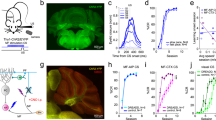
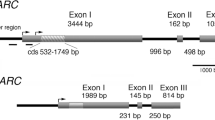
Abstract
An in vitro model of delay eyeblink classical conditioning was developed to investigate synaptic plasticity mechanisms underlying acquisition of associative learning. This was achieved by replacing real stimuli, such as an airpuff and tone, with patterned stimulation of the cranial nerves using an isolated brainstem preparation from turtle. Here, our primary findings regarding cellular and molecular mechanisms for learning acquisition using this unique approach are reviewed. The neural correlate of the in vitro eyeblink response is a replica of the actual behavior, and features of conditioned responses (CRs) resemble those observed in behavioral studies. Importantly, it was shown that acquisition of CRs did not require the intact cerebellum, but the appropriate timing did. Studies of synaptic mechanisms indicate that conditioning involves two stages of AMPA receptor (AMPAR) trafficking. Initially, GluA1-containing AMPARs are targeted to synapses followed later by replacement by GluA4 subunits that support CR expression. This two-stage process is regulated by specific signal transduction cascades involving PKA and PKC and is guided by distinct protein chaperones. The expression of the brain-derived neurotrophic factor (BDNF) protein is central to AMPAR trafficking and conditioning. BDNF gene expression is regulated by coordinated epigenetic mechanisms involving DNA methylation/demethylation and chromatin modifications that control access of promoters to transcription factors. Finally, a hypothesis is proposed that learning genes like BDNF are poised by dual chromatin features that allow rapid activation or repression in response to environmental stimuli. These in vitro studies have advanced our understanding of the cellular and molecular mechanisms that underlie associative learning.



Similar content being viewed by others
Data Availability
NA.
Code Availability
NA.
References
Gormezano I, Schneiderman N, Deaux E, Fuentes I (1962) Nictitating membrane: classical conditioning and extinction in the albino rabbit. Science 138:33–34
Gormezano I, Kehoe EJ, Marshall BS (1983) Twenty years of classical conditioning with the rabbit. Prog Psychobiol Physiol Psych 10:197–275
Thompson RF (1983) Neuronal substrates of simple associative learning: classical conditioning. Trends Neurosci 6:270–275
Delgado-Garcia JM, Gruart A (2006) Building new motor responses: eyelid conditioning revisited. Trends Neurosci 29:330–338
Bracha V, Zbarska S, Parker K, Carrel A, Zenitsky G, Bloedel JR (2009) The cerebellum and eye-blink conditioning: learning versus network performance hypotheses. Neurosci 162:787–796
Thompson F, Steinmetz JE (2009) The role of the cerebellum in classical conditioning of discrete behavioral responses. Neurosci 162:732–755
Mauk MD, Khilkevich A, Halverson H (2014) Cerebellar mechanisms of learning and plasticity revealed by delay eyelid conditioning. Int Rev Neurobiol 117:21–37
Clark GA, McCormick DA, Lavond DG, Thompson RF (1984) Effects of lesions of cerebellar nuclei on conditioned behavioral and hippocampal neuronal response. Brain Res 291:125–136
Yeo CH, Hardiman MJ, Glickstein M (1985a) Classical conditioning of the nictitating membrane response of the rabbit. I. Lesions of the cerebellar nuclei. Exp Brain Res 60:87–98
Yeo CH, Hardiman MJ, Glickstein M (1985b) Classical conditioning of the nictitating membrane response of the rabbit. II. Lesions of the cerebellar cortex. Exp Brain Res 60:99–113
Steinmetz JE, Lavond DG, Ivkovich D, Logan CG, Thompson RF (1992) Disruption of classical eyelid conditioning after cerebellar lesions: damage to a memory trace system or a simple performance deficit? J Neurosci 12:4403–4426
McCormick DA, Thompson RF (1984) Neuronal responses of the rabbit cerebellum during acquisition and performance of a classically conditioned nictitating membrane-eyelid response. J Neurosci 4:2811–2822
Berthier NE, Moore JW (1986) Cerebellar Purkinje cell activity related to the classically conditioned nictitating membrane response. Exp Brain Res 63:341–350
Berthier NE, Moore JW (1990) Activity of deep cerebellar nuclear cells during classical conditioning of nictitating membrane extension in rabbits. Exp Brain Res 83:44–54
Hesslow G, Ivarsson M (1994) Suppression of cerebellar Purkinje cells during conditioned responses in ferrets. Neuroreport 5:649–652
Norman RJ, Buchwald JS, Villablanca JR (1977) Classical conditioning with auditory discrimination of the eye blink in decerebrate cats. Science 196:551–553
Welsh JP, Harvey JA (1989) Cerebellar lesions and the nictitating membrane reflex: performance deficits of the conditioned and unconditioned response. J Neurosci 9:299–311
Kelly TM, Zuo C, Bloedel JR (1990) Classical conditioning of the eye-blink reflex in the decerebrate-decerebellate rabbit. Behav Brain Res 38:7–18
Gruart A, Blazquez P, Delgado-Garcia JM (1994) Kinematic analyses of classically-conditioned eyelid movements in the cat suggest a brain stem site for motor learning. Neurosci Lett 175:81–84
Anderson CW, Keifer J (1997) The cerebellum and red nucleus are not required for in vitro classical conditioning of the turtle abducens nerve response. J Neurosci 17:9736–9745
Nordholm AF, Lavond DG, Thompson RF (1991) Are eyeblink responses to tone in the decerebrate, decerebellate rabbit conditioned responses? Behav Brain Res 44:27–34
Chen L, Bao S, Lockard JM, Kim JJ, Thompson RF (1996) Impaired classical eyeblink conditioning in cerebellar-lesioned and Purkinje cell degeneration (pcd) mutant mice. J Neurosci 16:2829–2838
Ito M, Kano M (1982) Long-lasting depression of parallel fiber-Purkinje cell transmission induced by conjunctive stimulation of parallel fibers and climbing fibers in the cerebellar cortex. Neurosci Lett 33:253–258
Jirenhed DA, Bengtsson F, Hesslow G (2007) Acquisition, extinction, and reacquisition of a cerebellar cortical memory trace. J Neurosci 27:2493–2502
Freeman JH (2015) Cerebellar learning mechanisms. Brain Res 1621:260–269
Halversson HE, Khilkevich A, Mauk MD (2015) Relating cerebellar Purkinje cell activity to the timing and amplitude of conditioned eyelid responses. J Neurosci 35:7813–7832
Jirenhead DA, Hesslow G (2016) Are Purkinje cell pauses drivers of classically conditioned blink responses? Cerebellum 15:526–534
Freeman JH, Nicholson DA (1999) Neuronal activity in the cerebellar interpositus and lateral pontine nuclei during inhibitory classical conditioning of the eyeblink response. Brain Res 833:225–233
Aizenman CD, Linden DJ (2000) Rapid, synaptically driven increases in the intrinsic excitability of cerebellar deep nuclear neurons. Nat Neurosci 3:109–111
Ohyama T, Nores WL, Medina JF, Riusech FA, Mauk MD (2006) Learning-induced plasticity in deep cerebellar nucleus. J Neurosci 26:12656–12663
Pugh JR, Raman IM (2006) Potentiation of mossy fiber EPSCs in the cerebellar nuclei by NMDA receptor activation followed by post inhibitory rebound current. Neuron 51:113–123
Pugh JR, Raman IM (2008) Mechanisms of potentiation of mossy fiber EPSCs in the cerebellar nuclei by coincident synaptic excitation and inhibition. J Neurosci 28:10549–10560
Person AL, Raman IM (2010) Deactivation of L-type Ca current by inhibition controls LTP at excitatory synapses in the cerebellar nuclei. Neuron 66:550–559
Schreurs BG, Alkon DL (1993) Rabbit cerebellar slice analysis of long-term depression and its role in classical conditioning. Brain Res 631:235–240
Welsh JP, Yamaguchi H, Zeng XH, Kojo M, Nakada Y, Takagi A, Sugimori M, Llinas RR (2005) Normal motor learning during pharmacological prevention of Purkinje cell long-term depression. Proc Natl Acad Sci USA 102:17166–17171
Schonewille M, Gao Z, Boele H-J, Veloz MFV, Amerika WE, Simek AAM, De Jeu MT, Steinberg JP, Takamiya K, Hoebeek FE, Linden DJ, Huganir RL, De Zeeuw CI (2011) Reevaluating the role of LTD in cerebellar motor learning. Neuron 70:43–50
Hesslow G, Jirenhed D-A, Rasmussen A, Johansson F (2013) Classical conditioning of motor responses: what is the learning mechanism? Neural Netw 47:81–87
Johansson F, Jirenhed DA, Rasmussen A, Zucca R, Hesslow G (2018) Absence of parallel fibre to Purkinje cell LTD during eyeblink conditioning. Sci Reports 8:14777
Johansson F (2019) Intrinsic memory of temporal intervals in cerebellar Purkinje cells. Neurobiol Learn & Mem 166:107103
Keifer J, Houk JC (1991) Role of excitatory amino acids in mediating burst discharge of red nucleus neurons in the in vitro turtle brainstem-cerebellum. J Neurophysiol 65:454–467
Keifer J, Vyas D, Houk JC (1992) Sulforhodamine labeling of neural circuits engaged in motor pattern generation in the in vitro turtle brainstem-cerebellum. J Neurosci 12:3187–3199
Keifer J (1996) Effects of red nucleus inactivation on burst discharge in turtle cerebellum in vitro: evidence for positive feedback. J Neurophysiol 76:2200–2210
Keifer J (1993) In vitro eye-blink reflex model: role of excitatory amino acid receptors and labeling of network activity with sulforhodamine. Exp Brain Res 97:239–253
Keifer J, Armstrong KE, Houk JC (1995) In vitro classical conditioning of abducens nerve discharge in turtles. J Neurosci 15:5036–5048
Zheng Z, Sabirzhanov B, Keifer J (2012) Two-stage AMPA receptor trafficking in classical conditioning and selective role for glutamate receptor subunit 4 (tGluA4) flop splice variant. J Neurophysiol 108:101–111
Walls GL (1942) The vertebrate eye and its adaptive radiation. Canbrook Institute of Science, New York
Barbas-Henry HA, Lohman AHM (1988) The motor nuclei and sensory neurons of the IIIrd, IVth, and VIth cranial nerves in the monitor lizard, Varanus exanthematicus. J Comp Neurol 267:370–386
Herrick JL, Keifer J (1998) Central trigeminal and posterior eighth nerve projections in the turtle Chrysemys picta studied in vitro. Brain Behav Evol 51:183–201
Zhu D, Keifer J (2004) Pathways controlling trigeminal and auditory nerve-evoked abducens eyeblink reflexes in pond turtles. Brain Behav Evol 64:207–222
Powers AS, Evinger CE (1993) Characterization of blink responses in turtles. Second Int Behav Neurosci Soc Conf, Clearwater, FL
Barbas-Henry HA, Wouterlood FG (1988) Synaptic connections between primary trigeminal afferents and accessory abducens motoneurons in the monitor lizard, Varanus exanthematicus. J Comp Neurol 267:387–397
Weiss C, Disterhoft JF (1985) Connections of the rabbit abducens nucleus. Brain Res 326:172–178
Van Ham JJ, Yeo CH (1996) Trigeminal inputs to eyeblink motoneurons in the rabbit. Exp Neurol 142:244–257
Berthier NE, Moore JW (1983) The nictitating membrane response: an electrophysiological study of the abducens nerve and nucleus and the accessory abducens nucleus in rabbit. Brain Res 258:201–210
Disterhoft JF, Weiss C (1985) Motoneuronal control of eye retraction/nictitating membrane extension in rabbit. In: Alkon DL, Woody CD (eds) Neural mechanisms of conditioning. Plenum, New York, pp. 197–208
Evinger C, Sibony PA, Manning KA, Fiero RA (1988) A pharmacological distinction between the long and short latency pathways of the human blink reflex revealed with tobacco. Exp Brain Res 73:477–480
Pellegrini JJ, Horn AKE, Evinger C (1995) The trigeminally evoked blink reflex I. Neuronal circuits. Exp Brain Res 107:166–180
Delgado-Garcia JM, Evinger C, Escudero M, Baker R (1990) Behavior of accessory abducens motoneurons during eye retraction and rotationin the alert cat. J Neurophysiol 64:413–422
Medina JF, Garcia KS, Mauk MD (2001) A mechanism for savings in the cerebellum. J Neurosci 21:4081–4089
Perrett SP, Ruiz BP, Mauk MD (1993) Cerebellar cortex lesions disrupt learning-dependent timing of conditioned eyelid responses. J Neurosci 13:1708–1718
Hebb DO (1949) The organization of behavior: a neuropsychological theory. Wiley, New York
Tsien JZ (2000) Linking Hebb’s coincidence-detection to memory formation. Curr Opin Neurobiol 10:266–273
Jacobs S, Cui Z, Feng R, Wang H, Wang D, Tsien JZ (2014) Molecular and genetic determinants of the NMDA receptor for superior learning and memory functions. PLoS One 9:e111865
Keifer J, Mokin M (2004) Distribution of anterogradely labeled trigeminal and auditory nerve boutons on abducens motor neurons in turtles: Implications for in vitro classical conditioning. J Comp Neurol 471:144–152
Keifer J, Zheng Z (2015) Coincidence detection in a neural correlate of classical conditioning is initiated by bidirectional 3-phosphoinositide-dependent kinase-1 signalling and modulated by adenosine receptors. J Physiol 593:1581–1595
Moore JW, Desmond JE, Berthier NE (1989) Adaptively timed conditioned responses and the cerebellum: a neural network. Approach Biol Cybern 62:17–28
Anderson CW, Keifer J (1999) Properties of conditioned abducens nerve responses in a highly reduced in vitro brain stem preparation from the turtle. J Neurophysiol 81:1242–1250
Garcia KS, Mauk MD (1998) Pharmacological analysis of cerebellar contributions to the timing and expression of conditioned eyelid responses. Neuropharmacology 37:471–480
Kalmbach BE, Davis T, Ohyama T, Riusech F, Nores WL, Mauk MD (2010) Cerebellar cortex contributions to the expression and timing of conditioned eyelid responses. J Neurophysiol 103:2039–2049
Raymond JL, Lisberger SG, Mauk MD (1996) The cerebellum: a neuronal learning machine? Science 272:1126–1131
Koekkoek SKE, Hulscher HC, Dortland BR, Hensbroek RA, Elgersma Y, Ruigrok TJH, De Zeeuw CI (2003) Cerebellar LTD and learning-dependent timing of conditioned eyelid responses. Science 301:1736–1739
Gerwig M, Hajjar K, Dimitrova A, Maschke M, Kolb FP, Frings M, Thilmann AF, Forsting M, Diener HC, Timmann D (2005) Timing of conditioned eyeblink responses is impaired in cerebellar patients. J Neurosci 25:3919–3931
Svensson P, Jirenhed DA, Bengtsson F, Hesslow G (2010) Effect of conditioned stimulus parameters on timing of conditioned Purkinje cell responses. J Neurophysiol 103:1329–1336
Maas RPPWM, Schutter DJLG, Toni I, Timmann D, van de Warrenburg BPC (2022) Cerebellar transcranial direct current stimulation modulates timing but not acquisition of conditioned eyeblink responses in SCA3 patients. Brain Stimulation 15:806–813
Choquet D (2018) Linking nanoscale dynamics of AMPA receptor organization to plasticity of excitatory synapses and learning. J Neurosci 38:9318–9329
Diering GH, Huganir RL (2018) The AMPA receptor code of synaptic plasticity. Neuron 100:314–329
Keifer J (2022) Regulation of AMPAR trafficking in synaptic plasticity by BDNF and the impact of neurodegenerative disease. J Neurosci Res 100:979–991
Keifer J (2001) In vitro eye-blink classical conditioning is NMDA receptor-dependent and involves redistribution of AMPA receptor subunit GluR4. J Neurosci 21:2434–2441
Keifer J, Clark TG (2003) Abducens conditioning in in vitro turtle brain stem without cerebellum requires NMDA receptors and involves upregulation of GluR4 containing AMPA receptors. Exp Brain Res 151:405–410
Bliss TVP, Collingridge GL (1993) A synaptic model of memory: long-term potentiation in the hippocampus. Nature 361:31–39
Nicoll RA, Roche KW (2013) Long-term potentiation: peeling the onion. Neuropharmacol 74:18–22
Hayashi Y (2022) Molecular mechanism of hippocampal long-term potentiation – towards multiscale understanding of learning and memory. Neurosci Res 175:3–15
Servatius RJ, Shors TJ (1996) Early acquisition, but not retention, of the classically conditioned eyeblink response is N-methyl-D-aspartate (NMDA) receptor dependent. Behav Neurosci 110:1040–1048
Kishimoto Y, Kawahara S, Kirino Y, Kadotani H, Nakamura Y, Ikeda M, Yoshioka T (1997) Conditioned eyeblink response is impaired in mutant mice lacking NMDA receptor subunit NR2A. Neuroreport 8:3717–3721
Tocco G, Devgan KK, Hauge SA, Weiss C, Baudry M, Thompson RF (1991) Classical conditioning selectively increases AMPA receptor binding in rabbit hippocampus. Brain Res 559:331–336
Hauge SA, Tracy JA, Baudry M, Thompson RF (1998) Selective changes in AMPA receptors in rabbit cerebellum following classical conditioning of the eyelid-nictitating membrane response. Brain Res 803:9–18
Mokin M, Keifer J (2004) Targeting of GluR4-containing AMPA receptors to synaptic sites during in vitro classical conditioning. Neurosci 128:219–228
Mokin M, Keifer J (2006) Quantitative analysis of immune fluorescent punctate staining of synaptically localized proteins using confocal microscopy and stereology. J Neurosci Methods 157:218–224
Mokin M, Lindahl JS, Keifer J (2006) Immediate-early gene-encoded protein arc is associated with synaptic delivery of GluR4-containing AMPA receptors during in vitro classical conditioning. J Neurophysiol 95:215–224
Rao A, Craig AM (1997) Activity regulates the synaptic localization of the NMDA receptor in hippocampal neurons. Neuron 19:801–812
Liao D, Zhang X, O’Brian R, Ehlers MD, Huganir RL (1999) Regulation of morphological postsynaptic silent synapses in developing hippocampal neurons. Nat Neurosci 2:37–43
Liao D, Scannevin RH, Huganir R (2001) Activation of silent synapses by rapid activity-dependent synaptic recruitment of AMPA receptors. J Neurosci 21:6008–6017
Lissin DV, Carroll RC, Nicoll RA, Malenka RC, Von Zastrow M (1999) Rapid, activation-induced redistribution of ionotropic glutamate receptors in cultured hippocampal neurons. J Neurosci 19:1263–1272
Zheng Z, Keifer J (2009) PKA has a critical role in synaptic delivery of GluR1- and GluR4-containing AMPARs during initial stages of acquisition of in vitro classical conditioning. J Neurophysiol 101:2539–2549
Zheng Z, Keifer J (2014) Sequential delivery of synaptic GluA1 and GluA4-containing AMPARs by SAP97 anchored protein complexes in classical conditioning. J Biol Chem 289:10540–10550
Keifer J, Tiwari NK, Buse L, Zheng Z (2017) Subunit-specific synaptic delivery of AMPA receptors by auxiliary chaperone proteins TARPγ8 and GSG1L in classical conditioning. Neurosci Lett 645:53–59
Isaac JT, Nicoll RA, Malenka RC (1995) Evidence for silent synapses: implications for the expression of LTP. Neuron 15:427–434
Liao D, Hessler NA, Malinow R (1995) Activation of postsynaptically silent synapses during pairing-induced LTP in CA1 region of hippocampal slice. Nature 375:400–404
Shi SH, Hayashi Y, Petralia RS, Zaman SH, Wenthold RJ, Svoboda K, Malinow R (1999) Rapid spine delivery and redistribution of AMPA receptors after synaptic NMDA receptor activation. Science 284:1811–1816
Shi SH, Hayashi Y, Esteban JA, Malinow R (2001) Subunit-specific rules governing AMPA receptor trafficking to synapses in hippocampal pyramidal neurons. Cell 105:331–343
Diaz-Alonso J, Sun YJ, Granger AJ, Levy JM, Blankenship SM, Nicoll RA (2017) Subunit-specific role for the amino-terminal domain of AMPA receptors in synaptic targeting. Proc Nat Acad Sci USA 114:7136–7141
Zhou Z, Liu A, Xia S, Leung C, Qi J, Meng Y, Xie W, Park P, Collingridge GL, Jia Z (2018) The C-terminal tails of endogenous GluA1 and GluA2 differentially contribute to hippocampal synaptic plasticity and learning. Nat Neurosci 21:50–62
Adesnik H, Nicoll RA (2007) Conservation of glutamate receptor 2-containing AMPA receptors during long-term potentiation. J Neurosci 27:4598–4602
Rumpel S, LeDoux J, Zador A, Malinow R (2005) Postsynaptic receptor trafficking underlying a form of associative learning. Science 308:83–88
Clem R, Huganir RL (2010) Calcium-permeable AMPA receptor dynamics mediate fear memory erasure. Science 330:1108–1112
Penn AC, Zhang CL, Georges F, Rover L, Breillat C, Hosy E, Petersen JD, Humeau Y, Choquet D (2017) Hippocampal LTP and contextual learning require surface diffusion of AMPA receptors. Nature 549:384–388
Mokin M, Zheng Z, Keifer J (2007) Conversion of silent synapses into the active pool by selective GluR1-3 and GluR4 AMPAR trafficking during in vitro classical conditioning. J Neurophysiol 98:1278–1286
Keifer J, Zheng Z (2010) AMPA receptor trafficking and learning. Eur J Neurosci 32:269–277
Keifer J, Houk JC (2011) Modeling signal transduction in classical conditioning with network motifs. Front Mol Neurosci 4:9
Carlezon WA Jr, Duman RS, Nestler EJ (2005) The many faces of CREB. Trends Neurosci 28:436–445
Li W, Keifer J (2008) Coordinate action of pre- and postsynaptic brain-derived neurotrophic factor is required for AMPAR trafficking and acquisition of in vitro classical conditioning. Neuroscience 155:686–697
Li W, Keifer J (2009) BDNF-induced synaptic delivery of AMPAR subunits is differentially dependent on NMDA receptors and requires ERK. Neurobiol Learn & Mem 91:243–249
Keifer J, Sabirzhanov BE, Zheng Z, Li W, Clark TG (2009) Cleavage of proBDNF to BDNF by a tolloid-like metalloproteinase is required for acquisition of in vitro eyeblink classical conditioning. J Neurosci 29:14956–14964
Ambigapathy G, Zheng Z, Keifer J (2015) Regulation of BDNF chromatin status and promoter accessibility in a neural correlate of associative learning. Epigenetics 10:981–993
Ambigapathy G, Zheng Z, Keifer J (2013) Identification of a functionally distinct truncated BDNF mRNA splice variant and protein in Trachemys scripta elegans. PLoS One 8:e67141
Keifer J, Zheng Z, Zhu D (2007) MAPK signaling pathways mediate AMPA receptor trafficking in an in vitro model of classical conditioning. J Neurophysiol 97:2067–2074
Sharma SK, Sheriff CM, Stough S, Hsuan V, Carew TJ (2006) A tropomyosin-related kinase B ligand is required for ERK activation, long-term synaptic facilitation, and long-term memory in Aplysia. Proc Natl Acad Sci USA 103:14206–14210
Mosbacher J, Schoepfer R, Monyer H, Burnashev N, Seeburg PH, Rupersberg JP (1994) A molecular determinant for submillisecond desensitization in glutamate receptors. Science 266:1059–1062
Pilati N, Linley DM, Selvaskanden H, Uchitel O, Henning MH, Kopp-Scheinpflug C, Forsythe ID (2016) Acoustic trauma slows AMPA receptor-mediated EPSCs in the auditory brainstem, reducing GluA4 subunit expression as a mechanism to rescue binaural function. J Physiol 594:3683–3703
Zheng Z, Keifer J (2008) Protein kinase C-dependent and independent signaling pathways regulate synaptic GluR1 and GluR4 AMPAR subunits during in vitro classical conditioning. Neuroscience 156:872–884
Lisman J, Raghavachari S (2006) A unified model of the presynaptic and postsynaptic changes during LTP at CA1 synapses. Sci STKE 356:re11
Opazo P, Sainlos M, Choquet D (2012) Regulation of AMPA receptor surface diffusion by PSD-95 slots. Curr Opin Neurobiol 22:453–460
Chater TE, Goda Y (2022) The shaping of AMPA receptor surface distribution by neuronal activity. Front Syn Neurosci 14:833782
Humeau Y, Reisel D, Johnson AW, Borchardt T, Jensen V, Gebhardt BV, Gass P, Bannerman DM, Good MA, Hvalby O, Sprengel R, Luthi A (2007) A pathway-specific function for different AMPA receptor subunits in amygdala long-term potentiation and fear conditioning. J Neurosci 27:10947–10956
Nedelescu H, Kelso CM, Lazaro-Munoz G, Purpura M, Cain CK, LeDoux JE, Aoki C (2010) Endogenous GluR1-containing AMPA receptors translocate to asymmetric synapses in the lateral amygdala during early phase of fear memory formation: an electron microscopic immunocytochemical study. J Comp Neurol 518:4723–4739
Wong W, Scott JD (2004) AKAP signaling complexes: focal points in space and time. Nat Rev Mol Cell Biol 5:959–970
Nie T, McDonough CB, Huang T, Nguyen PV, Abel T (2007) Genetic disruption of protein kinase A anchoring reveals a role for compartmentalized kinase signaling in theta-burst long-term potentiation and spatial memory. J Neurosci 27:10278–10288
Purkey AM, Woolfrey KM, Crosby KC, Chick WS, Aoto J, Dell’Acqua ML (2018) AKAP150 palmitoylation regulates synaptic incorporation of Ca2-permeable AMPA receptors to control LTP. Cell Rep 25:974–987
Shalin SC, Hernandez CM, Dougherty MK, Morrison DK, Sweatt JD (2006) Kinase suppressor of Ras1 compartmentalizes hippocampal signal transduction and subserves synaptic plasticity and memory formation. Neuron 50:765–779
Jackson AC, Nicoll RA (2011) The expanding social network of ionotropic glutamate receptors: TARPs and other transmembrane auxiliary subunits. Neuron 70:178–199
Schwenk J, Harmel N, Brechet A, Zolles G, Berkefeld H, Swantje Muller C, Bildl W, Baehrens D, Huber B, Kulik A, Klocker N, Schulte U, Fakler B (2012) High-resolution proteomics unravel architecture and molecular diversity of native AMPA receptor complexes. Neuron 74:621–633
Keifer J, Zheng Z, Mokin M (2008) Synaptic localization of GluR4-containing AMPARs and Arc during acquisition, extinction and reacquisition of in vitro classical conditioning. Neurobiol Learn Mem 90:301–308
Zheng Z, Ambigapathy G, Keifer J (2019) Characterization and transcriptional activation of the immediate early gene ARC during a neural correlate of classical conditioning. J Mol Neurosci 69:380–390
Chowdhury S, Shepherd JD, Okuno H, Lyford G, Petralia RS, Plath N, Kuhl D, Huganir RL, Worley PF (2006) Arc/Arg3.1 interacts with the endocytic machinery to regulate AMPA receptor trafficking. Neuron 52:445–459
Rial Verde EM, Lee-Osbourne J, Worley PF, Malinow R, Cline HT (2006) Increased expression of the immediate-early gene Arc/Arg3.1 reduces AMPA receptor-mediated synaptic transmission. Neuron 52:461–474
Wall MJ, Collins DR, Chery SL, Allen ZD, Patuzyn ED, George AJ, Nikolova VD, Moy SS, Philpot BD, Shepherd JD, Muller J, Ehlers MD, Am M, Correa SAL (2018) The temporal dynamics of Arc expression regulate cognitive flexibility. Neuron 98:1124–1132
Ambigapathy G, Zheng Z, Keifer J (2014) Genomic organization and identification of promoter regions for the BDNF gene in the pond turtle Trachemys scripta elegans. J Mol Neurosci 53:626–636
Kaas GA, Zhong C, Eason DE, Ross DL, Vachhani RV, Ming G, King JR, Song H, Sweatt JD (2013) Tet1 controls CNS 5-methylcytosine hydroxylation, active DNA demethylation, gene transcription, and memory formation. Neuron 79:1086–1093
Rudenko A, Dawlaty MM, Seo J, Cheng AW, Meng J, Le T, Faull KF, Jaenisch R, Tsai L-H (2013) Tet1 is critical for neuronal activity-regulated gene expression and memory extinction. Neuron 79:1109–1122
Feng J, Shao N, Szulwach KE, Vialou V, Huynh J, Zhong C, Le T, Ferguson D et al (2015) Role of Tet1 and 5-hydroxymethylcytosine in cocaine action. Nat Neurosci 18:536–544
Wu H, Zhang Y (2011) Mechanisms and functions of Tet protein-mediated 5-methylcytosine oxidation. Genes Dev 25:2436–2452
Voigt P, Tee WW, Reinberg D (2013) A double take on bivalent promoters. Genes Dev 27:1318–1338
Harikumar A, Meshorer E (2015) Chromatin remodeling and bivalent histone modifications in embryonic stem cells. EMBO Rep 16:1609–1619
Lubin FD, Roth TL, Sweatt JD (2008) Epigenetic regulation of bdnf gene transcription in the consolidation of fear memory. J Neurosci 28:10576–10586
Gupta S, Kim SY, Artis S, Molfese DL, Schumacher A, Sweatt JD, Paylor RE, Lubin FD (2010) Histone methylation regulates memory formation. J Neurosci 30:3589–3599
Zheng Z, Ambigapathy G, Keifer J (2017) MeCP2 regulates Tet1-catalyzed demethylation, CTCF binding, and learning-dependent alternative splicing of the BDNF gene in turtle. eLife 6:e25384
Zheng Z, Keifer J (2021) Learning-dependent transcription regulation of BDNF by its truncated protein isoform in turtle. J Mol Neurosci 71:999–1014
Keifer J (2021) Comparative genomics of the BDNF gene, non-canonical modes of transcriptional regulation, and neurological disease. Mol Neurobiol 58:2851–2861
Flavell SW, Kim T-K, Gray JM, Harmin DA, Hemburg M, Hong EJ, Markenscoff-Papadimitriou E, Bear DM, Greenberg ME (2008) Genome-wide analysis of MEF2 transcriptional program reveals synaptic target genes and neuronal activity-dependent polyadenylation site selection. Neuron 60:1022–1038
Sala C, Futai K, Yamamoto K, Worley PF, Hayashi Y, Sheng M (2003) Inhibition of dendritic spine morphogenesis and synaptic transmission by activity-inducible protein Homer1a. J Neurosci 23:6327–6337
Niibori Y, Hayashi F, Hirai K, Matsui M, Inokuchi K (2007) Alternative poly(A) site-selection regulates the production of alternatively spliced vesl-1/homer1 isoforms that encode postsynaptic scaffolding proteins. Neurosci Res 57:399–410
Liu Q-R, Walther D, Drgon T, Polesskaya O, Lesnick TG, Strain KJ, de Andrade M, Bower JH, Maragonore DM, Uhl GR (2005) Human brain derived neurotrophic factor (BDNF) genes, splicing pattern, and assessments of associations with substance abuse and Parkinson’s disease. Am J Med Genet Pt B 134B:93–103
Pruunsild P, Kazantseva A, Aid T, Palm K, Timmusk T (2007) Dissecting the human BDNF locus: bidirectional transcription, complex splicing, and multiple promoters. Genomics 90:397–406
Lipovich L, Dachet F, Cai J, Bagla S, Balan K, Jia H, Loeb JA (2012) Activity-dependent human brain coding/noncoding gene regulatory networks. Genetics 192:1133–1148
Modarresi F, Faghihi MA, Lopez-Toledano A, Fatemi RP, Magistri M, Brothers SP, van der Brug MP, Wahlestedt C (2012) Inhibition of natural antisense transcripts in vivo results in gene-specific transcriptional upregulation. Nat Biotech 30:453–459
Modarresi F, Fatemi RP, Razavipour SF, Ricciardi N, Makhmutova M, Khoury N, Magistri M, Volmar C-H, Wahlestedt C, Faghihi MA (2021) A novel knockout mouse model of the noncoding antisense brain-derived neurotrophic factor (Bdnf) gene displays increased endogenous Bdnf protein and improved memory function following exercise. Heliyon 7:e07570
Tapia-Arancibia L, Aliaga E, Silhol M, Arancibia S (2008) New insights into brain BDNF function in normal aging and Alzheimer disease. Brain Res Rev 59:201–220
Nagahara AH, Tuszynski MH (2011) Potential therapeutic uses of BDNF in neurological and psychiatric disorders. Nat Rev Drug Disc 10:209–219
Bernstein BE, Mikkelsen TS, Xie X, Kamal M, Huebert DJ, Cuff J, Fry B, Meissner A et al (2006) A bivalent chromatin structure marks key developmental genes in embryonic stem cells. Cell 125:315–326
Keifer J (2017) Primetime for learning genes. Genes 8:69
Koo JW, Mazei-Robison MS, LaPlant Q, Egervari G, Braunscheidel KM, Adank DN, Ferguson D, Feng J et al (2015) Epigenetic basis of opiate suppression of Bdnf gene expression in the ventral tegmental area. Nat Neurosci 18:415–422
Palomer E, Martin-Segura A, Baliyan S, Ahmed T, Balschun D, Venero C, Martin MG, Dotti CG (2016) Aging triggers a repressive chromatin state at Bdnf promoters in hippocampal neurons. Cell Rep 16:2889–2900
Keifer J, Summers CH (2016) Putting the “biology” back into “neurobiology”: the strength of diversity in animal model systems for neuroscience research. Front Syst Neurosci 10:69
Funding
This work was funded for many years by the National Institutes of Health (NINDS, NIMH) and the National Science Foundation to J.K.
Author information
Authors and Affiliations
Contributions
JK conceptualized and wrote the paper.
Corresponding author
Ethics declarations
Ethics Approval
NA.
Consent to Participate
NA.
Consent for Publication
NA.
Conflict of Interest
The author declares no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Keifer, J. Synaptic Mechanisms of Delay Eyeblink Classical Conditioning: AMPAR Trafficking and Gene Regulation in an In Vitro Model. Mol Neurobiol 60, 7088–7103 (2023). https://doi.org/10.1007/s12035-023-03528-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-023-03528-z