Abstract
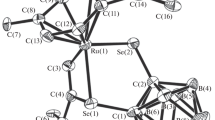
The reaction of [Ru(η 6-cymene)Cl2]2 and PPh2Cl in the ratio 1:2 gives a stable [Ru(η 6-cymene) Cl2(PPh2Cl)] complex. Attempts to make the cationic [Ru(η 6-cymene)Cl(PPh2Cl)2]Cl with excess PPh2Cl and higher temperatures led to adventitious hydrolysis and formation of [Ru(η 6-cymene)Cl2(PPh2OH)]. Attempts to make a phosphinite complex by reacting [Ru(η 6-cymene)Cl2]2 with PPh2Cl in the presence of an alcohol results in the reduction of PPh2Cl to give [Ru(η 6-cymene)Cl2(PPh2H)] and the expected phosphinite. The yield of the hydride complex is highest when the alcohol is 1-phenyl-ethane-1,2-diol. All three half-sandwich complexes are characterized by X-ray crystallography. Surprisingly, the conversion of chlorodiphenylphosphine to diphenylphosphine is mediated by 1-phenyl-ethane-1,2-diol even in the absence of the ruthenium half-sandwich precursor.

Synthesis and unusual reactivity of some ruthenium half-sandwich complexes including an unusual organocatalysed conversion of PPh2Cl to PPh2H are reported.














Similar content being viewed by others
References
Fey N, Orpen A G and Harvey J N 2009 Coord. Chem. Rev. 253 704
Goodman J and Macgregor S A 2010 Coord. Chem. Rev. 254 1295
Ciancaleoni G, Scafuri N, Bistoni G, Macchioni A, Tarantelli F, Zuccaccia D and Belpassi L 2014 Inorg. Chem. 53 9907
McMullin C L, Fey N and Harvey J N 2014 Dalton Trans. 43 13545
Ficks A, Clegg W, Harrington R W and Higham L J 2014 Organometallics 33 6319
Truzzi D R and Franco D W 2014 Polyhedron 81 238
Clarke M L and Frew J J R 2009 In Organometallic Chemistry Vol. 35 (London: The Royal Society of Chemistry) pp. 19–46
Brisdon A K and Herbert C J 2013 Coord. Chem. Rev. 257 880
Fei Z, Scopelliti R and Dyson P J 2003 Inorg. Chem. 42 2125
Gray G M and Kraihanzel C S 1982 J. Organomet. Chem. 238 209
Wong E H and Bradley F C 1981 Inorg. Chem. 20 2333
Gray G M and Kraihanzel C S 1978 J. Organomet. Chem. 146 23
Ibrahim Sk Md, Ganesamoorthy C and Balakrishna M S 2013 Ind. J. Chem. 52 A 1400
Esteban M, Pequerul A, Carmona D, Lahoz F J, Marlin A and Oro L A 1999 J. Organomet. Chem. 402 421
Treichel P M and Rosenhein L D 1981 Inorg. Chem. 20 1539
Pereira R M S, Paula V I, Buffon R, Tomazela D M and Eberlin M N 2004 Inorg. Chim. Acta 357 2100
Khan H, Badshah A, Said M, Murtaza G, Ahmad J, Jean-Claude B J, Todorova M and Butler I S 2013 Appl. Organomet. Chem. 27 387
Torres-Lubián R, Rosales-Hoz M J, Arif A M, Ernst R D and Paz-Sandoval M A 1999 J. Organomet. Chem. 585 68
Jantscher F, Kirchner K and Mereiter K 2009 Acta Cryst. E65 m941
Sharma R K, Nethaji M and Samuelson A G 2008 Tetrahedron: Asymmetry 19 655
Sharma R K and Samuelson A G 2007 Tetrahedron: Asymmetry 18 2387
Bergamini P, Bertolasi V and Giordani R 2005 Inorg. Chim. Acta 358 2031
Song L -C, Zeng G -H, Lou S -X, Zan H -N, Ming J -B and Hu Q -M 2008 Organometallics 27 3714
Suss-Fink G 2014 J. Organomet. Chem. 751 2
Kechaou-Perrot M, Vendier L, Bastin S, Sotiropoulos J M, Miqueu K, Menendez-Rodriguez L, Crochet P, Cadierno V and Igau A 2014 Organometallics 33 6294
Kumar P, Gupta R K and Pandey D S 2014 Chem. Soc. Rev. 43 707
Aydemir M, Meric N, Baysal A, Turgut Y, Kayan C, Seker S, Togrul M and Gümgüm B 2011 J. Organomet. Chem. 696 1541
Sheeba M M, Tamizh M M, Farrugia L J, Endo A and Karvembu R 2014 Organometallics 33 540
Bugarcic T, Habtemariam A, Deeth R J, Fabbiani F P A, Parsons S and Sadler P J 2009 Inorg. Chem. 48 9444
Reddy A R, Ranjini A S, Das P K and Samuelson A G 2007 Inorg. Chim. Acta 360 2778
De S, Mitra R, Samuelson A G and Das P K 2015 J. Organomet. Chem. 785 72
Siemens 1995 Area Detector Control and Integration Software. Siemens Analytical X-ray Instruments Inc., Madison, Wisconsin, USA.
Sheldrick G 1993 In SADABS User Guide (University of Gottingen: Gottingen, Germany)
Sheldrick G 2008 Acta. Cryst. A 64 112
Ikariya T and Blacker A J 2007 Acc. Chem. Res. 40 1300
Noyori R and Hashiguchi S 1997 Acc. Chem. Res. 30 97
Ouellet S G, Walji A M and Macmillan D W C 2007 Acc. Chem. Res. 40 1327
Li G and Antilla J C 2009 Org. Lett. 11 1075
Martin N J A, Ozores L and List B 2007 J. Am. Chem. Soc. 129 8976
Ouellet S G, Tuttle J B and MacMillan D W C 2005 J. Am. Chem. Soc. 127 32
Tuttle J B, Ouellet S G and MacMillan D W C 2006 J. Am. Chem. Soc. 128 12662
Gulyás H, Benet-Buchholz J, Escudero-Adan E C, Freixa Z and van Leeuwen P W N M 2007 Chem. Eur. J. 13 3424
Acknowledgements
Financial support from the Department of Science and Technology, New Delhi vide Project number, SR/S5/MBD-02/2007, is gratefully acknowledged. AKP acknowledges CSIR, New Delhi for a SRF Fellowship.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information
CCDC 884833, 884834 and 880813 contain the supplementary crystallographic data for [Ru(η 6-cymene) Cl2(PPh2Cl)] 2 [Ru(η 6-cymene)Cl2(PPh2OH)] 4 and [Ru(η 6-cymene)Cl2(PPh2H)] 5, respectively. These data can be obtained free of charge from The Cambridge Crystallographic Data Centre via www.ccdc.cam.ac.uk/data_request/cif. NMR spectra of complexes (figures S1-S15) are available at www.ias.ac.in/chemsci.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
PANDIAKUMAR, A.K., SAMUELSON, A.G. Synthesis and unexpected reactivity of [Ru(η 6-cymene)Cl2(PPh2Cl)], leading to [Ru(η 6-cymene)Cl2(PPh2H)] and [Ru(η 6-cymene)Cl 2 (PPh2OH)] complexes. J Chem Sci 127, 1329–1338 (2015). https://doi.org/10.1007/s12039-015-0905-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12039-015-0905-z