Abstract
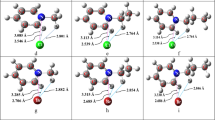
Halogen bonding interactions of type X⋯O=C are important in various fields including biological systems. In this work, theoretical calculations were carried out using B3LYP/6-31 ++G**, MP2/6-31 ++G** and MP2/aug-cc-pVDZ methods on a series of O⋯X halogen bonds between CH2O and CH3CHO as halogen bond acceptor with X-Y (X = Cl, Br; Y = CF3, CF2H, CFH2, CN, CCH, CCCN) as halogen bond donors. The strength of interaction energy for O⋯Br halogen-bonded complexes varies from −2.16 to −5.26 kcal/mol while for O⋯Cl complexes, it is between −1.65 to −3.67 kcal/mol, which indicate the O⋯Br bond to be stronger in comparison to O⋯Cl bond. SAPT analysis suggests that the strength of halogen bonding arises from the electrostatic and induction forces while dispersion is playing a comparatively smaller role. The halogen-bonded interaction energies were found to correlate well with positive electrostatic potential VS,max, halogen bonded distances, and the change in s-character of C-X bond. The halogen-bonded interaction energies were also evaluated for O⋯I bonded complexes and thus these complexes were found to be stronger than O⋯Br and O⋯Cl bonded complexes.

Theoretical calculations were carried out on halogen bonded complexes CH2O⋯X-Y and CH3CHO⋯X-Y (X = Cl, Br, I; Y = CF3, CF2H, CFH2, CN, CCH, CCCN). The interaction energies increase in the order O⋯Cl < O⋯Br < O⋯I. The interaction energies were found to be correlate well with the VS,max, halogen bonded distances, and the change in s-character of C-X bond.











Similar content being viewed by others
References
Legon A C 2010 Phys. Chem. Chem. Phys. 12 7736
Politzer P, Lane P, Concha M C, Ma Y and Murray J S 2007 J. Mol. Model. 13 305
Hauchecorne D, Szostak R, Herrebout W A and van der Veken B J 2009 ChemPhysChem 10 2105
Hauchecorne D, van der Veken B J, Moiana A and Herrebout W A 2010 Chem. Phys. 374 30
Hauchecorne D, Moiana A, van der Veken B J and Herrebout W A 2011 Phys. Chem. Chem. Phys. 13 10204
Politzer P and Murray J S 2013 ChemPhysChem 14 278
Troff R W, Mäkelä T, Topić F, Valkonen A, Raatikainen K and Rissanen K 2013 Eur. J. Org. Chem. 2013 1617
Metrangolo P and Resnati G 2012 Cryst. Growth Des. 12 5835
Jentzsch A V, Emery D, Mareda J, Nayak S K, Metrangolo P, Resnati G, Sakai N and Matile S 2012 Nat. Commun. 3 905
Riley K E and Hobza P 2013 Acc. Chem. Res. 46 927
Yang X, Gan L, Han L, Wang E and Wang J 2013 Angew. Chem., Int. Ed. Engl. 52 2022
Poznanski J and Shugar D 2013 Biochim. Biophys. Acta 1834 1381
Metrangolo P, Neukirch H, Pilati T and Resnati G 2005 Acc. Chem. Res. 38 386
Alkorta I, Blanco F, Deya P M, Elguero J, Estarellas C, Frontera A and Quinonero D 2009 Theor. Chem. Acc. 126 1
Mooibroek T J and Gamez P 2013 CrystEngComm. 15 1802
Meazza L, Foster J A, Fucke K, Metrangolo P, Resnati G and Steed J W 2013 Nat. Chem. 5 42
Khavasi H R and Azhdari Tehrani A 2013 Inorg. Chem. 52 2891
Metrangolo P, Meyer F, Pliati T, Resnati G and Terraneo G 2008 Angew. Chem., Int. Ed. Engl. 47 6114
Rosokha S V and Vinakos M K 2012 Cryst. Growth Des. 12 4149
Politzer P, Murray J S and Concha M C 2008 J. Mol. Model. 14 659
Clark T, Hennemann M, Murray J S and Politzer P 2007 J. Mol. Model. 13 291
Murray J S, Lane P, Clark T and Politzer P 2007 J. Mol. Model. 13 1033
Murray J S, Concha M C, Lane P, Hobza P and Politzer P 2008 J. Mol. Model. 14 699
Murray J S, Lane P and Politzer P 2009 J. Mol. Model. 15 723
Politzer P, Murray J S and Concha M C 2007 J. Mol. Model. 13 643
Legon A C 1999 Angew. Chem., Int. Ed. Engl. 38 2686
Metrangolo P, Murray J S, Pliati T, Politzer P, Resnati G and Terraneo G 2011 CrystEngComm. 13 6593
Chopra D and Guru Row T N 2011 CrystEngComm. 13 2175
Pinter B, Nagels N, Herrebout W A and De Proft F 2013 Chem. Eur. J. 19 519
Glaser R, Chen N J, Wu H, Knotts N and Kaupp M 2004 J. Am. Chem. Soc. 126 4412
Romaniello P and Lelj F 2002 J. Phys. Chem. A. 106 9114
Valeiro G, Raos G, Meille S V, Metrangolo P and Resnati G 2000 J. Phys. Chem. A. 104 1617
Sarwar M G, Dragisic B, Dimitrijevic E and Taylor M S 2013 Chem. Eur. J. 19 2050
Zou J W, Jiang Y J, Guo M, Hu G X, Zhiang B, Liu H C and Yu Q S 2005 Chem. Eur. J. 11 740
Larsen D W and Allred A L 1965 J. Am. Chem. Soc. 87 1216
Larsen D W and Allred A L 1965 J. Am. Chem. Soc. 87 1219
Amezaga N J M, Pamies S C, Peruchena N M and Sosa G L 2010 J. Phys. Chem. A. 114 552
Battistutta R, Mazzorana M, Sarno S, Kazimierczuk Z, Zanotti G and Pinna L A 2005 Chem. Biol. 12 1211
Ghosh M, Meerts I A T M, Cook A, Bergman A, Brouwer A and Johnson L N 2000 Acta. Crystallogr., Sect. D: Biol. Crystallogr. 56 1085
Jiang Y, Alcaraz A A, Chen J M, Kobayashi H, Lu Y J and Synder J P 2006 J. Med. Chem. 49 1891
Lopez-Rodriguez M L, Murcia M, Benhamu B, Viso A, Campillo M and Pardo L 2002 J. Med. Chem. 45 4806
Auffinger P, Hays F A and Westhof E S H P 2004 Proc. Natl. Acad. Sci. USA 101 16789
Riley K E and Hobza P 2007 J. Chem. Theory Comput. 4 232
Riley K E, Murray J S, Politzer P, Concha M C and Hobza P 2009 J. Chem. Theory Comput. 5 155
Zierkiewicz W, Wieczorek R, Hobza P and Michalska D 2011 Phys. Chem. Chem. Phys. 13 5105
Li Q, Xu X, Liu T, Jing B, Li W, Cheng J, Gong B and Sun J 2010 Phys. Chem. Chem. Phys. 12 6837
Boys S F, Moran D and Radom L 2007 J. Phys. Chem. A. 111 11683
Frisch M J, Trucks G W, Schlegel H B, Scuseria G E, Robb M A, Cheeseman J R, Scalmani G, Barone V, Mennucci B, Petersson G A, Nakatsuji H, Caricato M, Li X, Hratchian H P, Izmaylov A F, Bloino J, Zheng G, Sonnenberg J L, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, KItao O, Nakai H, Vreven T, Montgomery J A, Peralta J E, Ogliaro F, Bearpark M, Heyd J J, Brothers E, Kudin K N, Staroverov V N, Kobayashi R, Normand J, Raghavachari K, Rendel A, Burant J C, Iyengar S S, Tomasi J, Cossi M, Rega N, Millam J M, Klene M, Knox J E, Cross J B, Bakken V, Adamo C, Jaramillo C, Gomperts R, Stratmann R E, Yazyev O, Austin A J, Cammi R, Pomelli C, Ochterski J W, Martin R L, Morokuma K, Zakrzewski V G, Voth G A, Salvador P, Dannenberg J J, Dapprich S, Daniels A D, Farkas O, Foresman J B, Ortiz J V, Cioslowski J and Fox D J Gaussian, Inc.: Wallingford CT, 2009
Bulat F A, Toro-Labbe A, Brinck T, Murray J S and Politzer P 2010 J. Mol. Model. 16 1679
Politzer P and Truhlar D G 1981 In Chemical Applications of Atomic and Molecular Electrostatic Potentials (New York: Plenum)
Reed A E, Curtiss L A and Weinhold F 1988 Chem. Rev. 88 899
Bader R F W 1990 In Atoms in Molecules: A Quantum Theory (Oxford: Clarendon Press)
Bader R F W 1991 Chem. Rev. 91 893
Bader R F W 1998 In Encyclopedia of Computational Chemistry Schleyer P V R, Allinger N L T, Gasteiger C J, Kollman P A, Schaefer H F S, III, Schreiner P R, (Chichester: Wiley) vol. 1, p 64
Biegler-König F and Schönbohm, J 2002 AIM2000: Version 2.0, Germany
Jeziorski B, Moszyński R and Szalewicz K 1994 Chem. Rev. 94 1887
Moszyński R, Heijmen T G A and Jeziorski B J 1996 Mol. Phys. 88 741
Bukowski R, Cencek W, Jankowski P, Jeziorska M, Jeziorski B, Kucharski SA, Lotrich V F, Misquitta A J, Moszyński R, Patkowski K, Podeszwa R, Rybak S, Szalewicz K, Williams H L, Wheatley R J, Wormer P E S and żuchowski P S 2008 University of Delaware and University of Warsaw: Newark (DE) and Warsaw
Naray-Szabo G and Ferenczy G G 1995 Chem. Rev. 95 829
Murray J S and Politzer P 1998 THEOCHEM 425 107
Politzer P, Murray J S and Concha M C 2002 Int. J. Quantum Chem. 88 19
Bader R F W, Carroll M T, Chesseman J R and Chang C 1987 J. Am. Chem. Soc. 109 7968
Brick T, Murray J S and Politzer P 1992 Int. J. Quantum Chem. 44 57
Murray-Rust P, Stallings W C, Monti C T, Preston R K and Glusker J P 1983 J. Am. Chem. Soc. 105 3206
Ramasubbu N, Parthasartahy P and Murray-Rust P 1986 J. Am. Chem. Soc. 108 4308
Murray-Rust P and Motherwell W D S 1979 J. Am. Chem. Soc 101 4374
Riley K E, Murray J S, Fanfrlik J, Rezac J, Sola R J, Concha M C, Ramos F M and Politzer P 2011 J. Mol. Model. 17 3309
Shields Z P, Murray J S and Politzer P 2010 Int. J. Quantum Chem. 110 2823
Keefe C D and Istvankova Z 2011 Can. J. Chem. 89 34
Kaur D and Khanna S 2011 THEOCHEM 963 71
Kaur D and Khanna S 2012 Struct. Chem. 23 755
Kaur D, Khanna S and Aulakh D 2013 Struct. Chem. 24 357
Kaur D, Sharma R and Aulakh D 2011 Struct. Chem. 22 1015
Syzgantseva O A, Tognetti V and Joubert L 2013 J. Phys. Chem. A 117 8969
Duarte D J, Sosa G L and Peruchena N M 2013 J. Mol. Model. 19 2035
Zeng Y, Zhang X, Li X, Zheng S and Meng L 2010 Int. J. Quantum Chem. 3725
Madzhidov T I, Chmutova G A and Martin Pendas A 2011 J. Phys. Chem. A 115 10069
Koch U and Popelier P L A 1995 J. Phys. Chem. 99 9747
Mata I, Alkorta I, Molins E and Espinosa E 2010 Chem. Eur. J. 16 2442
Alkorta I and Elguero J 2004 Struct. Chem. 15 117
Jabloński M and Palusiak M 2012 J. Phys. Chem. A 116 2322
Peterson K A, Figgen D, Goll E, Stoll H and Dolg M 2003 J. Chem. Phys. 119 11113
Moller C and Plesset M S 1934 Phys. Rev. 46 618
Dunning T H 1989 J. Chem. Phys. 90 1007
Wood D E and Dunning T H 1995 J. Chem. Phys. 103 4572
Acknowledgement
We are highly thankful to the Department of Science and Technology (DST) (INSPIRE Fellowship Programme) for the financial assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information
The differences of the halogen bonding distances between the values in the complex and in the isolated molecule for C-X bond of X-Y molecules and for C=O bond of carbonyl molecules are given in table S1 at MP2/aug-cc-pVDZ level. Tables S2–S19 include the optimized parameters for the CH2O and CH3CHO along with their complexes with X-Y (X = Cl, Br; Y = CF3, CF2H, CFH2, CN, CCH and CCCN) at B3LYP/6-31 ++G** and MP2/aug-cc-pVDZ theoretical levels. Table S21 lists the percentage s-character of C in C-X bond of the halogen donors for the monomers and the complexes at MP2/aug-cc-pVDZ level. The atomic charges on the halogen bonding atoms obtained using NBO analysis at MP2/aug-cc-pVDZ level are given in table S22. Figure S1 shows a correlation between SAPT-based interaction energies and interaction energies obtained at MP2/cc-pVDZ level for both CH2O and CH3CHO complexes under study. The optimized parameters for O ⋯I bond complexes of CH2O and CH3CHO at MP2/cc-pVDZ-PP level are reported in tables S23–S28.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
KAUR, D., KAUR, R. Theoretical study on O⋯Br and O⋯Cl halogen bonds in some small model molecular systems. J Chem Sci 126, 1763–1779 (2014). https://doi.org/10.1007/s12039-014-0717-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12039-014-0717-6