Abstract
We report six new 2,6-disubstituted fluorene-based conjugated molecules carrying orthogonal triphenylamine and pyridine moiety as donor-acceptor units at the C-9 position. The geminal donor-acceptor is connected to the fluorene core by σ-conjugation. Optical/solvatochromic studies, investigated by UV-VIS absorption and fluorescence spectroscopy, show blue emission with good quantum yield (40–70%) in solution and in thin film state for the synthesized compounds. In microsecond order, the derivatives give a shorter delayed lifetime (both in solution and thin films), facilitating reverse intersystem crossing (RISC). TD-DFT studies for all the derivatives show minimal singlet-triplet splitting in the gas phase (close to 0.2 eV), and the high thermal stability confirmed by the compound's TGA method (320–398 °C) makes them very useful in the optoelectronic market.
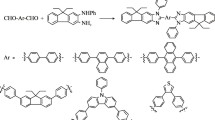
Graphical abstract
D-σ-π-σ-A based blue emitters carrying orthogonal triphenylamine and pyridine moiety as donor-acceptor unit at C-9 position were synthesized. Optical/solvatochromic studies, investigated by UV-VIS absorption and fluorescence spectroscopy, show blue emission with good quantum yield (40–70%) in solution and in thin film state for the synthesized compounds.







Similar content being viewed by others
References
(a) Tang C W and Slyke S A Van 1987 Organic electroluminescent diodes Appl. Phys. Lett. 51 913; (b) Jenekhe S A 1995 Excited-state complexes of conjugated polymers Adv. Mater. 7 309; (c) Miyata S and Nalwa H S (Eds.) 1997 Organic Electroluminescent Materials and Derivatives (Gordon and Breach: New York); (d) Kraft A, Grimsdale A C and Holms A B 1998 Electroluminescent Conjugated Polymers–Seeing Polymersina New Light Angew. Chem., Int. Ed. 37 402; (e) Shirota Y 2000 Organic materials for electronic and optoelectronic devices J. Mater. Chem. 10 1; (f) Segura J L 1998 The chemistry of electroluminescent organic materials Acta Polym. 49 319
Seo J H, Lee S C, Kim Y K and Kim Y S 2009 Efficient red electrophosphorescent organic lightemitting diodes based on the new sensitized heteroleptic tris-cyclometalated Ir(III) complexes Thin Solid Films 517 4119
Danel KS, Gąsiorski P, Matusiewicz M, Całus S, Uchacz T and Kityk A V 2010 UV–vis spectroscopy and semiempirical quantum chemical studies on methyl derivatives of annulated analogues of azafluoranthene and azulene dyes Spectrochim. Acta Part A 77 16
Całus S, Danel K S, Uchacz T and Kityk A V 2010 Optical absorption and fluorescence spectra of novel annulated analogues of azafluoranthene and azulene dyes Mater. Chem. Phys. 121 477
Wang B, Lv X, Tan J, Zhang Q, Huang Z, Yi W et al., 2016 Bipolar phenanthroimidazole–diazacarbazole hybrids with appropriate band gaps for highly efficient and low roll-off red, green and blue electroluminescent devices J. Mater. Chem. C 4 8473
Monkman A 2022 Why Do We Still Need a Stable Long Lifetime Deep Blue OLED Emitter? ACS Appl. Mater. Interfaces 14 20463
Uoyama H, Goushi K, Shizu K, Nomura H and Adachi C 2012 Highly Efficient Organic Light-Emitting Diodes from Delayed Fluorescence Nature 492 234
Wong M Y and Colman E Z 2017 Purely Organic Thermally Activated Delayed Fluorescence Materials for Organic Light-Emitting Diodes Adv. Mater. 29 1605444
Liu Y, Li C, Ren Z, Yan S and Bryce M R 2018 All-Organic Thermally Activated Delayed Fluorescence Materials for Organic Light-Emitting Diodes Nat. Rev. Mater. 3 18020
Bryden M A and Colman E Z 2021 Organic Thermally Activated Delayed Fluorescence (TADF) Compounds Used in Photocatalysis Chem. Soc. Rev. 50 7587
Nguyen V N, Kumar A, Lee M H and Yoon J 2020 Recent Advances in Biomedical Applications of Organic Fluorescence Materials with Reduced Singlet-Triplet Energy Gaps Coord. Chem. Rev. 425 213545
Goushi K, Yoshida K, Sato K and Adachi C 2012 Organic Light-Emitting Diodes Employing Efficient Reverse Intersystem Crossing for Triplet-to-Singlet State Conversion Nat. Photon. 6 253
Park I S, Komiyama H and Yasuda T 2017 Pyrimidine-based twisted donor–acceptor delayed fluorescence molecules: a new universal platform for highly efficient blue electroluminescence Chem. Sci. 8 953
Wang Y, Liu J, Yuan W, Wang Y, Zhou H, Liu X, Cao J and Zhang C 2019 Unique twisted donor–acceptor cruciform π-architectures exhibiting aggregation-induced emission and stimuli-response behaviours Dyes Pigm. 167 135
Karaman M, Gupta A K, Suresh S M, Matulaitis T, Mardegan L, Tordera D, et al. 2022 Ionic multiresonant thermally activated delayed fluorescence emitters for light emitting electrochemical cells Beilstein J. Org. Chem. 18 1311
Ahn D H, Lee H, Kim S W, Karthik D, Lee J, Jeong H, et al. 2019 Highly Twisted Donor–Acceptor Boron Emitter and High Triplet Host Material for Highly Efficient Blue Thermally Activated Delayed Fluorescent Device ACS Appl. Mater. Interfaces 11 14909
Keruckiene R, Guzauskas M, Volyniuk D, Matulis V E, Lyakhovc D A and Grazulevicius J V 2022 Theoretical and experimental insights into the properties of donor–r–acceptor type derivatives of quinoxaline and methanone containing different donor moieties New J. Chem. 46 20768
Wu Y, Zhu Y, Zhan Z, Zhao C, He J, Yan C and Meng H 2022 Narrowband Deep-Blue Multi-Resonance Induced Thermally Activated Delayed Fluorescence: Insights from the Theoretical Molecular Design Molecules 27 348
Park J, Lim J, Lee J H, Jang B, Han J H, Yoon S S and Lee J Y 2021 Asymmetric Blue Multi resonance TADF Emitters with a Narrow Emission Band ACS Appl. Mater. Interfaces 13 45798
Gao H, Shen S, Qin Y, Liu G, Gao T, Dong X, et al. 2022 Ultrapure Blue Thermally Activated Delayed Fluorescence (TADF) Emitters Based on Rigid Sulfur/Oxygen-Bridged Triaryl boron Acceptor: MR TADF and D−A TADF J. Phys. Chem. Lett. 13 7561
Chan C Y, Suresh S M, Lee Y T, Tsuchiya Y, Matulaitis T, Hall D, et al. 2022 Two boron atoms versus one: high-performance deep-blue multi-resonance thermally activated delayed fluorescence emitters Chem. Commun. 58 9377
Im Y, Kim M, Cho Y J, Seo J A, Yook K S and Lee J Y 2017 Molecular Design Strategy of Organic Thermally Activated Delayed Fluorescence Emitters Chem. Mater. 29 1946
Xia G, Qu C, Zhu Y, Ye J, Ye K, Zhang Z and Wang Y A 2021 TADF Emitter Featuring Linearly Arranged Spiro-Donor and Spiro Acceptor Groups: Efficient Nondoped and Doped Deep-Blue OLEDs with CIEy< 0.1 Angew. Chem. Int. Ed. 60 9598
Woo S J, Kim Y H and Kim J J 2021 Dihedral Angle Distribution of Thermally Activated Delayed Fluorescence Molecules in Solids Induces Dual Phosphorescence from Charge-Transfer and Local Triplet States Chem. Mater 33 5618
Song Y, Tian M, Yu R and He L 2021 Through-Space Charge-Transfer Emitters Developed by Fixing the Acceptor for High-Efficiency Thermally Activated Delayed Fluorescence ACS Appl. Mater. Interfaces 13 60269
Jayabharathi J, Ramya R, Thanikachalam V, Jeevaa P and Sarojpurani 2019 Efficient full-colour organic light-emitting diodes based on donor–acceptor electroluminescent materials with a reduced singlet–triplet splitting energy gap RSC Adv. 9 2948
Zhang X, Ji X, Jiang S, Liu L, Weeks B L and Zhang Z 2011 Highly efficient synthesis of 9-fluorenones from 9H-fluorenes by air oxidation Green Chem. 13 1891
Feng X, Hu JY, Tomiyasu H, Seto N, Redshaw C, Elsegoodd M R J and Yamato T 2013 Synthesis and photophysical properties of novel butterfly-shaped blue emitters based on pyrene Org. Biomol. Chem. 11 8366
Anandhan K, Ceron M, Perumal V, Ceballos P, Guerra P G, Gutierrez E P, Castillo A E, Thamotharanb S and Percino M J 2019 Solvatochromism and pH effect on the emission of triphenylimidazole-phenylacrylonitrile derivative: experimental and DFT studies RSC Adv. 9 12085
Zhang Y, Li D, Lia Y and Yu J 2014 Solvatochromic AIE luminoges as supersensitive waterdetectors in organic solvents and highly efficient cyanide chemosensors in water Chem. Sci. 5 2710
Nagy M, Fiser B, Ori M, Vanyorek L and Viskolcz B 2022 Optical study of solvatochromicisocyanoaminoanthracene Dyes and 1,5-diaminoanthracene Int. J. Mol. Sci. 23 1315
Li X, Baryshnikov G, Deng C, Bao X, Wu B, Zhou Y, et al. 2019 A three-dimensional ratiometric sensing strategy on unimolecular fluorescence–thermally activated delayed fluorescence dual emission Nat. Commun. 10 731
Serevicius T, Skaisgiris R, Dodonova J, Jagintavicius L, Banevicius D, Kazlauskas K, et al. 2020 Achieving Submicrosecond Thermally Activated Delayed Fluorescence Lifetime and Highly Efficient Electroluminescence by Fine-Tuning of the Phenoxazine−Pyrimidine Structure ACS Appl. Mater. Interfaces 12 10727
Luo Y, Zhao B, Zhang B, Lan Y, Chen L, Zhang Y, Bao Y and Niu Li 2022 The Scaffold of Thermally Activated Delayed Fluorescence Polymer Dots Towards Aqueous Electrochemiluminescence and Biosensing Application Analyst 147 2442
Pander P and Dias F B 2016 Photophysical Characterisation of Thermally Activated Delayed Fluorescence (TADF) Materials Display Imaging 00 1
Rajamalli P, Chen D, Suresh S M, Tsuchiya Y, Adachi C and Colman E Z 2021 Planar and Rigid Pyrazine-Based TADF Emitter for Deep Blue Bright Organic Light-Emitting Diodes Eur. J. Org. Chem. 2285
Yang J G, Song X F, Kaili J W, Chang X, Tan L Y, Liu C X, et al. 2021 Highly Efficient Thermally Activated Delayed Fluorescence from Pyrazine-Fused Carbene Au(I) Emitters Chem. Eur. J. 27 17834
Dawei Y, Xiaojuan Z, Zhiming W, Bing Y, Yuguang M and Yuyu P 2018 Theoretical investigation of the effects of various substituents on the large energy gap between triplet excited-states of anthracene RSC Adv. 8 27979
Acknowledgments
We are thankful to the Sophisticated Analytical Instrumental Facilities (SAIF), IIEST Shibpur, and India, for NMR & HRMS measurement & R.M. is thankful to the Indian Institute of Engineering Science and Technology, Shibpur, Howrah, India, for an Institute Fellowship.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Majumder, R., Jana, D. & Ghorai, B.K. Fluorene-based conjugates with geminal donor-acceptor: synthesis, photophysical properties and theoretical studies. J Chem Sci 136, 20 (2024). https://doi.org/10.1007/s12039-024-02249-7
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12039-024-02249-7