Abstract
The compounds trizImEA, Cu-trizImEA and Zn-trizImEA were synthesized and characterized by CHN, IR, NMR and mass measurements. They were interacted with CT DNA, and human telomere DNA, HTelo8 and HTelo20 to elucidate their binding capacity with different DNA structures. The interactions were followed by circular dichrosim (CD) measurements, fluorescent intercalator displacement (FID) assays and molecular docking studies through MOE program. The CD studies reveal the G-quadruplex stabilization of both HTelo8 and HTelo20 under salt conditions by the pure ligand, Cu-trizImEA and Zn-trizImEA. The compounds also induce and stabilize, selectively antiparallel G-quadruplex conformation on d(TTAGGG)n under no salt conditions. The binding constant (Kb) values calculated for the ligand and complexes are in the range of 5.3 × 104 M−1 to 9.4 × 105 M−1 revealing the strong binding. The FID assays clearly indicate the strong binding of the compounds on the G-quadruplex structure, as they displace thiazole orange (TO) which was bound to G-quadruplex already through specific intercalation. Molecular docking studies revealed classical intercalation as the preferred mode of interaction for all the three compounds in case of the stabilization towards antiparallel conformation. The compound's ability on antitelomerase activity and anticancer activities were performed and was found to be very effective in various cancer cell lines. The IC50 values observed were in the range of 50–100 nM for anticancer activity. Thus the compounds can be considered as effective anticancer agents as they stabilize G-quadruplex structures in a more facile and effective way towards antitelomerase action.
Graphical abstract
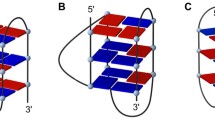
The pure ligand, Cu-trizImEA and Zn-trizImEA compounds were induce and stabilize, selectively antiparallel G-quadruplex conformation on d(TTAGGG)n under no salt conditions. The formed G-quadruplexes were thermodynamically stable and irreversible, thus resulting in proficient antitelomerase and anticancer activity, in-vitro.














Similar content being viewed by others
References
Shay J W and Wright W E 2019 Telomeres and telomerase: three decades of progress Nat. Rev. Genet. 20 299
Oganesian L and Bryan T M 2007 Physiological relevance of telomeric G-quadruplex formation: A potential drug target BioEssays 29 155
Hänsel-Hertsch R, Di Antonio M and Balasubramanian S 2017 DNA G-quadruplexes in the human genome: Detection, functions and therapeutic potential Nat. Rev. Mol. Cell Biol. 18 279
Müller S and Rodriguez R 2014 G-quadruplex interacting small molecules and drugs: From bench toward bedside Expert Rev. Clin. Pharmacol. 7 663
O’Hagan M P, Morales J C and Galan M C 2019 Binding and Beyond: What Else Can G-Quadruplex Ligands Do? Eur. J. Org. Chem. 2019 4995
Neidle S and Parkinson G 2002 Telomere maintenance as a target for anticancer drug discovery Nat. Rev. Drug Discov. 1 383
Dai J, Carver M, Punchihewa C, Jones R A and Yang D 2007 Structure of the hybrid-2 type intramolecular human telomeric G-quadruplex in K+ solution: Insights into structure polymorphism of the human telomeric sequence Nucleic Acids Res. 35 4927
Alessandrini I, Recagni M, Zaffaroni N and Folini M 2021 On the road to fight cancer: The potential of g-quadruplex ligands as novel therapeutic agents Int. J. Mol. Sci. 22 1
Awadasseid A and Ma X 2021 Wu Y and Zhang W 2021 G-quadruplex stabilization via small-molecules as a potential anti-cancer strategy Biomed. Pharmacother. 139 111550
Cao Q et al. 2017 G-quadruplex DNA targeted metal complexes acting as potential anticancer drugs Inorg. Chem. Front. 4 10
Asamitsu S, Obata S, Yu Z, Bando T and Sugiyama H 2019 Recent progress of targeted G-quadruplex-preferred ligands toward cancer therapy Molecules 24 429
Mitrasinovic P M 2020 G-quadruplexes: Emerging targets for the structure-based design of potential anti-cancer and antiviral therapies Acta Chim. Slov. 67 683
Kosiol N, Juranek S, Brossart P, Heine A and Paeschke K 2021 G-quadruplexes: a promising target for cancer therapy Mol. Cancer 20 1
Ruden M and Puri N 2013 Novel anticancer therapeutics targeting telomerase Cancer Treat. Rev. 39 444
Terenzi A et al. 2014 Selective G-quadruplex stabilizers: Schiff-base metal complexes with anticancer activity RSC Adv. 4 33245
Reed J E, Arnal A A, Neidle S and Vilar R 2006 Stabilization of G-quadruplex DNA and inhibition of telomerase activity by square-planar nickel(II) complexes J. Am. Chem. Soc. 128 5992
Müller S, Kumari S, Rodriguez R and Balasubramanian S 2010 Small-molecule-mediated G-quadruplex isolation from human cells Nat. Chem. 2 1095
Kim M, Vankayalapati H, Shin-ya K, Wierzba K and Hurley L H 2002 Telomestatin, a Potent Telomerase Inhibitor That Interacts Quite Specifically with the Human Telomeric Intramolecular G-Quadruplex JACS Commun. 124 2098
Zhang W J et al. 2007 9-Substituted berberine derivatives as G-quadruplex stabilizing ligands in telomeric DNA Bioorg. Med. Chem. 15 5493
Lavrado J, Reszka A P, Moreira R, Neidle S and Paulo A 2010 C-11 diamino cryptolepine derivatives NSC748392, NSC748393, and NSC748394: Anticancer profile and G-quadruplex stabilization Bioorg. Med. Chem. Lett. 20 7042
Hudson J S, Brooks S C and Graves D E 2009 Interactions of actinomycin D with human telomeric G-quadruplex DNA Biochemistry 48 4440
Majumder P and Dasgupta D 2011 Effect of DNA groove binder distamycin a upon chromatin structure PLoS ONE 6 e26486
Miglietta G, Russo M, Duardo R C and Capranico G 2021 G-quadruplex binders as cytostatic modulators of innate immune genes in cancer cells Nucleic Acids Res. 49 6673
Percivalle C et al. 2014 Aryl ethynyl anthraquinones: A useful platform for targeting telomeric G-quadruplex structures Org. Biomol. Chem. 12 3744
Martins C et al. 2007 Structure-based design of benzylamino-acridine compounds as G-quadruplex DNA telomere targeting agents Bioorg. Med. Chem. Lett. 17 2293
Haq I, Trent J O, Chowdhry B Z and Jenkins T C 1999 Intercalative G-Tetraplex Stabilization of Telomeric DNA by a Cationic Porphyrin J. Am. Chem. Soc. 9 1768
Le Vy Thi T, Han S, Chae J and Park H-J 2012 G-Quadruplex Binding Ligands: from Naturally Occurring to Rationally Designed Molecules Curr. Pharm. Des. 18 1948
Abd Karim N H et al. 2014 Salphen metal complexes as tunable G-quadruplex binders and optical probes RSC Adv. 4 3355
Gama S et al. 2016 Anthracene-terpyridine metal complexes as new G-quadruplex DNA binders J. Inorg. Biochem. 160 275
Reed J E, Neidle S and Vilar R 2007 Stabilisation of human telomeric quadruplex DNA and inhibition of telomerase by a platinum-phenanthroline complex Chem. Commun. 42 4366
Ali A, Kamra M, Roy S, Muniyappa K and Bhattacharya S 2016 Novel Oligopyrrole Carboxamide based Nickel(II) and Palladium(II) Salens, Their Targeting of Human G-Quadruplex DNA, and Selective Cancer Cell Toxicity Chem. Asian J. 11 2542
Dik-Lung M, Che C M and Yan S C 2009 Platinum(II) complexes with dipyridophenazine ligands as human telomerase inhibitors and luminescent probes for G-quadruplex DNA J. Am. Chem. Soc. 131 1835
Tuntiwechapikul W and Salazar M 2001 Cleavage of telomeric G-quadruplex DNA with perylene-EDTA·Fe(II) Biochemistry 40 13652
Randazzo A, Spada G P and da S M W 2012 Circular Dichroism of Quadruplex Structures Top. Curr. Chem. 310 1
Bhat H R, Masih A, Shakya A, Ghosh S K and Singh U P 2020 Design, synthesis, anticancer, antibacterial, and antifungal evaluation of 4-aminoquinoline-1,3,5-triazine derivatives J. Heterocycl. Chem. 57 390
Shelke M E 2021 Design, Synthesis, and Antimicrobial Evaluation of Novel 1,3,5-Triazine Derivatives Am. J. Pharmacol. 4 6
Yadav N and Jain P D A 2020 Biological Evaluation Of Synthesized Trsubstituted 1, 3, 5 Triazine Derivatives For Anti Bacterial Activity Int. J. Adv. Sci. Technol. 29 10477
Cascioferro S et al. 2017 1,3,5-Triazines: A promising scaffold for anticancer drugs development Eur. J. Med. Chem. 142 523
Mahdi M A, Jasim L S and Mohamed M H 2020 Synthesis and anticancer activity evaluation of novel ligand 2- [2 - (5-Chloro carboxy phenyl) Azo] 1-Methyl Imidazole (1-Mecpai) with Some Metal Complexes Syst. Rev. Pharm. 11 1979
De Hoog P, Gamez P, Driessen W L and Reedijk J 2002 New polydentate and polynucleating N-donor ligands from amines and 2,4,6-trichloro-1,3,5-triazine Tetrahedron Lett. 43 6783
Maheswari P U et al. 2006 Crystallographic evidence of nitrate-π interactions involving the electron-deficient 1,3,5-triazine ring Inorg. Chem. 45 6637
Wolfe A, Shimer G H and Meehan T 1987 Polycyclic aromatic hydrocarbons physically intercalate into duplex regions of denatured DNA Polycyclic Aromatic Hydrocarbons Physically Intercalate into Duplex Regions of Denatured DNA? Biochemistry 26 6392
Adams D M and Lock P J 1967 Copper-Halogen Stretching Frequencies J. Chem. Soc. 620
Balasubramanian S, Hurley L H and Neidle S 2011 Targeting G-quadruplexes in gene promoters: A novel anticancer strategy? Nat. Rev. Drug Discov. 10 261
Bhattacharjee A J et al. 2011 Induction of G-quadruplex DNA structure by Zn(II) 5,10,15,20-tetrakis(N- methyl-4-pyridyl)porphyrin Biochimie 93 1297
Sun L et al. 2014 Unfolding and conformational variations of thrombin-binding DNA aptamers: Synthesis, circular dichroism and molecular dynamics simulations ChemMedChem 9 993
Siters K E, Fountain M A and Morrow J R 2014 Selective Binding of Zn2+ Complexes to Human Telomeric G-Quadruplex DNA Inorg. Chem. 53 11540
Bonsignore R et al. 2016 G-quadruplex vs. duplex-DNA binding of nickel(II) and zinc(II) Schiff base complexes J. Inorg. Biochem. 161 115
Bonsignore R et al. 2018 The interaction of Schiff Base complexes of nickel(II) and zinc(II) with duplex and G-quadruplex DNA J. Inorg. Biochem. 178 106
Fu B et al. 2008 Cationic metal-corrole complexes: Design, synthesis, and properties of guanine-quadruplex stabilizers Chem. A Eur. J. 14 9431
Xu L et al. 2010 Trinuclear Ru(II) polypyridyl complexes as human telomeric quadruplex DNA stabilizers Inorg. Chem. Commun. 13 1050
Roshatiara S, Sahudin M A, Hassan N H and Karim N H A 2017 Interaction of n, n’-bis[4-[1-(2-hydroxyethoxy)] salicylidene]-phenyldiamine- nickel(II) and copper(II) complexes with G-quadruplex DNA Malays J. Anal. Sci. 21 544
Monchaud D, Allain C and Teulade-Fichou M P 2006 Development of a fluorescent intercalator displacement assay (G4-FID) for establishing quadruplex-DNA affinity and selectivity of putative ligands Bioorg. Med. Chem. Lett. 16 4842
Largy E, Hamon F and Teulade-Fichou M P 2011 Development of a high-throughput G4-FID assay for screening and evaluation of small molecules binding quadruplex nucleic acid structures Anal. Bioanal. Chem. 400 3419
Lubitz I, Zikich D and Kotlyar A 2010 Specific high-affinity binding of thiazole orange to triplex and g-quadruplex DNA Biochemistry 49 3567
Parkinson G N, Lee M P H and Neidle S 2002 Crystal structure of parallel quadruplexes from human telomeric DNA Nature 417 876
Wilson T, Costa P J, Félix V, Williamson M P and Thomas J A 2013 Structural studies on dinuclear ruthenium(II) complexes that bind diastereoselectively to an antiparallel folded human telomere sequence J. Med. Chem. 56 8674
Lin C et al. 2018 Molecular Recognition of the Hybrid-2 Human Telomeric G-Quadruplex by Epiberberine: Insights into Conversion of Telomeric G-Quadruplex Structures Angew. Chem. Int. Ed. 57 10888
Mender I and Shay J W 2016 Telomerase Repeat Amplification Protocol (TRAP) Bio-Protocol 5 e1657
Shin-ya K et al. 2001 Telomestatin, a novel telomerase inhibitor from Streptomyces anulatus [17] J. Am. Chem. Soc. 123 1262
Maheswari P U et al. 2006 The square-planar cytotoxic [CuII(pyrimol)Cl] complex acts as an efficient DNA cleaver without reductant J. Am. Chem. Soc. 128 710
Li L et al. 2019 Design, synthesis, and biological evaluation of 1-substituted -2-aryl imidazoles targeting tubulin polymerization as potential anticancer agents Eur. J. Med. Chem. 184 111732
Bin Sayeed I, Vishnuvardhan M V P S, Nagarajan A, Kantevari S and Kamal A 2018 Imidazopyridine linked triazoles as tubulin inhibitors, effectively triggering apoptosis in lung cancer cell line Bioorg. Chem. 80 714
Narasimha Rao M P et al. 2018 Synthesis of imidazo-thiadiazole linked indolinone conjugates and evaluated their microtubule network disrupting and apoptosis inducing ability Bioorg. Chem. 76 420
Acknowledgements
Prof. Raghavan, MBU, IISc., Bangalore is thanked for CD facility. DST-WOS-A (SR/WOS-A/CS-75/2016), India is acknowledged for financial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Duraisamy, R., Palanisamy, U., Sheriffa Begum, K.M.M. et al. Facile induction and stabilization of intramolecular antiparallel G-quadruplex of d(TTAGGG)n on interaction with triazine-2-imidazole ethyl amine compound and its Cu(II), Zn(II) complexes under no-salt conditions. J Chem Sci 134, 12 (2022). https://doi.org/10.1007/s12039-021-01996-1
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12039-021-01996-1