non-randomized trial [35, 36]. No statistically significant decrease in change scores between ATE and watchful waiting was noted (SMD = −0.62, 95%CI −1.13 to −0.10, p = 0.02). Moderate heterogeneity was found (I2 = 34%; p = 0.22) (Fig. 7).
Forest plot for the assessment of AHI in children with moderate or severe OSAS. Two different subgroups are considered. Vertical line demonstrates no difference between the two comparison groups. SMD = standardized mean difference, IV = inverse variance, SD = standard deviation, CI = confidence interval
Quality Assessment
Overall, the two randomized trials were judged to be at low risk of bias [34, 35] (Table 3). All the included non-randomized trials were deemed to be at moderate risk of bias (Table 4) [31,32,33, 36, 37].
Discussion
The goal of the treatment of OSAS is to restore optimal breathing during the night to relieve associated symptoms, improve daytime functioning, and minimize negative impact. However, the applied interventions are varied, complex, and often multidisciplinary. Given the fact that hypertrophy of upper airway lymphadenoid tissues constitutes the most common factor underlying the presence of OSAS in children [11], guidelines from both the American Academy of Otolaryngology-Head and Neck Surgery (AAO-HNS) and the American Academy of Pediatrics (AAP) recommend adenotonsillectomy (ATE) as the first-line treatment [15].
However, ATE may only partially solve the problem of airway obstruction [15], since residual and recurrent disease is reported in a large proportion of children and can be attributed to multiple factors [41]. To elaborate further, persistent OSAS after ATE may occur between 13 and 29% among children categorized as low-risk patients, whereas in higher-risk groups such as children with craniofacial and upper respiratory malformations or extreme obesity, residual OSAS may be present in up to 75% [42]. Further risk factors for the persistence of OSAS after ATE include age > 7 years, asthma, allergic rhinitis, and the severity of OSAS prior to ATE [43].
Hence, taking into consideration the undeniable intraoperative and postoperative dangers of a surgical procedure with general endotracheal anesthesia in children, in addition to the usual phenomenon of residual OSAS after ATE, in this meta-analysis, we sought to compare ATE with the conservative watchful waiting strategy. Non-surgical treatment [11] such as weight loss, CPAP, intranasal steroids, and antileukotrienes could not be considered in this study because of the lack of relevant trials.
There is an increasing concern as to whether watchful waiting would be considered an appropriate if not recommended, alternative approach depending on the severity of the disease. Coincident with this perspective, data from the CHAT [35] study suggest that young children with non-severe OSAS and the absence of sleep-related complications could be managed non-surgically. Although this trial provided evidence for the beneficial effects of early ATE, including improvements in symptoms, parent-reported behavior, quality of life, and polysomnographic findings in the treatment group, polysomnographic abnormalities were also resolved in 46% of the children in the watchful waiting group. This means that half the children in the watchful-waiting group showed normalization of AHI [35]. Additionally, this conservative watchful waiting approach is affirmed by a study by Calhoun et al. [44] which demonstrated that children with mild OSAS have neurocognitive functioning equivalent to control patients. Furthermore, proponents of the watchful waiting claim a low prevalence of tonsillar hypertrophy after children turn 8 years old [44].
Contrary to the former findings, a network meta-analysis assessed the effectiveness of various interventions for pediatric OSAS. Fourteen comparative studies involving 1064 otherwise healthy children with adenotonsillar hypertrophy were included. The study concluded that ATE was still the most effective intervention compared with no treatment in terms of improvement in AHI [45]. Furthermore, Katidis et al. [46] concluded that ATE may be beneficial in children with 1–5 events/h associated with the following conditions: cardiovascular or central nervous system-associated morbidity, enuresis, somatic growth delay or growth failure, decreased quality of life, and persistent risk factors for OSDB.
For mild OSAS especially, limited evidence is available regarding the outcomes in children with mild OSAS who have not undergone ATE. A systematic review by Tan et al. showed that mild OSAS is relieved in approximately two-thirds of the children as they grow older [47]. In any case, children with tonsillar hypertrophy should be monitored closely for the early detection of worsening OSAS [48]. On the other hand, in a prospective cohort study, Li et al. reported that not only there was no spontaneous resolution of mild OSAS in untreated children, but also a worsening in 29% after two years of follow-up [48]. Moreover, another prospective trial demonstrated significant improvement in pediatric patients undergoing ATE for mild OSAS compared to the controls, regarding the quality of life, at a follow-up of 8 months [32]. An aggressive surgical approach is further encouraged by several studies that present neurocognitive deficits in children with only mild airway obstruction, including primary snoring [49, 50].
All in all, this study proved clinically the superiority of surgical intervention compared to a conservative watchful waiting strategy in terms of improvement of the AHI and OSA-18 score. Surprisingly enough, the improvement regarding the mean SpO2 score between the ATE and watchful waiting group was not found to be clinically higher in the former. However, this finding could be justified by the substantial heterogeneity that was detected.
What children with mild and mild to moderate OSAS concerns, for which the therapeutic strategy is still arguable, the subgroup analysis indicated significant clinical improvement of OSAS in children subjected to ATE, as indicated through the improvement of the AHI. On the contrary, it was unexpected that the clinical difference between moderate and severe OSAS in children subjected to surgical treatment was not significant, but again moderate heterogeneity was present in this comparison.
Thus, this systematic review and meta-analysis support the notion that ATE could be justified in most of the patients without contraindications, compared to watchful waiting.
Limitations
We recognize that this study is not without limitations. First, the level of evidence we have provided was of moderate strength, due to the inclusion of non-randomized studies. Second, in the current meta-analysis we only considered three outcome measures, that is AHI index, OSA-18 score, and mean SpO2 levels, which reflects the fact that limited data were available for statistical pooling. To elaborate, investigating further parameters such as the Modified Epsworth Sleepiness Scale, Pediatric Sleep Questionnaire (PSQ), oxygen desaturation index (ODI), arousal counts, or Sleep-Related Breathing Disorder Scale (PSQ-SRBD) would have been of the essence for us to draw more robust conclusions on ATE clinical outcomes. Furthermore, the provision of neurocognitive measurements would have helped a great deal to determine the clinical relevance of the findings of this meta-analysis. Last but not least, synthesizing data from particular subgroups such as patients with Trisomy 21, craniofacial syndromes, obesity, and different ethnic backgrounds would have helped in delineating the extent of residual OSAS following ATE.
Conclusion
The current meta-analysis provides evidence of moderate strength which supports surgical treatment over watchful waiting for the management of pediatric OSAS. Further RCTs are required to strengthen the results of this study.
References
Ioachimescu OC, Allam JS, Samarghandi A, Anand N, Fields BG, Dholakia SA, Venkateshiah SB, Eisenstein R, Ciavatta MM, Collop NA (2020) Performance of peripheral arterial tonometry-based testing for the diagnosis of obstructive sleep apnea in a large sleep clinic cohort. J Clin Sleep Med 16(10):1663–1674. https://doi.org/10.5664/jcsm.8620
Ioachimescu OC, Dholakia SA, Venkateshiah SB, Fields B, Samarghandi A, Anand N, Eisenstein R, Ciavatta MM, Allam JS, Collop NA (2020) Improving the performance of peripheral arterial tonometry-based testing for the diagnosis of obstructive sleep apnea. J Investig Med 68(8):1370–1378. https://doi.org/10.1136/jim-2020-001448
American Academy of Sleep Medicine (2014)
Li HY, Lee LA (2009) Sleep-disordered breathing in children. Chang Gung Med J 32(3):247–257
Sateia MJ (2014) International classification of sleep disorders-third edition: highlights and modifications. Chest 146(5):1387–1394. https://doi.org/10.1378/chest.14-0970
Li HY, Lee LA Sleep-disordered breathing in children. Chang Gung Med J 2009 May-Jun ;32(3):247–257
Tsara V, Amfilochiou A, Papagrigorakis JM, Georgopoulos D, Liolios E, Kadiths A, Koudoumnakis E, Aulonitou E, Emporiadou M, Tsakanikos M, Chatzis A, Choulakis M, Chrousos G (2010) Guidelines for diagnosing and treating sleep related breathing disorders in adults and children (part 3: obstructive sleep apnea in children, diagnosis and treatment). Hippokratia 14(1):57–62
Savini S, Ciorba A, Bianchini C, Stomeo F, Corazzi V, Vicini C, Pelucchi S (2019) Assessment of obstructive sleep apnoea (OSA) in children: an update. Acta Otorhinolaryngol Ital 39(5):289–297. https://doi.org/10.14639/0392-100X-N0262
Weinstock TG, Rosen CL, Marcus CL, Garetz S, Mitchell RB, Amin R, Paruthi S, Katz E, Arens R, Weng J, Ross K, Chervin RD, Ellenberg S, Wang R, Redline S (2014) Predictors of obstructive sleep apnea severity in adenotonsillectomy candidates. Sleep 37(2):261–269. https://doi.org/10.5665/sleep.3394
Qualls H, Rimell F (2015) Surgical management of sleep-disordered breathing in children. Oper Tech Otolaryngol - Head Neck Surg 26(2):100–104
Venekamp RP, Hearne BJ, Chandrasekharan D, Blackshaw H, Lim J, Schilder AG (2015) Tonsillectomy or adenotonsillectomy versus non-surgical management for obstructive sleep-disordered breathing in children. Cochrane Database Syst Rev 2015(10):CD011165. https://doi.org/10.1002/14651858.CD011165.pub2
Fregosi RF, Quan SF, Kaemingk KL, Morgan WJ, Goodwin JL, Cabrera R, Gmitro A (2003) Sleep-disordered breathing, pharyngeal size and soft tissue anatomy in children. J Appl Physiol (1985) 95(5):2030–2038. https://doi.org/10.1152/japplphysiol.00293.2003
Farrell PC, Richards G (2017) Recognition and treatment of sleep-disordered breathing: an important component of chronic disease management. J Transl Med 15(1):114. https://doi.org/10.1186/s12967-017-1211-y
Brietzke SE, Katz ES, Roberson DW (2004) Can history and physical examination reliably diagnose pediatric obstructive sleep apnea/hypopnea syndrome? A systematic review of the literature. Otolaryngol Head Neck Surg 131(6):827–832. https://doi.org/10.1016/j.otohns.2004.07.002
Marcus CL, Brooks LJ, Draper KA, Gozal D, Halbower AC, Jones J, Schechter MS, Ward SD, Sheldon SH, Shiffman RN, Lehmann C, Spruyt K, American Academy of Pediatrics (2012) Diagnosis and management of childhood obstructive sleep apnea syndrome. Pediatrics 130(3):e714–e755. https://doi.org/10.1542/peds.2012-1672
Roland PS, Rosenfeld RM, Brooks LJ, Friedman NR, Jones J, Kim TW, Kuhar S, Mitchell RB, Seidman MD, Sheldon SH, Jones S, Robertson P, American Academy of Otolaryngology—Head and Neck Surgery Foundation (2011) Clinical practice guideline: Polysomnography for sleep-disordered breathing prior to tonsillectomy in children. Otolaryngol Head Neck Surg 145(1 Suppl):S1–15. https://doi.org/10.1177/0194599811409837
Brouillette RT, Morielli A, Leimanis A, Waters KA, Luciano R, Ducharme FM (2000) Nocturnal pulse oximetry as an abbreviated testing modality for pediatric obstructive sleep apnea. Pediatrics 105(2):405–412. https://doi.org/10.1542/peds.105.2.405
Loughlin GM, Brouillette RT, Brooke LJ, Carroll JL, Chipps BE, England SJ, Ferber P, Ferraro NF, Gaultier C, Givan DC, Haddad GG, Maddern BR, Mallory GB, Nathanson IT, Rosen CL, Thach BT, Davidson-Ward SL, Weese-Mayer DE, Wohl ME (1996) Standards and indications for cardiopulmonary sleep studies in children. Am J Respir Crit Care Med 153(2):866–878. https://doi.org/10.1164/ajrccm.153.2.8564147
Kushida CA, Littner MR, Morgenthaler T, Alessi CA, Bailey D, Coleman J Jr, Friedman L, Hirshkowitz M, Kapen S, Kramer M, Lee-Chiong T, Loube DL, Owens J, Pancer JP, Wise M (2005) Practice parameters for the indications for polysomnography and related procedures: an update for 2005. Sleep 28(4):499–521. https://doi.org/10.1093/sleep/28.4.499
Locci C, Cenere C, Sotgiu G, Puci MV, Saderi L, Rizzo D, Bussu F, Antonucci R (2023) Adenotonsillectomy in children with obstructive sleep apnea syndrome: clinical and functional outcomes. J Clin Med 12(18):5826. https://doi.org/10.3390/jcm12185826
Xu ZF, Ni X (2021) Debates in pediatric obstructive sleep apnea treatment. World J Otorhinolaryngol Head Neck Surg 7(3):194–200. https://doi.org/10.1016/j.wjorl.2021.05.001
Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gøtzsche PC, Ioannidis JP, Clarke M, Devereaux PJ, Kleijnen J, Moher D (2009) The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. J Clin Epidemiol 62(10):e1–34. https://doi.org/10.1016/j.jclinepi.2009.06.006
Higgins JP, Altman DG (2008) Assessing Risk of Bias in Included Studies. In Cochrane Handbook for Systematic Reviews of Interventions (eds J.P. Higgins and S. Green). https://doi.org/10.1002/9780470712184.ch8
Sterne JA, Hernán MA, Reeves BC, Savović J, Berkman ND, Viswanathan M, Henry D, Altman DG, Ansari MT, Boutron I, Carpenter JR, Chan AW, Churchill R, Deeks JJ, Hróbjartsson A, Kirkham J, Jüni P, Loke YK, Pigott TD, Ramsay CR, Regidor D, Rothstein HR, Sandhu L, Santaguida PL, Schünemann HJ, Shea B, Shrier I, Tugwell P, Turner L, Valentine JC, Waddington H, Waters E, Wells GA, Whiting PF, Higgins JP (2016) ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ 355:i4919. https://doi.org/10.1136/bmj.i4919
Review Manager (RevMan) (2014) [Computer program]. Version 5.3. The Nordic Cochrane Centre, The Cochrane Collaboration, Copenhagen
Deeks JJ, Higgins JP, Altman DG (2008) Analysing Data and Undertaking Meta-analyses. Cochrane Handb Syst Rev Interv Cochrane B Ser. ;243–296
Fletcher J (2007) What is heterogeneity and is it important? BMJ 334(7584):94–96. https://doi.org/10.1136/bmj.39057.406644.68
Higgins JPTTS (2002) Quantifying heterogeneity in a meta-analysis. Stat Power Anal Behav Sci 21(11):1539
Sterne JAC, Egger M, Moher D, Boutron IChapter 10: Addressing reporting biases. In:, Higgins JPT, Churchill R, Chandler J, Cumpston MS (eds) Cochrane Handbook for Systematic Reviews of Interventions version 5.2.0 (updated June 2017), Cochrane, 2017
Cohen J (1988) Statistical Power Analysis for the Behavioral Sciences (2nd ed.). Routledge. https://doi.org/10.4324/9780203771587
Trosman SJ, Eleff DJ, Krishna J, Anne S (2016) Polysomnography results in pediatric patients with mild obstructive sleep apnea: Adenotonsillectomy vs. watchful waiting. Int J Pediatr Otorhinolaryngol 83:25–30. https://doi.org/10.1016/j.ijporl.2016.01.012
Volsky PG, Woughter MA, Beydoun HA, Derkay CS, Baldassari CM (2014) Adenotonsillectomy vs observation for management of mild obstructive sleep apnea in children. Otolaryngol Head Neck Surg 150(1):126–132. https://doi.org/10.1177/0194599813509780
de Martínez-Ruiz P, Carrasco-Llatas M, Esteller-Moré E (2020) Surgical versus non-surgical treatment in the obstructive sleep apnea-hypopnea syndrome. Int J Pediatr Otorhinolaryngol 138:110310. https://doi.org/10.1016/j.ijporl.2020.110310
Fehrm J, Nerfeldt P, Browaldh N, Friberg D (2020) Effectiveness of Adenotonsillectomy vs Watchful Waiting in Young Children with mild to moderate obstructive sleep apnea: a Randomized Clinical Trial. JAMA Otolaryngol Head Neck Surg 146(7):647–654. https://doi.org/10.1001/jamaoto.2020.0869. Erratum in: JAMA Otolaryngol Head Neck Surg. 2020;146(12):1181
Marcus CL, Moore RH, Rosen CL, Giordani B, Garetz SL, Taylor HG, Mitchell RB, Amin R, Katz ES, Arens R, Paruthi S, Muzumdar H, Gozal D, Thomas NH, Ware J, Beebe D, Snyder K, Elden L, Sprecher RC, Willging P, Jones D, Bent JP, Hoban T, Chervin RD, Ellenberg SS, Redline S (2013) Childhood adenotonsillectomy trial (CHAT). A randomized trial of adenotonsillectomy for childhood sleep apnea. N Engl J Med 368(25):2366–2376. https://doi.org/10.1056/NEJMoa1215881
Ben-Israel N, Zigel Y, Tal A, Segev Y, Tarasiuk A (2011) Adenotonsillectomy improves slow-wave activity in children with obstructive sleep apnoea. Eur Respir J 37(5):1144–1150. https://doi.org/10.1183/09031936.00106710
Huang YS, Guilleminault C, Li HY, Yang CM, Wu YY, Chen NH (2007) Attention-deficit/hyperactivity disorder with obstructive sleep apnea: a treatment outcome study. Sleep Med 8(1):18–30. https://doi.org/10.1016/j.sleep.2006.05.016
Katz ES, Moore RH, Rosen CL, Mitchell RB, Amin R, Arens R, Muzumdar H, Chervin RD, Marcus CL, Paruthi S, Willging P, Redline S (2014) Growth after adenotonsillectomy for obstructive sleep apnea: an RCT. Pediatrics 134(2):282–289. https://doi.org/10.1542/peds.2014-0591
Quante M, Wang R, Weng J, Rosen CL, Amin R, Garetz SL, Katz E, Paruthi S, Arens R, Muzumdar H, Marcus CL, Ellenberg S, Redline S (2015) Childhood adenotonsillectomy trial (CHAT). The Effect of Adenotonsillectomy for Childhood Sleep Apnea on Cardiometabolic measures. Sleep 38(9):1395–1403. https://doi.org/10.5665/sleep.4976
Garetz SL, Mitchell RB, Parker PD, Moore RH, Rosen CL, Giordani B, Muzumdar H, Paruthi S, Elden L, Willging P, Beebe DW, Marcus CL, Chervin RD, Redline S (2015) Quality of life and obstructive sleep apnea symptoms after pediatric adenotonsillectomy. Pediatrics 135(2):e477–e486. https://doi.org/10.1542/peds.2014-0620
Imanguli M, Ulualp SO (2016) Risk factors for residual obstructive sleep apnea after adenotonsillectomy in children. Laryngoscope 126(11):2624–2629. https://doi.org/10.1002/lary.25979
Bhattacharjee R, Kheirandish-Gozal L, Spruyt K, Mitchell RB, Promchiarak J, Simakajornboon N, Kaditis AG, Splaingard D, Splaingard M, Brooks LJ, Marcus CL, Sin S, Arens R, Verhulst SL, Gozal D (2010) Adenotonsillectomy outcomes in treatment of obstructive sleep apnea in children: a multicenter retrospective study. Am J Respir Crit Care Med 182(5):676–683. https://doi.org/10.1164/rccm.200912-1930OC
Tagaya M, Nakata S, Yasuma F, Mitchell RB, Sasaki F, Miyazaki S, Morinaga M, Otake H, Teranishi M, Nakashima T (2012) Children with severe or moderate obstructive sleep apnoea syndrome show a high incidence of persistence after adenotonsillectomy. Acta Otolaryngol 132(11):1208–1214. https://doi.org/10.3109/00016489.2012.695088
Calhoun SL, Mayes SD, Vgontzas AN, Tsaoussoglou M, Shifflett LJ, Bixler EO (2009) No relationship between neurocognitive functioning and mild sleep disordered breathing in a community sample of children. J Clin Sleep Med 5(3):228–234
Lin SY, Su YX, Wu YC, Chang JZ, Tu YK (2020) Management of paediatric obstructive sleep apnoea: a systematic review and network meta-analysis. Int J Paediatr Dent 30(2):156–170. https://doi.org/10.1111/ipd.12593
Kaditis AG, Alonso Alvarez ML, Boudewyns A, Alexopoulos EI, Ersu R, Joosten K, Larramona H, Miano S, Narang I, Trang H, Tsaoussoglou M, Vandenbussche N, Villa MP, Van Waardenburg D, Weber S, Verhulst S (2016) Obstructive sleep disordered breathing in 2- to 18-year-old children: diagnosis and management. Eur Respir J 47(1):69–94. https://doi.org/10.1183/13993003.00385-2015
Tan HL, Alonso Alvarez ML, Tsaoussoglou M, Weber S, Kaditis AG (2017) When and why to treat the child who snores? Pediatr Pulmonol 52(3):399–412. https://doi.org/10.1002/ppul.23658
Li AM, Au CT, Ng SK, Abdullah VJ, Ho C, Fok TF, Ng PC, Wing YK (2010) Natural history and predictors for progression of mild childhood obstructive sleep apnoea. Thorax 65(1):27–31. https://doi.org/10.1136/thx.2009.120220
Kennedy JD, Blunden S, Hirte C, Parsons DW, Martin AJ, Crowe E, Williams D, Pamula Y, Lushington K (2004) Reduced neurocognition in children who snore. Pediatr Pulmonol 37(4):330–337. https://doi.org/10.1002/ppul.10453
O’Brien LM, Mervis CB, Holbrook CR, Bruner JL, Klaus CJ, Rutherford J, Raffield TJ, Gozal D (2004) Neurobehavioral implications of habitual snoring in children. Pediatrics 114(1):44–49. https://doi.org/10.1542/peds.114.1.44
Funding
Open access funding provided by HEAL-Link Greece.
Author information
Authors and Affiliations
Contributions
A.T. was the primary author. S.A. assisted the primary author with data extraction, study selection, quality assessment, and literature research, whereas F.D. was involved in writing part of this paper. K.G., and A.F. reviewed the final version of the manuscript. S.T. and K.T. critically reviewed and revised the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tsikopoulos, A., Tsikopoulos, K., Dilmperis, F. et al. Adenotonsillectomy Versus Watchful Waiting for Children with Obstructive Sleep Apnea Syndrome: A Systematic Review with Meta-Analysis. Indian J Otolaryngol Head Neck Surg (2024). https://doi.org/10.1007/s12070-024-04738-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12070-024-04738-0