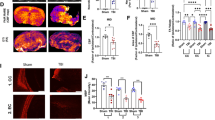
Abstract
Harmful stimuli trigger mutations lead to uncontrolled accumulation of hnRNPA2/B1 in the cytoplasm, exacerbating neuronal damage. Kapβ2 mediates the bidirectional transport of most substances between the cytoplasm and the nucleus. Kapβ2 guides hnRNPA2/B1 back into the nucleus and restores its function, alleviating related protein toxicity. Here, we aim to explore the involvement of Kapβ2 in neurodegeneration in rats with MCI following sevoflurane anesthesia and surgery. Firstly, novel object recognition test and Barnes maze were conducted to assess behavioral performances, and we found Kapβ2 positively regulated the recovery of memory and cognitive function. In vivo electrophysiological experiments revealed that the hippocampal theta rhythm energy distribution was disrupted, coherence was reduced, and long-term potentiation was attenuated in MCI rats. LTP was greatly improved with positive modulation of Kapβ2. Next, functional MRI and BOLD imaging will be employed to examine the AFLL and FC values of dynamic connectivity between the cortex and hippocampus of the brain. The findings show that regulating Kapβ2 in the hippocampus region enhances functional activity and connections between brain regions in MCI rats. WB results showed that increasing Kapβ2 expression improved the expression and recovery of cognitive-related proteins in the hippocampus of MCI rats. Finally, WB and immunofluorescence were used to examine the changes in hnRNPA2/B1 expression in the nucleus and cytoplasm after overexpression of Kapβ2, and it was found that nucleocytoplasmic mis location was alleviated. Overall, these data show that Kapβ2 reverses the nucleoplasmic misalignment of hnRNPA2/B1, which slows neurodegeneration towards dementia in MCI after sevoflurane anesthesia and surgery. Our findings may lead to new approaches for perioperative neuroprotection of MCI patients.








Similar content being viewed by others
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Petersen RC (2016) Mild cognitive impairment. CONTINUUM: Lifelong Learn Neurol 22:404–418
Langa KM, Levine DA (2014) The diagnosis and management of mild cognitive impairment: a clinical review. JAMA 312:2551
Brodaty H, Connors MH, Ames D et al (2014) Progression from mild cognitive impairment to dementia: A 3-year longitudinal study. Aust N Z J Psychiatry 48:1137–1142
Meles SK, Pagani M, Arnaldi D et al (2017) The Alzheimer’s disease metabolic brain pattern in mild cognitive impairment. J Cereb Blood Flow Metab 37:3643–3648
Roberts RO, Knopman DS, Mielke MM et al (2014) Higher risk of progression to dementia in mild cognitive impairment cases who revert to normal. Neurology 82:317–325
Kandiah N, Chan YF, Chen C et al (2021) Strategies for the use of Ginkgo biloba extract, EGb 761 ®, in the treatment and management of mild cognitive impairment in Asia: Expert consensus. CNS Neurosci Ther 27:149–162
Thomas KR, Osuna JR, Weigand AJ et al (2021) Regional hyperperfusion in older adults with objectively-defined subtle cognitive decline. J Cereb Blood Flow Metab 41:1001–1012
Gao S, Unverzagt FW, Hall KS et al (2014) Mild cognitive impairment, incidence, progression, and reversion: findings from a community-based cohort of elderly african americans. Am J Geriatr Psychiatry 22:670–681
Pappa M, Theodosiadis N, Tsounis A, Sarafis P (2017) Pathogenesis and treatment of post-operative cognitive dysfunction. Electron Physician 9:3768–3775. https://doi.org/10.19082/3768
Evered L, Silbert B, Knopman DS et al (2018) Recommendations for the nomenclature of cognitive change associated with anaesthesia and surgery—2018. Anesthesiology 129:872–879
Wan Y, Xu J, Ma D et al (2007) Postoperative impairment of cognitive function in rats. Anesthesiology 106:436–443
Monk TG, Weldon BC, Garvan CW et al (2008) Predictors of cognitive dysfunction after major noncardiac surgery. Anesthesiology 108:18–30
Li K, Guo Z-W, Zhai X-M et al (2020) RBPTD: a database of cancer-related RNA-binding proteins in humans. Database 2020:baz156
Wolozin B, Ivanov P (2019) Stress granules and neurodegeneration. Nat Rev Neurosci 20:649–666
Loïodice I, Alves A, Rabut G et al (2004) The entire Nup107-160 complex, including three new members, is targeted as one entity to kinetochores in mitosis. MBoC 15:3333–3344
Soniat M, Chook YM (2015) Nuclear localization signals for four distinct karyopherin-β nuclear import systems. Biochem J 468:353–362
Görlich D, Kutay U (1999) Transport between the cell nucleus and the cytoplasm. Annu Rev Cell Dev Biol 15:607–660
Xu D, Farmer A, Chook YM (2010) Recognition of nuclear targeting signals by Karyopherin-β proteins. Curr Opin Struct Biol 20:782–790
Lee BJ, Cansizoglu AE, Süel KE et al (2006) Rules for nuclear localization sequence recognition by Karyopherinβ2. Cell 126:543–558
Won J-S, Kim J, Annamalai B et al (2013) Protective role of S-Nitrosoglutathione (GSNO) against cognitive impairment in rat model of chronic cerebral hypoperfusion. JAD 34:621–635
Duan W, Chun-Qing Z, Zheng J et al (2011) Relief of carotid stenosis improves impaired cognition in a rat model of chronic cerebral hypoperfusion. Acta Neurobiol Exp (Wars) 71:233–243
Zhou Z, Zhang Y, Zhu C et al (2012) Cognitive functions of carotid artery stenosis in the aged rat. Neuroscience 219:137–144
Harry LE, Sandison A, Paleolog EM et al (2008) Comparison of the healing of open tibial fractures covered with either muscle or fasciocutaneous tissue in a murine model. J Orthop Res 26:1238–1244
Bokil H, Andrews P, Kulkarni JE et al (2010) Chronux: a platform for analyzing neural signals. J Neurosci Methods 192:146–151
Yan C-G, Wang X-D, Zuo X-N, Zang Y-F (2016) DPABI: data processing & analysis for (Resting-State) brain imaging. Neuroinform 14:339–351
Papazoglou A, Soos J, Lundt A et al (2016) Gender-specific hippocampal dysrhythmia and aberrant hippocampal and cortical excitability in the APPswePS1dE9 model of alzheimer’s disease. Neural Plast 2016:1–16
Silbert B, Evered L, Scott DA (2011) Cognitive decline in the elderly: Is anaesthesia implicated? Best Pract Res Clin Anaesthesiol 25:379–393
Roberts R, Knopman DS (2013) Classification and epidemiology of MCI. Clin Geriatr Med 29:753–772
McKhann GM, Knopman DS, Chertkow H et al (2011) The diagnosis of dementia due to Alzheimer’s disease: Recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement 7:263–269
Wang J, Yang C, Wang H et al (2020) A new rat model of chronic cerebral hypoperfusion resulting in early-stage vascular cognitive impairment. Front Aging Neurosci 12:86
Smith I, Nathanson M, White PF (1996) Sevoflurane-a long-awaited volatile anaesthetic. Br J Anaesth 76:435–445
Culley DJ, Xie Z, Crosby G (2007) General anesthetic-induced neurotoxicity: an emerging problem for the young and old? Curr Opin Anaesthesiol 20:408–413
Satomoto M, Satoh Y, Terui K et al (2009) Neonatal exposure to sevoflurane induces abnormal social behaviors and deficits in fear conditioning in mice. Anesthesiology 110:628–637
Shen X, Liu Y, Xu S et al (2013) Early life exposure to sevoflurane impairs adulthood spatial memory in the rat. Neurotoxicology 39:45–56
Wang J, Zhang L, Huang Q et al (2017) Monitoring the end-tidal concentration of sevoflurane for preventing awareness during anesthesia (MEETS-PANDA): A prospective clinical trial. Int J Surg 41:44–49
Logginidou HG, Li B-H, Li D-P et al (2003) Propofol suppresses the cortical somatosensory evoked potential in rats. Anesth Analg 97:1784–1788
Rosenfeld CS, Ferguson SA (2014) Barnes maze testing strategies with small and large rodent models. JoVE 84:51194
Cohen SJ, Stackman RW Jr (2015) Assessing rodent hippocampal involvement in the novel object recognition task. A review. Behav Brain Res 285:105–117
Perl DP (2010) Neuropathology of alzheimer’s disease: D. P. PERL: NEUROPATHOLOGY OF ALZHEIMER’S DISEASE. Mt Sinai J Med 77:32–42
Kylkilahti TM, Berends E, Ramos M et al (2021) Achieving brain clearance and preventing neurodegenerative diseases—A glymphatic perspective. J Cereb Blood Flow Metab 41:2137–2149
Sun S, Zhou J, Li Z et al (2022) Progranulin promotes hippocampal neurogenesis and alleviates anxiety-like behavior and cognitive impairment in adult mice subjected to cerebral ischemia. CNS Neurosci Ther 28:775–787
Burgess N, Maguire EA, O’Keefe J (2002) The human hippocampus and spatial and episodic memory. Neuron 35:625–641
Suo Z, Yang J, Zhou B et al (2022) Whole-transcriptome sequencing identifies neuroinflammation, metabolism and blood–brain barrier related processes in the hippocampus of aged mice during perioperative period. CNS Neurosci Ther 28:1576–1595
Buzsáki G, Draguhn A (2004) Neuronal oscillations in cortical networks. Science 304:1926–1929
Colgin LL (2013) Mechanisms and functions of theta rhythms. Annu Rev Neurosci 36:295–312
Tuesta LM, Zhang Y (2014) Mechanisms of epigenetic memory and addiction. EMBO J 33:1091–1103
Biswal B, ZerrinYetkin F, Haughton VM, Hyde JS (1995) Functional connectivity in the motor cortex of resting human brain using echo-planar mri. Magn Reson Med 34:537–541
Pan P, Zhu L, Yu T et al (2017) Aberrant spontaneous low-frequency brain activity in amnestic mild cognitive impairment: A meta-analysis of resting-state fMRI studies. Ageing Res Rev 35:12–21
Tahmasian M, Eickhoff SB, Giehl K et al (2017) Resting-state functional reorganization in Parkinson’s disease: An activation likelihood estimation meta-analysis. Cortex 92:119–138
Logothetis NK, Pauls J, Augath M et al (2001) Neurophysiological investigation of the basis of the fMRI signal. Nature 412:150–157
Yu-Feng Z, Yong H, Chao-Zhe Z et al (2007) Altered baseline brain activity in children with ADHD revealed by resting-state functional MRI. Brain Develop 29:83–91
Bullmore E, Sporns O (2009) Complex brain networks: graph theoretical analysis of structural and functional systems. Nat Rev Neurosci 10:186–198
Martin SJ, Grimwood PD, Morris RGM (2000) Synaptic plasticity and memory: An evaluation of the hypothesis. Annu Rev Neurosci 23:649–711
Krecic AM, Swanson MS (1999) hnRNP complexes: composition, structure, and function. Curr Opin Cell Biol 11:363–371
Ruan QT, Yazdani N, Beierle JA et al (2018) Changes in neuronal immunofluorescence in the C- versus N-terminal domains of hnRNP H following D1 dopamine receptor activation. Neurosci Lett 684:109–114
Gao Y, Tatavarty V, Korza G et al (2008) Multiplexed dendritic targeting of α calcium calmodulin-dependent protein kinase II, neurogranin, and activity-regulated cytoskeleton-associated protein RNAs by the A2 pathway. MBoC 19:2311–2327
Cohen SM, Suutari B, He X, Wang Y, Sanchez S, Tirko NN et al (2018) Calmodulin shuttling mediates cytonuclear signaling to trigger experience-dependent transcription and memory. Nat Commun 9:2451
Nakamura TY, Nakao S, Nakajo Y, Takahashi JC, Wakabayashi S, Yanamoto H (2017) Possible signaling pathways mediating neuronal calcium sensor-1-dependent spatial learning and memory in mice. PLoS ONE 12:e0170829
Raymundi AM, da Silva TR, Zampronio AR et al (2020) A time-dependent contribution of hippocampal CB1, CB2 and PPARγ receptors to cannabidiol-induced disruption of fear memory consolidation. Br J Pharmacol 177(4):945–957
Estañ MC, Fernández-Núñez E, Zaki MS et al (2019) Recessive mutations in muscle-specific isoforms of FXR1 cause congenital multi-minicore myopathy. Nat Commun 10:797
Li L, Yu J, Ji SJ (2021) Axonal mRNA localization and translation: local events with broad roles. Cell Mol Life Sci 78:7379–7395
Boeynaems S, Alberti S, Fawzi NL et al (2018) Protein phase separation: a new phase in cell biology. Trends Cell Biol 28:420–435
Buchan JR, Parker R (2009) Eukaryotic stress granules: the ins and outs of translation. Mol Cell 36:932–941
Protter DSW, Parker R (2016) Principles and properties of stress granules. Trends Cell Biol 26:668–679. https://doi.org/10.1016/j.tcb.2016.05.004
Guo L, Kim HJ, Wang H et al (2018) Nuclear-import receptors reverse aberrant phase transitions of RNA-binding proteins with prion-like domains. Cell 173:677-692.e20
Pak CW, Kosno M, Holehouse AS et al (2016) Sequence determinants of intracellular phase separation by complex coacervation of a disordered protein. Mol Cell 63:72–85
Geuens T, Bouhy D, Timmerman V (2016) The hnRNP family: insights into their role in health and disease. Hum Genet 135:851–867
Kim HJ, Kim NC, Wang Y-D et al (2013) Mutations in prion-like domains in hnRNPA2B1 and hnRNPA1 cause multisystem proteinopathy and ALS. Nature 495:467–473
Xia T, Yang C, Wang X et al (2022) Heterogeneous nuclear ribonucleoprotein A2/B1 as a novel biomarker in elderly patients for the prediction of postoperative neurocognitive dysfunction: A prospective nested case-control study. Front Aging Neurosci 14:1034041
Ramaswami M, Taylor JP, Parker R (2013) Altered ribostasis: RNA-Protein granules in degenerative disorders. Cell 154:727–736
Baradaran-Heravi Y, Van Broeckhoven C, van der Zee J (2020) Stress granule mediated protein aggregation and underlying gene defects in the FTD-ALS spectrum. Neurobiol Dis 134:104639
Lu J, Cao Q, Hughes MP et al (2020) CryoEM structure of the low-complexity domain of hnRNPA2 and its conversion to pathogenic amyloid. Nat Commun 11:4090
Acknowledgements
We would like to express my gratitude to all these who helped us during the writing of this manuscript. Thanks to all the peer reviewers for their opinions and suggestions.
Funding
This work was supported by grants from the National Natural Science Foundation of China (82071220, 82371205), Natural Science Foundation of Tianjin (20JCYBJC01290), and the Science and Technology Foundation of Tianjin Health Commission (MS20013), and Tianjin key Medical Discipline (Specialty) Construction Project (TJYXZDXK-072C), Tianjin Multiple Investment Foundation of Applied Basic Research (21JCQNJC01020), Tianjin Health Research Project (TJWJ2023XK019, TJWJ2023QN042).
Author information
Authors and Affiliations
Contributions
Guarantor of integrity of the entire study: Miao Zhang, Haiyun Wang; study concepts: Miao Zhang, Haiyun Wang; study design: Miao Zhang; definition of intellectual content: Qiang Wang, Miao Zhang, Qingkai Tang; literature research: Feiyu Jia, Miao Zhang, Xinyi Wang; experimental studies: Feiyu Jia, Miao Zhang; data acquisition: Feiyu Jia, Miao Zhang, Tianyue Liu; data analysis: Miao Zhang, Qiang Wang; statistical analysis: Miao Zhang, Feiyu Jia, Chenyi Yang; manuscript preparation: Miao Zhang, Feiyu Jia, Qiang Wang; manuscript editing: Zhuo Yang; manuscript review: Haiyun Wang; All the authors approved for the final version.
Corresponding authors
Ethics declarations
Ethics Approval
Tianjin Medical University's Institutional Animal Conservation and Utilization Committee reviewed the animal study and approved it.
Consent to Participate
Not applicable
Consent for Publication
Not Applicable
Conflict of Interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, M., Jia, F., Wang, Q. et al. Kapβ2 Inhibits Perioperative Neurocognitive Disorders in Rats with Mild Cognitive Impairment by Reversing the Nuclear-Cytoplasmic Mislocalization of hnRNPA2/B1. Mol Neurobiol (2023). https://doi.org/10.1007/s12035-023-03789-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12035-023-03789-8