Abstract
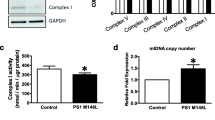
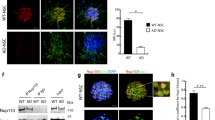
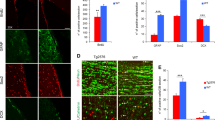
Alzheimer’s disease (AD) is the most common neurodegenerative disease and is characterized by the accumulation of amyloid β peptide (Aβ). Although most AD mouse models present a decline in neurogenesis, they express mutated genes which regulate neurogenesis per se and are not present in most AD patients, thus masking the real impact of Aβ on adult neurogenesis. Mitochondrion, a well-known target of Aβ in neurons, is a main regulator of neural stem cell (NSC) fate. Here, we aimed to investigate the impact of Aβ on NSC mitochondria and cell fate decisions, namely whether and how Aβ affects neurogenesis. NSC fate and mitochondrial parameters, including biogenesis, dynamics, and oxidative stress, were evaluated. Our results showed that Aβ impaired NSC viability and proliferation and indirectly blocked neurogenic differentiation, by disrupting mitochondrial signaling of self-renewing NSCs. Importantly, Aβ decreased ATP levels, generated oxidative stress, and affected the radical scavenger system through SOD2 and SIRT3. Aβ also reduced mtDNA and mitochondrial biogenesis proteins, such as Tfam, PGC-1α, and NRF1, and inhibited activation of PGC-1α-positive regulator CREB. Moreover, Aβ triggered mitochondrial fragmentation in self-renewing NSCs and reduced mitochondrial fusion proteins, such as Mfn2 and ERRα. Notably, Aβ compromised NSC commitment and survival by irreversibly impairing mitochondria and thwarting any neurogenic rescue through mitochondrial biogenesis, dynamics, or radical scavenger system. Altogether, this study brings new perspective to rethink the molecular targets relevant for endogenous NSC-based strategies in AD.






Similar content being viewed by others
Abbreviations
- Aβ:
-
Amyloid β
- AD:
-
Alzheimer’s disease
- APP:
-
Amyloid precursor protein
- BrdU:
-
Bromodeoxyuridine
- CREB:
-
cAMP-responsive element-binding protein
- DRP1:
-
Dynamin-related protein 1
- ERRα:
-
Estrogen-related receptor α
- FBS:
-
Fetal bovine serum
- FGF:
-
Fibroblast growth factor
- GAPDH:
-
Glyceraldehyde 3-phosphate dehydrogenase
- GFAP:
-
Glial fibrillary acidic protein
- GFP:
-
Green fluorescence protein
- MAP2:
-
Microtubule-associated protein 2
- Mfn2:
-
Mitofusin 2
- mtDNA:
-
Mitochondrial DNA
- mtROS:
-
Mitochondrial ROS
- NRF1:
-
Nuclear respiratory factor 1
- NSC:
-
Neural stem cell
- PBS:
-
Phosphate-buffered saline
- PGC-1α:
-
Peroxisome proliferator-activated receptor γ coactivator-1 α
- PSEN1:
-
Presenilin 1
- ROS:
-
Reactive oxygen species
- Sirt3:
-
Sirtuin-3
- SOD2:
-
Superoxide dismutase 2
- Tfam:
-
Mitochondrial transcription factor A
References
Selkoe DJ (2001) Alzheimer’s disease: genes, proteins, and therapy. Physiol Rev 81(2):741–766. https://doi.org/10.1152/physrev.2001.81.2.741
LaFerla FM, Green KN, Oddo S (2007) Intracellular amyloid-beta in Alzheimer’s disease. Nat Rev Neurosci 8(7):499–509. https://doi.org/10.1038/nrn2168
Lazarov O, Mattson MP, Peterson DA, Pimplikar SW, van Praag H (2010) When neurogenesis encounters aging and disease. Trends Neurosci 33(12):569–579. https://doi.org/10.1016/j.tins.2010.09.003
Bizon JL, Lee HJ, Gallagher M (2004) Neurogenesis in a rat model of age-related cognitive decline. Aging Cell 3(4):227–234. https://doi.org/10.1111/j.1474-9728.2004.00099.x
Li B, Yamamori H, Tatebayashi Y, Shafit-Zagardo B, Tanimukai H, Chen S, Iqbal K, Grundke-Iqbal I (2008) Failure of neuronal maturation in Alzheimer disease dentate gyrus. J Neuropathol Exp Neurol 67(1):78–84. https://doi.org/10.1097/nen.0b013e318160c5db
Mu Y, Gage FH (2011) Adult hippocampal neurogenesis and its role in Alzheimer’s disease. Mol Neurodegener 6:85. https://doi.org/10.1186/1750-1326-6-85
Waldau B, Shetty AK (2008) Behavior of neural stem cells in the Alzheimer brain. Cell Mol Life Sci 65(15):2372–2384. https://doi.org/10.1007/s00018-008-8053-y
Choi SH, Veeraraghavalu K, Lazarov O, Marler S, Ransohoff RM, Ramirez JM, Sisodia SS (2008) Non-cell-autonomous effects of presenilin 1 variants on enrichment-mediated hippocampal progenitor cell proliferation and differentiation. Neuron 59(4):568–580. https://doi.org/10.1016/j.neuron.2008.07.033
Haughey NJ, Nath A, Chan SL, Borchard AC, Rao MS, Mattson MP (2002) Disruption of neurogenesis by amyloid beta-peptide, and perturbed neural progenitor cell homeostasis, in models of Alzheimer’s disease. J Neurochem 83(6):1509–1524
Donovan MH, Yazdani U, Norris RD, Games D, German DC, Eisch AJ (2006) Decreased adult hippocampal neurogenesis in the PDAPP mouse model of Alzheimer’s disease. J Comp Neurol 495(1):70–83. https://doi.org/10.1002/cne.20840
Hamilton A, Holscher C (2012) The effect of ageing on neurogenesis and oxidative stress in the APP(swe)/PS1(deltaE9) mouse model of Alzheimer’s disease. Brain Res 1449:83–93. https://doi.org/10.1016/j.brainres.2012.02.015
Hamilton LK, Aumont A, Julien C, Vadnais A, Calon F, Fernandes KJ (2010) Widespread deficits in adult neurogenesis precede plaque and tangle formation in the 3xTg mouse model of Alzheimer’s disease. Eur J Neurosci 32(6):905–920. https://doi.org/10.1111/j.1460-9568.2010.07379.x
Ghosal K, Stathopoulos A, Pimplikar SW (2010) APP intracellular domain impairs adult neurogenesis in transgenic mice by inducing neuroinflammation. PLoS One 5(7):e11866. https://doi.org/10.1371/journal.pone.0011866
Pan H, Wang D, Zhang X, Zhou D, Zhang H, Qian Q, He X, Liu Z et al (2016) Amyloid beta is not the major factor accounting for impaired adult hippocampal neurogenesis in mice overexpressing amyloid precursor protein. Stem Cell Reports 7(4):707–718. https://doi.org/10.1016/j.stemcr.2016.08.019
Wen PH, Hof PR, Chen X, Gluck K, Austin G, Younkin SG, Younkin LH, DeGasperi R et al (2004) The presenilin-1 familial Alzheimer disease mutant P117L impairs neurogenesis in the hippocampus of adult mice. Exp Neurol 188(2):224–237. https://doi.org/10.1016/j.expneurol.2004.04.002
Lazarov O, Marr RA (2010) Neurogenesis and Alzheimer’s disease: at the crossroads. Exp Neurol 223(2):267–281. https://doi.org/10.1016/j.expneurol.2009.08.009
Kadowaki H, Nishitoh H, Urano F, Sadamitsu C, Matsuzawa A, Takeda K, Masutani H, Yodoi J et al (2005) Amyloid beta induces neuronal cell death through ROS-mediated ASK1 activation. Cell Death Differ 12(1):19–24. https://doi.org/10.1038/sj.cdd.4401528
Nitta A, Fukuta T, Hasegawa T, Nabeshima T (1997) Continuous infusion of beta-amyloid protein into the rat cerebral ventricle induces learning impairment and neuronal and morphological degeneration. Jpn J Pharmacol 73(1):51–57
Manczak M, Anekonda TS, Henson E, Park BS, Quinn J, Reddy PH (2006) Mitochondria are a direct site of A beta accumulation in Alzheimer’s disease neurons: implications for free radical generation and oxidative damage in disease progression. Hum Mol Genet 15(9):1437–1449. https://doi.org/10.1093/hmg/ddl066
Mattson MP, Gleichmann M, Cheng A (2008) Mitochondria in neuroplasticity and neurological disorders. Neuron 60(5):748–766. https://doi.org/10.1016/j.neuron.2008.10.010
Veereshwarayya V, Kumar P, Rosen KM, Mestril R, Querfurth HW (2006) Differential effects of mitochondrial heat shock protein 60 and related molecular chaperones to prevent intracellular beta-amyloid-induced inhibition of complex IV and limit apoptosis. J Biol Chem 281(40):29468–29478. https://doi.org/10.1074/jbc.M602533200
Khacho M, Clark A, Svoboda DS, Azzi J, MacLaurin JG, Meghaizel C, Sesaki H, Lagace DC et al (2016) Mitochondrial dynamics impacts stem cell identity and fate decisions by regulating a nuclear transcriptional program. Cell Stem Cell 19(2):232–247. https://doi.org/10.1016/j.stem.2016.04.015
Sena LA, Chandel NS (2012) Physiological roles of mitochondrial reactive oxygen species. Mol Cell 48(2):158–167. https://doi.org/10.1016/j.molcel.2012.09.025
Owusu-Ansah E, Banerjee U (2009) Reactive oxygen species prime Drosophila haematopoietic progenitors for differentiation. Nature 461(7263):537–541. https://doi.org/10.1038/nature08313
Zhang J, Khvorostov I, Hong JS, Oktay Y, Vergnes L, Nuebel E, Wahjudi PN, Setoguchi K et al (2016) UCP2 regulates energy metabolism and differentiation potential of human pluripotent stem cells. EMBO J 35(8):899. https://doi.org/10.15252/embj.201694054
Mitra K (2013) Mitochondrial fission-fusion as an emerging key regulator of cell proliferation and differentiation. Bioessays 35(11):955–964. https://doi.org/10.1002/bies.201300011
Xavier JM, Rodrigues CM, Sola S (2015) Mitochondria: major regulators of neural development. Neuroscientist 22:346–358. https://doi.org/10.1177/1073858415585472
Facucho-Oliveira JM, Alderson J, Spikings EC, Egginton S, St John JC (2007) Mitochondrial DNA replication during differentiation of murine embryonic stem cells. J Cell Sci 120(Pt 22):4025–4034. https://doi.org/10.1242/jcs.016972
Xavier JM, Morgado AL, Sola S, Rodrigues CM (2014) Mitochondrial translocation of p53 modulates neuronal fate by preventing differentiation-induced mitochondrial stress. Antioxid Redox Signal 21(7):1009–1024. https://doi.org/10.1089/ars.2013.5417
Pollard SM, Conti L, Sun Y, Goffredo D, Smith A (2006) Adherent neural stem (NS) cells from fetal and adult forebrain. Cereb Cortex 16(Suppl 1):i112–i120. https://doi.org/10.1093/cercor/bhj167
Conti L, Pollard SM, Gorba T, Reitano E, Toselli M, Biella G, Sun Y, Sanzone S et al (2005) Niche-independent symmetrical self-renewal of a mammalian tissue stem cell. PLoS Biol 3(9):e283. https://doi.org/10.1371/journal.pbio.0030283
Fuentealba LC, Rompani SB, Parraguez JI, Obernier K, Romero R, Cepko CL, Alvarez-Buylla A (2015) Embryonic origin of postnatal neural stem cells. Cell 161(7):1644–1655. https://doi.org/10.1016/j.cell.2015.05.041
Fonseca MB, Sola S, Xavier JM, Dionisio PA, Rodrigues CM (2013) Amyloid beta peptides promote autophagy-dependent differentiation of mouse neural stem cells: Abeta-mediated neural differentiation. Mol Neurobiol 48(3):829–840. https://doi.org/10.1007/s12035-013-8471-1
Glaser T, Pollard SM, Smith A, Brustle O (2007) Tripotential differentiation of adherently expandable neural stem (NS) cells. PLoS One 2(3):e298. https://doi.org/10.1371/journal.pone.0000298
Vandersteen A, Hubin E, Sarroukh R, De Baets G, Schymkowitz J, Rousseau F, Subramaniam V, Raussens V et al (2012) A comparative analysis of the aggregation behavior of amyloid-beta peptide variants. FEBS Lett 586(23):4088–4093. https://doi.org/10.1016/j.febslet.2012.10.022
Morgado AL, Xavier JM, Dionisio PA, Ribeiro MF, Dias RB, Sebastiao AM, Sola S, Rodrigues CM (2015) MicroRNA-34a modulates neural stem cell differentiation by regulating expression of synaptic and autophagic proteins. Mol Neurobiol 51(3):1168–1183. https://doi.org/10.1007/s12035-014-8794-6
Xavier JM, Morgado AL, Rodrigues CM, Sola S (2014) Tauroursodeoxycholic acid increases neural stem cell pool and neuronal conversion by regulating mitochondria-cell cycle retrograde signaling. Cell Cycle 13(22):3576–3589. https://doi.org/10.4161/15384101.2014.962951
Eaton JS, Lin ZP, Sartorelli AC, Bonawitz ND, Shadel GS (2007) Ataxia-telangiectasia mutated kinase regulates ribonucleotide reductase and mitochondrial homeostasis. J Clin Invest 117(9):2723–2734. https://doi.org/10.1172/JCI31604
Chen H, Detmer SA, Ewald AJ, Griffin EE, Fraser SE, Chan DC (2003) Mitofusins Mfn1 and Mfn2 coordinately regulate mitochondrial fusion and are essential for embryonic development. J Cell Biol 160(2):189–200. https://doi.org/10.1083/jcb.200211046
Doe CQ (2008) Neural stem cells: balancing self-renewal with differentiation. Development 135(9):1575–1587. https://doi.org/10.1242/dev.014977
Ma DK, Bonaguidi MA, Ming GL, Song H (2009) Adult neural stem cells in the mammalian central nervous system. Cell Res 19(6):672–682. https://doi.org/10.1038/cr.2009.56
Sundaresan NR, Gupta M, Kim G, Rajamohan SB, Isbatan A, Gupta MP (2009) Sirt3 blocks the cardiac hypertrophic response by augmenting Foxo3a-dependent antioxidant defense mechanisms in mice. J Clin Invest 119(9):2758–2771. https://doi.org/10.1172/JCI39162
Kong X, Wang R, Xue Y, Liu X, Zhang H, Chen Y, Fang F, Chang Y (2010) Sirtuin 3, a new target of PGC-1alpha, plays an important role in the suppression of ROS and mitochondrial biogenesis. PLoS One 5(7):e11707. https://doi.org/10.1371/journal.pone.0011707
Cho YM, Kwon S, Pak YK, Seol HW, Choi YM, Park DJ, Park KS, Lee HK (2006) Dynamic changes in mitochondrial biogenesis and antioxidant enzymes during the spontaneous differentiation of human embryonic stem cells. Biochem Biophys Res Commun 348(4):1472–1478. https://doi.org/10.1016/j.bbrc.2006.08.020
Prigione A, Adjaye J (2010) Modulation of mitochondrial biogenesis and bioenergetic metabolism upon in vitro and in vivo differentiation of human ES and iPS cells. Int J Dev Biol 54(11–12):1729–1741. https://doi.org/10.1387/ijdb.103198ap
Collu-Marchese M, Shuen M, Pauly M, Saleem A, Hood DA (2015) The regulation of mitochondrial transcription factor A (Tfam) expression during skeletal muscle cell differentiation. Biosci Rep 35(3). https://doi.org/10.1042/BSR20150073
Fernandez-Marcos PJ, Auwerx J (2011) Regulation of PGC-1alpha, a nodal regulator of mitochondrial biogenesis. Am J Clin Nutr 93 (4):884S-890. doi:https://doi.org/10.3945/ajcn.110.001917
Piantadosi CA, Suliman HB (2006) Mitochondrial transcription factor A induction by redox activation of nuclear respiratory factor 1. J Biol Chem 281(1):324–333. https://doi.org/10.1074/jbc.M508805200
Naqvi S, Martin KJ, Arthur JS (2014) CREB phosphorylation at Ser133 regulates transcription via distinct mechanisms downstream of cAMP and MAPK signalling. Biochem J 458(3):469–479. https://doi.org/10.1042/BJ20131115
Karbowski M, Youle RJ (2003) Dynamics of mitochondrial morphology in healthy cells and during apoptosis. Cell Death Differ 10(8):870–880. https://doi.org/10.1038/sj.cdd.4401260
Fang D, Yan S, Yu Q, Chen D, Yan SS (2016) Mfn2 is required for mitochondrial development and synapse formation in human induced pluripotent stem cells/hiPSC derived cortical neurons. Sci Rep 6:31462. https://doi.org/10.1038/srep31462
Ryan JJ, Marsboom G, Fang YH, Toth PT, Morrow E, Luo N, Piao L, Hong Z et al (2013) PGC1alpha-mediated mitofusin-2 deficiency in female rats and humans with pulmonary arterial hypertension. Am J Respir Crit Care Med 187(8):865–878. https://doi.org/10.1164/rccm.201209-1687OC
Breton-Provencher V, Lemasson M, Peralta MR 3rd, Saghatelyan A (2009) Interneurons produced in adulthood are required for the normal functioning of the olfactory bulb network and for the execution of selected olfactory behaviors. J Neurosci 29(48):15245–15257. https://doi.org/10.1523/JNEUROSCI.3606-09.2009
Deng W, Aimone JB, Gage FH (2010) New neurons and new memories: how does adult hippocampal neurogenesis affect learning and memory? Nat Rev Neurosci 11(5):339–350. https://doi.org/10.1038/nrn2822
Demars M, Hu YS, Gadadhar A, Lazarov O (2010) Impaired neurogenesis is an early event in the etiology of familial Alzheimer’s disease in transgenic mice. J Neurosci Res 88(10):2103–2117. https://doi.org/10.1002/jnr.22387
Verret L, Jankowsky JL, Xu GM, Borchelt DR, Rampon C (2007) Alzheimer’s-type amyloidosis in transgenic mice impairs survival of newborn neurons derived from adult hippocampal neurogenesis. J Neurosci 27(25):6771–6780. https://doi.org/10.1523/JNEUROSCI.5564-06.2007
Zhang C, McNeil E, Dressler L, Siman R (2007) Long-lasting impairment in hippocampal neurogenesis associated with amyloid deposition in a knock-in mouse model of familial Alzheimer’s disease. Exp Neurol 204(1):77–87. https://doi.org/10.1016/j.expneurol.2006.09.018
He N, Jin WL, Lok KH, Wang Y, Yin M, Wang ZJ (2013) Amyloid-beta(1-42) oligomer accelerates senescence in adult hippocampal neural stem/progenitor cells via formylpeptide receptor 2. Cell Death Dis 4:e924. https://doi.org/10.1038/cddis.2013.437
Shruster A, Eldar-Finkelman H, Melamed E, Offen D (2011) Wnt signaling pathway overcomes the disruption of neuronal differentiation of neural progenitor cells induced by oligomeric amyloid beta-peptide. J Neurochem 116(4):522–529. https://doi.org/10.1111/j.1471-4159.2010.07131.x
Lee IS, Jung K, Kim IS, Park KI (2013) Amyloid-beta oligomers regulate the properties of human neural stem cells through GSK-3beta signaling. Exp Mol Med 45:e60. https://doi.org/10.1038/emm.2013.125
Chiang MC, Cheng YC, Chen SJ, Yen CH, Huang RN (2016) Metformin activation of AMPK-dependent pathways is neuroprotective in human neural stem cells against Amyloid-beta-induced mitochondrial dysfunction. Exp Cell Res 347:322–331. https://doi.org/10.1016/j.yexcr.2016.08.013
Lovell MA, Geiger H, Van Zant GE, Lynn BC, Markesbery WR (2006) Isolation of neural precursor cells from Alzheimer’s disease and aged control postmortem brain. Neurobiol Aging 27(7):909–917. https://doi.org/10.1016/j.neurobiolaging.2005.05.004
Kincaid B, Bossy-Wetzel E (2013) Forever young: SIRT3 a shield against mitochondrial meltdown, aging, and neurodegeneration. Front Aging Neurosci 5:48. https://doi.org/10.3389/fnagi.2013.00048
Park D, Xiang AP, Mao FF, Zhang L, Di CG, Liu XM, Shao Y, Ma BF et al (2010) Nestin is required for the proper self-renewal of neural stem cells. Stem Cells 28(12):2162–2171. https://doi.org/10.1002/stem.541
Antico Arciuch VG, Elguero ME, Poderoso JJ, Carreras MC (2012) Mitochondrial regulation of cell cycle and proliferation. Antioxid Redox Signal 16(10):1150–1180. https://doi.org/10.1089/ars.2011.4085
Kumar A, Singh A (2015) A review on mitochondrial restorative mechanism of antioxidants in Alzheimer’s disease and other neurological conditions. Front Pharmacol 6:206. https://doi.org/10.3389/fphar.2015.00206
Esposito L, Raber J, Kekonius L, Yan F, Yu GQ, Bien-Ly N, Puolivali J, Scearce-Levie K et al (2006) Reduction in mitochondrial superoxide dismutase modulates Alzheimer’s disease-like pathology and accelerates the onset of behavioral changes in human amyloid precursor protein transgenic mice. J Neurosci 26(19):5167–5179. https://doi.org/10.1523/JNEUROSCI.0482-06.2006
Wu S, Zhou F, Zhang Z, Xing D (2011) Mitochondrial oxidative stress causes mitochondrial fragmentation via differential modulation of mitochondrial fission-fusion proteins. FEBS J 278(6):941–954. https://doi.org/10.1111/j.1742-4658.2011.08010.x
Youle RJ, van der Bliek AM (2012) Mitochondrial fission, fusion, and stress. Science 337(6098):1062–1065. https://doi.org/10.1126/science.1219855
Manczak M, Calkins MJ, Reddy PH (2011) Impaired mitochondrial dynamics and abnormal interaction of amyloid beta with mitochondrial protein Drp1 in neurons from patients with Alzheimer’s disease: implications for neuronal damage. Hum Mol Genet 20(13):2495–2509. https://doi.org/10.1093/hmg/ddr139
Park J, Choi H, Min JS, Kim B, Lee SR, Yun JW, Choi MS, Chang KT et al (2015) Loss of mitofusin 2 links beta-amyloid-mediated mitochondrial fragmentation and Cdk5-induced oxidative stress in neuron cells. J Neurochem 132(6):687–702. https://doi.org/10.1111/jnc.12984
Soriano FX, Liesa M, Bach D, Chan DC, Palacin M, Zorzano A (2006) Evidence for a mitochondrial regulatory pathway defined by peroxisome proliferator-activated receptor-gamma coactivator-1 alpha, estrogen-related receptor-alpha, and mitofusin 2. Diabetes 55(6):1783–1791. https://doi.org/10.2337/db05-0509
Sato A, Nakada K, Hayashi J (2009) Mitochondrial complementation preventing respiratory dysfunction caused by mutant mtDNA. Biofactors 35(2):130–137. https://doi.org/10.1002/biof.14
Wang W, Esbensen Y, Kunke D, Suganthan R, Rachek L, Bjoras M, Eide L (2011) Mitochondrial DNA damage level determines neural stem cell differentiation fate. J Neurosci 31(26):9746–9751. https://doi.org/10.1523/JNEUROSCI.0852-11.2011
Chen H, Vermulst M, Wang YE, Chomyn A, Prolla TA, McCaffery JM, Chan DC (2010) Mitochondrial fusion is required for mtDNA stability in skeletal muscle and tolerance of mtDNA mutations. Cell 141(2):280–289. https://doi.org/10.1016/j.cell.2010.02.026
Jarvis P, Lopez-Juez E (2013) Biogenesis and homeostasis of chloroplasts and other plastids. Nat Rev Mol Cell Biol 14(12):787–802. https://doi.org/10.1038/nrm3702
Quiros PM, Mottis A, Auwerx J (2016) Mitonuclear communication in homeostasis and stress. Nat Rev Mol Cell Biol 17(4):213–226. https://doi.org/10.1038/nrm.2016.23
Qin W, Haroutunian V, Katsel P, Cardozo CP, Ho L, Buxbaum JD, Pasinetti GM (2009) PGC-1alpha expression decreases in the Alzheimer disease brain as a function of dementia. Arch Neurol 66(3):352–361. https://doi.org/10.1001/archneurol.2008.588
Pugazhenthi S, Wang M, Pham S, Sze CI, Eckman CB (2011) Downregulation of CREB expression in Alzheimer’s brain and in Abeta-treated rat hippocampal neurons. Mol Neurodegener 6:60. https://doi.org/10.1186/1750-1326-6-60
Bartolome F, de la Cueva M, Pascual C, Antequera D, Fernandez T, Gil C, Martinez A, Carro E (2018) Amyloid beta-induced impairments on mitochondrial dynamics, hippocampal neurogenesis, and memory are restored by phosphodiesterase 7 inhibition. Alzheimers Res Ther 10(1):24. https://doi.org/10.1186/s13195-018-0352-4
Amato S, Man HY (2011) Bioenergy sensing in the brain: the role of AMP-activated protein kinase in neuronal metabolism, development and neurological diseases. Cell Cycle 10(20):3452–3460. https://doi.org/10.4161/cc.10.20.17953
Hardie DG, Ross FA, Hawley SA (2012) AMPK: a nutrient and energy sensor that maintains energy homeostasis. Nat Rev Mol Cell Biol 13(4):251–262. https://doi.org/10.1038/nrm3311
Acknowledgements
We are thankful to Dr. Jorge L. Ruas (Karolinska Institutet, Sweden) and Dr. David C. Chan (California Institute of Technology, USA) for kindly providing PGC-1α and Mfn2-Myc overexpression vectors, respectively. Finally, we thank all members of the laboratory, and especially Marta Fernandes, for insightful discussions.
Funding
This work was supported by UID/DTP/04138/2013, SFRH/BD/100674/2014, and SAICTPAC/0019/2015 from Fundação para a Ciência e Tecnologia, Portugal.
Author information
Authors and Affiliations
Contributions
MFR, CMPR, and SS conceived and designed the experiments. MFR performed the experiments. TG contributed in establishing the in vitro model. MFR, SS, and CMPR analyzed and interpreted the data. ACR contributed with reagents. MFR wrote the manuscript. CMPR and SS critically reviewed the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Electronic Supplementary Material
ESM 1
Tris buffer did not affect NSC viability. Mouse NSCs were treated with volumes of Tris buffer equivalent to 1.5, 5 or 10 μM Aβ in self-renewal conditions. After 24 h of Aβ treatment, cells were collected to assess viability and proliferation as described in Materials and Methods. Data represents mean values ± SEM for at least three independent experiments. (PNG 31 kb)
ESM 2
Aβ peptide blocks NSC cycle re-enter. Mouse NSCs were treated with 10 μM Aβ in self-renewal conditions. After 48 h of treatment, cells were collected to assess proliferation by qRT-PCR of Ki67 expression. Data represents mean values ± SEM for at least three independent experiments. ***p < 0.001 compared to untreated cells. (PNG 20 kb)
ESM 3
Neurogenesis inhibition by Aβ is not due to a double dosage the amyloid peptide. Five micromolar of Aβ was incubated at the same time of neuronal differentiation induction, for 48 h, with 24 h of 5 μM Aβ pre-incubation in self-renewal conditions. Samples were collected for flow cytometry analysis. Histograms of positive cells for nestin, βIII-tubulin, and MAP2 (top). Experimental scheme representing the protocol of treatment (bottom). (PNG 98 kb)
ESM 4
Aβ inhibits gliogenic differentiation. Mouse NSCs were treated with Aβ (10 μM) for 48 h at the same time of glial differentiation induction. Samples were collected for flow cytometry analysis. Percentage of positive cells for nestin and GFAP were analyzed (top). Experimental scheme representing the protocol of treatment (bottom). Data represents mean values ± SEM for at least three independent experiments. *p < 0.05, **p < 0.01 and ***p < 0.001 compared to untreated cells. (PNG 38 kb)
Rights and permissions
About this article
Cite this article
Ribeiro, M.F., Genebra, T., Rego, A.C. et al. Amyloid β Peptide Compromises Neural Stem Cell Fate by Irreversibly Disturbing Mitochondrial Oxidative State and Blocking Mitochondrial Biogenesis and Dynamics. Mol Neurobiol 56, 3922–3936 (2019). https://doi.org/10.1007/s12035-018-1342-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-018-1342-z