Abstract
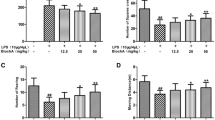
Gallic acid (3,4,5-trihydroxybenzoic acid, GA), a phenolic acid, is ubiquitous in almost all parts of the plant. In the present study, a neuroinflammatory rat model using intranigral infusion of lipopolysaccharides (LPS, 4 μg/μL) was employed to study the neuroprotective effect of GA which was orally administered daily. Compared with the vehicle-treated rats, systemic administration of GA (100 mg/kg) significantly attenuated LPS-induced increases in glial fibrillary acidic protein (a biomarker of activated astrocytes) and ED-1 (a biomarker of activated microglia), as well as inducible nitric oxide synthase (iNOS, a proinflammatory enzyme) and interleukin-1β (a proinflammatory cytokine), in the LPS-infused substantia nigra (SN) of rat brain. At the same time, GA attenuated LPS-induced elevation in heme oxygenase-1 level (a redox-regulated protein) and α-synuclein aggregation (a hallmark of CNS neurodegeneration), suggesting that GA is capable of inhibiting LPS-induced oxidative stress and protein conjugation. Furthermore, GA prevented LPS-induced caspase 3 activation (a biomarker of programmed cell death) and LPS-induced increases in receptor-interacting protein kinase (RIPK)-1 and RIPK-3 levels (biomarkers of necroptosis), indicating that GA inhibited LPS-induced apoptosis and necroptosis in the nigrostriatal dopaminergic system of rat brain. Moreover, an in vitro study was employed to investigate the anti-inflammatory effect of GA on BV2 microglial cells which were subjected to LPS (1 μg/mL) treatment. Consistently, co-incubation of GA diminished LPS-induced increases in iNOS mRNA and iNOS protein expression in the treated BV-2 cells as well as NO production in the culture medium. The anti-oxidative activity of GA was evaluated using iron-induced lipid peroxidation of brain homogenates. After 3-h incubation at 37 °C, GA was more potent than glutathione and less potent than trolox in inhibiting iron-induced lipid peroxidation. Conclusively, the present study suggests that GA is anti-inflammatory via attenuating LPS-induced neuroinflammation, oxidative stress, and protein conjugation. Furthermore, GA prevented LPS-induced programmed cell deaths of nigrostriatal dopaminergic neurons of the rat brain, suggesting that GA may be neuroprotective by attenuating neuroinflammation in CNS neurodegenerative diseases.





Similar content being viewed by others
References
Manoharan S, Guillemin GJ (2016) The role of reactive oxygen species in the pathogenesis of Alzheimer’s disease, Parkinson’s disease, and Huntington’s disease: a mini review. 2016:8590578. https://doi.org/10.1155/2016/8590578
Flores-Martinez YM, Fernandez-Parrilla MA (2018) Acute neuroinflammatory response in the substantia nigra pars compacta of rats after a local injection of lipopolysaccharide. 2018:1838921. https://doi.org/10.1155/2018/1838921
Reinert KR, Umphlet CD, Quattlebaum A, Boger HA (2014) Short-term effects of an endotoxin on substantia nigra dopamine neurons. Brain Res 1557:164–170. https://doi.org/10.1016/j.brainres.2014.02.005
Rutherford NJ, Sacino AN, Brooks M, Ceballos-Diaz C, Ladd TB, Howard JK, Golde TE, Giasson BI (2015) Studies of lipopolysaccharide effects on the induction of alpha-synuclein pathology by exogenous fibrils in transgenic mice. Mol Neurodegener 10:32. https://doi.org/10.1186/s13024-015-0029-4
Yang QQ, Zhou JW (2018) Neuroinflammation in the central nervous system: symphony of glial cells. https://doi.org/10.1002/glia.23571
Lin AM, Fang SF, Chao PL, Yang CH (2007) Melatonin attenuates arsenite-induced apoptosis in rat brain: involvement of mitochondrial and endoplasmic reticulum pathways and aggregation of alpha-synuclein. J Pineal Res 43(2):163–171. https://doi.org/10.1111/j.1600-079X.2007.00456.x
Wang YT, Lin HC, Zhao WZ, Huang HJ, Lo YL, Wang HT, Lin AM (2017) Acrolein acts as a neurotoxin in the nigrostriatal dopaminergic system of rat: involvement of alpha-synuclein aggregation and programmed cell death. Sci Rep 7:45741. https://doi.org/10.1038/srep45741
Hung KC, Huang HJ, Wang YT, Lin AM (2016) Baicalein attenuates alpha-synuclein aggregation, inflammasome activation and autophagy in the MPP(+)-treated nigrostriatal dopaminergic system in vivo. J Ethnopharmacol 194:522–529. https://doi.org/10.1016/j.jep.2016.10.040
Fu Y, Yang J, Wang X, Yang P, Zhao Y, Li K, Chen Y (2018) Herbal compounds play a role in neuroprotection through the inhibition of microglial activation. 2018:9348046. https://doi.org/10.1155/2018/9348046
Jiang X, Ganesan P, Rengarajan T, Choi DK, Arulselvan P (2018) Cellular phenotypes as inflammatory mediators in Parkinson’s disease: interventional targets and role of natural products. Biomed Pharmacother 106:1052–1062. https://doi.org/10.1016/j.biopha.2018.06.162
Wasik A, Antkiewicz-Michaluk L (2017) The mechanism of neuroprotective action of natural compounds. Pharmacol Rep 69(5):851–860. https://doi.org/10.1016/j.pharep.2017.03.018
Daglia M, Di Lorenzo A, Nabavi SF, Talas ZS, Nabavi SM (2014) Polyphenols: well beyond the antioxidant capacity: gallic acid and related compounds as neuroprotective agents: you are what you eat! Curr Pharm Biotechnol 15(4):362–372
Rai SN, Birla H, Singh SS, Zahra W, Patil RR, Jadhav JP, Gedda MR, Singh SP (2017) Mucuna pruriens protects against MPTP intoxicated neuroinflammation in Parkinson’s disease through NF-kappaB/pAKT signaling pathways. Front Aging Neurosci 9:421. https://doi.org/10.3389/fnagi.2017.00421
Chandrasekhar Y, Phani Kumar G, Ramya EM, Anilakumar KR (2018) Gallic acid protects 6-OHDA induced neurotoxicity by attenuating oxidative stress in human dopaminergic cell line. Neurochem Res 43(6):1150–1160. https://doi.org/10.1007/s11064-018-2530-y
Mansouri MT, Farbood Y, Sameri MJ, Sarkaki A, Naghizadeh B, Rafeirad M (2013) Neuroprotective effects of oral gallic acid against oxidative stress induced by 6-hydroxydopamine in rats. Food Chem 138(2–3):1028–1033. https://doi.org/10.1016/j.foodchem.2012.11.022
Lively S, Schlichter LC (2018) Microglia responses to pro-inflammatory stimuli (LPS, IFNgamma+TNFalpha) and reprogramming by resolving cytokines (IL-4, IL-10). Front Cell Neurosci 12:215. https://doi.org/10.3389/fncel.2018.00215
Cazareth J, Guyon A, Heurteaux C, Chabry J, Petit-Paitel A (2014) Molecular and cellular neuroinflammatory status of mouse brain after systemic lipopolysaccharide challenge: importance of CCR2/CCL2 signaling. J Neuroinflammation 11:132. https://doi.org/10.1186/1742-2094-11-132
Hughes CD, Choi ML, Ryten M, Hopkins L, Drews A, Botia JA, Iljina M, Rodrigues M et al (2018) Picomolar concentrations of oligomeric alpha-synuclein sensitizes TLR4 to play an initiating role in Parkinson’s disease pathogenesis. doi:https://doi.org/10.1007/s00401-018-1907-y
Codolo G, Plotegher N, Pozzobon T, Brucale M, Tessari I, Bubacco L, de Bernard M (2013) Triggering of inflammasome by aggregated alpha-synuclein, an inflammatory response in synucleinopathies. PLoS One 8(1):e55375. https://doi.org/10.1371/journal.pone.0055375
Couch Y, Alvarez-Erviti L, Sibson NR, Wood MJ, Anthony DC (2011) The acute inflammatory response to intranigral alpha-synuclein differs significantly from intranigral lipopolysaccharide and is exacerbated by peripheral inflammation. J Neuroinflammation 8:166. https://doi.org/10.1186/1742-2094-8-166
Fricker M, Vilalta A, Tolkovsky AM, Brown GC (2013) Caspase inhibitors protect neurons by enabling selective necroptosis of inflamed microglia. J Biol Chem 288(13):9145–9152. https://doi.org/10.1074/jbc.M112.427880
Maya S, Prakash T, Goli D (2018) Evaluation of neuroprotective effects of wedelolactone and gallic acid on aluminium-induced neurodegeneration: relevance to sporadic amyotrophic lateral sclerosis. Eur J Pharmacol 835:41–51. https://doi.org/10.1016/j.ejphar.2018.07.058
Qin S, Yang C, Huang W, Du S, Mai H, Xiao J, Lu T (2018) Sulforaphane attenuates microglia-mediated neuronal necroptosis through down-regulation of MAPK/NF-kappaB signaling pathways in LPS-activated BV-2 microglia. Pharmacol Res 133:218–235. https://doi.org/10.1016/j.phrs.2018.01.014
Sun J, Li YZ, Ding YH, Wang J, Geng J, Yang H, Ren J, Tang JY et al (2014) Neuroprotective effects of gallic acid against hypoxia/reoxygenation-induced mitochondrial dysfunctions in vitro and cerebral ischemia/reperfusion injury in vivo. Brain Res 1589:126–139. https://doi.org/10.1016/j.brainres.2014.09.039
Sarkaki A, Farbood Y, Gharib-Naseri MK, Badavi M, Mansouri MT, Haghparast A, Mirshekar MA (2015) Gallic acid improved behavior, brain electrophysiology, and inflammation in a rat model of traumatic brain injury. Can J Physiol Pharmacol 93(8):687–694. https://doi.org/10.1139/cjpp-2014-0546
Strlic M, Radovic T, Kolar J, Pihlar B (2002) Anti- and prooxidative properties of gallic acid in fenton-type systems. J Agric Food Chem 50(22):6313–6317
Moghtaderi H, Sepehri H, Delphi L, Attari F (2018) Gallic acid and curcumin induce cytotoxicity and apoptosis in human breast cancer cell MDA-MB-231. BioImpacts : BI 8 (3):185-194. Doi:https://doi.org/10.15171/bi.2018.21
Lee HL, Lin CS, Kao SH, Chou MC (2017) Gallic acid induces G1 phase arrest and apoptosis of triple-negative breast cancer cell MDA-MB-231 via p38 mitogen-activated protein kinase/p21/p27 axis. Anti-Cancer Drugs 28(10):1150–1156. https://doi.org/10.1097/cad.0000000000000565
Acknowledgments
The authors express their gratitude to Dr. C.Y. Chai at the Institute of Biomedical Sciences, Academia Sinica, for his encouragement and support. Special thanks are due to Dr. R.K. Freund at the Department of Pharmacology, University of Colorado, Anschutz, CO, USA, for editing this paper.
Funding
This study was supported by MOST107-2320-B-010-019-MY3 and 107DN08, Taipei, Taiwan, R.O.C.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The use of animals has been approved by the Institutional Animal Care and Use Committee of Taipei Veterans General Hospital, Taipei, Taiwan, R.O.C.. The approval number is IACUC2018. All experiments were performed in the accordance with relevant guidelines and regulation.
Conflict of Interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Supplemental data
Effects of GA on LPS-induced HO-1 in BV-2 cells. (A) BV-2 cells were incubated with LPS (1 μg/mL) plus GA (25-50 μM) for 24 h. Western blot assay was employed for measuring HO-1 expression. Graphs show statistical results from HO-1 protein expression. Values are the mean ± S.E.M. (n = 3/group). *, p < 0.05 in the LPS-treated BV-2 cells compared with vehicle-treated BV-2 cells; #, p < 0.05 in the LPS plus GA-treated BV-2 cells compared with LPS-treated BV-2 cells by t-test. (PNG 199 kb)
Rights and permissions
About this article
Cite this article
Liu, YL., Hsu, CC., Huang, HJ. et al. Gallic Acid Attenuated LPS-Induced Neuroinflammation: Protein Aggregation and Necroptosis. Mol Neurobiol 57, 96–104 (2020). https://doi.org/10.1007/s12035-019-01759-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-019-01759-7