Abstract
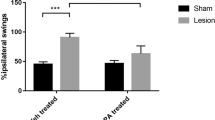
Parkinson’s disease (PD) is a neurodegenerative disease characterized by degeneration of dopaminergic neurons. Aging is a major risk factor for idiopathic PD. Several prior studies examined the neuroprotective effects of palmitoylethanolamide (PEA), alone or combined with antioxidants, in a model of PD induced by the dopaminergic toxin 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP). Here, we analyzed the pretreatment effect of micronized PEA (PEAm) on neuroinflammation and neuronal cell death in the MPTP model. Male CD mice (21 months of age) were pre-treated for 60 days with PEAm. After this time, they received four intraperitoneal injections of MPTP over a 24-h period and were killed 7 days later. On the 8th day, brains were processed. Pretreatment with PEAm ameliorated behavioral deficits and the reductions in expression of tyrosine hydroxylase and dopamine transporter, while blunting the upregulation of α-synuclein and β3-tubulin in the substantia nigra after MPTP induction. Moreover, PEAm reduced proinflammatory cytokine expression and showed a pro-neurogenic effect in hippocampus. These findings propose this strategy as a valid approach to prevent neurodegenerative diseases associated with old age.














Similar content being viewed by others
References
Lee YI, Kang H, Ha YW, Chang KY, Cho SC, Song SO, Kim H, Jo A et al (2016) Diaminodiphenyl sulfone-induced parkin ameliorates age-dependent dopaminergic neuronal loss. Neurobiol Aging 41:1–10. https://doi.org/10.1016/j.neurobiolaging.2015.11.008
Cascianelli S, Tranfaglia C, Fravolini ML, Bianconi F, Minestrini M, Nuvoli S, Tambasco N, Dottorini ME, Palumbo B (2017) Right putamen and age are the most discriminant features to diagnose Parkinson's disease by using (123)I-FP-CIT brain SPET data by using an artificial neural network classifier, a classification tree (ClT). Hell J Nucl Med 20 Suppl:165
Calabrese V, Santoro A, Monti D, Crupi R, Di Paola R, Latteri S, Cuzzocrea S, Zappia M et al (2018) Aging and Parkinson's disease: inflammaging, neuroinflammation and biological remodeling as key factors in pathogenesis. Free Radic Biol Med 115:80–91. https://doi.org/10.1016/j.freeradbiomed.2017.10.379
Baluchnejadmojarad T, Eftekhari SM, Jamali-Raeufy N, Haghani S, Zeinali H, Roghani M (2017) The anti-aging protein klotho alleviates injury of nigrostriatal dopaminergic pathway in 6-hydroxydopamine rat model of Parkinson's disease: Involvement of PKA/CaMKII/CREB signaling. Exp Gerontol 100:70–76. https://doi.org/10.1016/j.exger.2017.10.023
Olanow CW, Tatton WG (1999) Etiology and pathogenesis of Parkinson's disease. Annu Rev Neurosci 22:123–144. https://doi.org/10.1146/annurev.neuro.22.1.123
Becker G (2003) Methods for the early diagnosis of Parkinson's disease. Nervenarzt 74(Suppl 1):S7–11. https://doi.org/10.1007/s00115-003-1482-9
Rodriguez M, Rodriguez-Sabate C, Morales I, Sanchez A, Sabate M (2015) Parkinson's disease as a result of aging. Aging Cell 14(3):293–308. https://doi.org/10.1111/acel.12312
Branch SY, Sharma R, Beckstead MJ (2014) Aging decreases L-type calcium channel currents and pacemaker firing fidelity in substantia nigra dopamine neurons. The Journal of neuroscience : the official journal of the Society for Neuroscience 34(28):9310–9318. https://doi.org/10.1523/JNEUROSCI.4228-13.2014
Esposito E, Di Matteo V, Benigno A, Pierucci M, Crescimanno G, Di Giovanni G (2007) Non-steroidal anti-inflammatory drugs in Parkinson's disease. Exp Neurol 205(2):295–312. https://doi.org/10.1016/j.expneurol.2007.02.008
Schulz JB, Falkenburger BH (2004) Neuronal pathology in Parkinson's disease. Cell Tissue Res 318(1):135–147. https://doi.org/10.1007/s00441-004-0954-y
Liberatore GT, Jackson-Lewis V, Vukosavic S, Mandir AS, Vila M, McAuliffe WG, Dawson VL, Dawson TM et al (1999) Inducible nitric oxide synthase stimulates dopaminergic neurodegeneration in the MPTP model of Parkinson disease. Nat Med 5(12):1403–1409. https://doi.org/10.1038/70978
Huang D, Wang Z, Tong J, Wang M, Wang J, Xu J, Bai X, Li H et al (2018) Long-term changes in the nigrostriatal pathway in the MPTP mouse model of Parkinson's disease. Neuroscience 369:303–313. https://doi.org/10.1016/j.neuroscience.2017.11.041
Kohutnicka M, Lewandowska E, Kurkowska-Jastrzebska I, Czlonkowski A, Czlonkowska A (1998) Microglial and astrocytic involvement in a murine model of Parkinson's disease induced by 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP). Immunopharmacology 39(3):167–180
Liu J, Huang D, Xu J, Tong J, Wang Z, Huang L, Yang Y, Bai X et al (2015) Tiagabine protects dopaminergic neurons against neurotoxins by inhibiting microglial activation. Sci Rep 5:15720. https://doi.org/10.1038/srep15720
Shimoi K, Masuda S, Furugori M, Esaki S, Kinae N (1994) Radioprotective effect of antioxidative flavonoids in gamma-ray irradiated mice. Carcinogenesis 15(11):2669–2672
Lin Y, Shi R, Wang X, Shen HM (2008) Luteolin, a flavonoid with potential for cancer prevention and therapy. Curr Cancer Drug Targets 8(7):634–646
Lambert DM, Vandevoorde S, Jonsson KO, Fowler CJ (2002) The palmitoylethanolamide family: a new class of anti-inflammatory agents? Curr Med Chem 9(6):663–674
Hansen HS (2010) Palmitoylethanolamide and other anandamide congeners. Proposed role in the diseased brain. Exp Neurol 224(1):48–55. https://doi.org/10.1016/j.expneurol.2010.03.022
De Filippis D, D'Amico A, Iuvone T (2008) Cannabinomimetic control of mast cell mediator release: new perspective in chronic inflammation. J Neuroendocrinol 20(Suppl 1):20–25. https://doi.org/10.1111/j.1365-2826.2008.01674.x
Koch M, Kreutz S, Bottger C, Benz A, Maronde E, Ghadban C, Korf HW, Dehghani F (2011) Palmitoylethanolamide protects dentate gyrus granule cells via peroxisome proliferator-activated receptor-alpha. Neurotox Res 19(2):330–340. https://doi.org/10.1007/s12640-010-9166-2
Sasso O, Russo R, Vitiello S, Raso GM, D'Agostino G, Iacono A, Rana GL, Vallee M et al (2012) Implication of allopregnanolone in the antinociceptive effect of N-palmitoylethanolamide in acute or persistent pain. Pain 153(1):33–41. https://doi.org/10.1016/j.pain.2011.08.010
Esposito E, Impellizzeri D, Mazzon E, Paterniti I, Cuzzocrea S (2012) Neuroprotective activities of palmitoylethanolamide in an animal model of Parkinson's disease. PLoS One 7(8):e41880. https://doi.org/10.1371/journal.pone.0041880
Esposito E, Paterniti I, Mazzon E, Genovese T, Di Paola R, Galuppo M, Cuzzocrea S (2011) Effects of palmitoylethanolamide on release of mast cell peptidases and neurotrophic factors after spinal cord injury. Brain Behav Immun 25(6):1099–1112. https://doi.org/10.1016/j.bbi.2011.02.006
Genovese T, Esposito E, Mazzon E, Di Paola R, Meli R, Bramanti P, Piomelli D, Calignano A et al (2008) Effects of palmitoylethanolamide on signaling pathways implicated in the development of spinal cord injury. J Pharmacol Exp Ther 326(1):12–23. https://doi.org/10.1124/jpet.108.136903
Antzoulatos E, Jakowec MW, Petzinger GM, Wood RI (2011) MPTP neurotoxicity and testosterone induce dendritic remodeling of striatal medium spiny neurons in the C57Bl/6 mouse. Parkinson's disease 2011:138471–138410. https://doi.org/10.4061/2011/138471
Fleming SM, Salcedo J, Fernagut PO, Rockenstein E, Masliah E, Levine MS, Chesselet MF (2004) Early and progressive sensorimotor anomalies in mice overexpressing wild-type human alpha-synuclein. The Journal of neuroscience : the official journal of the Society for Neuroscience 24(42):9434–9440. https://doi.org/10.1523/JNEUROSCI.3080-04.2004
Hwang DY, Fleming SM, Ardayfio P, Moran-Gates T, Kim H, Tarazi FI, Chesselet MF, Kim KS (2005) 3,4-dihydroxyphenylalanine reverses the motor deficits in Pitx3-deficient aphakia mice: behavioral characterization of a novel genetic model of Parkinson's disease. The Journal of neuroscience : the official journal of the Society for Neuroscience 25(8):2132–2137. https://doi.org/10.1523/JNEUROSCI.3718-04.2005
Pellow S, Chopin P, File SE, Briley M (1985) Validation of open:closed arm entries in an elevated plus-maze as a measure of anxiety in the rat. J Neurosci Methods 14(3):149–167
Siracusa R, Paterniti I, Impellizzeri D, Cordaro M, Crupi R, Navarra M, Cuzzocrea S, Esposito E The Association of Palmitoylethanolamide with Luteolin Decreases Neuroinflammation and Stimulates Autophagy in Parkinson's Disease Model. CNS Neurol Disord Drug Targets 14 (10):1350–1365. doi:CNSNDDT-EPUB-69743 [pii]
Siracusa R, Paterniti I, Cordaro M, Crupi R, Bruschetta G, Campolo M, Cuzzocrea S, Esposito E (2017) Neuroprotective effects of Temsirolimus in animal models of Parkinson's disease. Mol Neurobiol. https://doi.org/10.1007/s12035-017-0496-4
Lee KW, Zhao X, Im JY, Grosso H, Jang WH, Chan TW, Sonsalla PK, German DC et al (2012) Apoptosis signal-regulating kinase 1 mediates MPTP toxicity and regulates glial activation. PLoS One 7(1):e29935. https://doi.org/10.1371/journal.pone.0029935
Siracusa R, Impellizzeri D, Cordaro M, Crupi R, Esposito E, Petrosino S, Cuzzocrea S (2017) Anti-inflammatory and neuroprotective effects of co-ultraPEALut in a mouse model of vascular dementia. Front Neurol 8:233. https://doi.org/10.3389/fneur.2017.00233
Cordaro M, Paterniti I, Siracusa R, Impellizzeri D, Esposito E, Cuzzocrea S (2017) KU0063794, a dual mTORC1 and mTORC2 inhibitor, reduces neural tissue damage and locomotor impairment after spinal cord injury in mice. Mol Neurobiol 54(4):2415–2427. https://doi.org/10.1007/s12035-016-9827-0
Demetrius L (2006) Aging in mouse and human systems: a comparative study. Ann N Y Acad Sci 1067:66–82. https://doi.org/10.1196/annals.1354.010
Dutta S, Sengupta P (2016) Men and mice: relating their ages. Life Sci 152:244–248. https://doi.org/10.1016/j.lfs.2015.10.025
Erro R, Picillo M, Vitale C, Amboni M, Moccia M, Santangelo G, Pellecchia MT, Barone P (2016) The non-motor side of the honeymoon period of Parkinson's disease and its relationship with quality of life: a 4-year longitudinal study. Eur J Neurol 23(11):1673–1679. https://doi.org/10.1111/ene.13106
Hall H, Reyes S, Landeck N, Bye C, Leanza G, Double K, Thompson L, Halliday G et al (2014) Hippocampal Lewy pathology and cholinergic dysfunction are associated with dementia in Parkinson's disease. Brain : a journal of neurology 137(Pt 9):2493–2508. https://doi.org/10.1093/brain/awu193
van Mierlo TJ, Chung C, Foncke EM, Berendse HW, van den Heuvel OA (2015) Depressive symptoms in Parkinson's disease are related to decreased hippocampus and amygdala volume. Movement disorders: official journal of the Movement Disorder Society 30(2):245–252. https://doi.org/10.1002/mds.26112
Ouchi Y, Yoshikawa E, Sekine Y, Futatsubashi M, Kanno T, Ogusu T, Torizuka T (2005) Microglial activation and dopamine terminal loss in early Parkinson's disease. Ann Neurol 57(2):168–175. https://doi.org/10.1002/ana.20338
McGeer PL, McGeer EG (2002) Innate immunity, local inflammation, and degenerative disease. Science of aging knowledge environment : SAGE KE 2002 (29):re3. https://doi.org/10.1126/sageke.2002.29.re3
Hald A, Lotharius J (2005) Oxidative stress and inflammation in Parkinson's disease: is there a causal link? Exp Neurol 193(2):279–290. https://doi.org/10.1016/j.expneurol.2005.01.013
Marchetti B, Abbracchio MP (2005) To be or not to be (inflamed)—is that the question in anti-inflammatory drug therapy of neurodegenerative disorders? Trends Pharmacol Sci 26(10):517–525. https://doi.org/10.1016/j.tips.2005.08.007
Calignano A, La Rana G, Piomelli D (2001) Antinociceptive activity of the endogenous fatty acid amide, palmitylethanolamide. Eur J Pharmacol 419(2–3):191–198
Naccarato M, Chiodo Grandi F, Dennis M, Sandercock PA (2010) Physical methods for preventing deep vein thrombosis in stroke. The Cochrane database of systematic reviews 8:CD001922. https://doi.org/10.1002/14651858.CD001922.pub3
Crupi R, Paterniti I, Ahmad A, Campolo M, Esposito E, Cuzzocrea S (2013) Effects of palmitoylethanolamide and luteolin in an animal model of anxiety/depression. CNS & neurological disorders drug targets 12(7):989–1001
Palee S, Apaijai N, Shinlapawittayatorn K, Chattipakorn SC, Chattipakorn N (2016) Acetylcholine attenuates hydrogen peroxide-induced intracellular calcium dyshomeostasis through both muscarinic and nicotinic receptors in cardiomyocytes. Cellular physiology and biochemistry : international journal of experimental cellular physiology, biochemistry, and pharmacology 39(1):341–349. https://doi.org/10.1159/000445628
Bertolino B, Crupi R, Impellizzeri D, Bruschetta G, Cordaro M, Siracusa R, Esposito E, Cuzzocrea S (2017) Beneficial effects of co-ultramicronized palmitoylethanolamide/luteolin in a mouse model of autism and in a case report of autism. CNS neuroscience & therapeutics 23(1):87–98. https://doi.org/10.1111/cns.12648
Asakawa T, Fang H, Sugiyama K, Nozaki T, Hong Z, Yang Y, Hua F, Ding G et al (2016) Animal behavioral assessments in current research of Parkinson's disease. Neurosci Biobehav Rev 65:63–94. https://doi.org/10.1016/j.neubiorev.2016.03.016
Abeliovich A, Schmitz Y, Farinas I, Choi-Lundberg D, Ho WH, Castillo PE, Shinsky N, Verdugo JM et al (2000) Mice lacking alpha-synuclein display functional deficits in the nigrostriatal dopamine system. Neuron 25(1):239–252
Nemani VM, Lu W, Berge V, Nakamura K, Onoa B, Lee MK, Chaudhry FA, Nicoll RA et al (2010) Increased expression of alpha-synuclein reduces neurotransmitter release by inhibiting synaptic vesicle reclustering after endocytosis. Neuron 65(1):66–79. https://doi.org/10.1016/j.neuron.2009.12.023
Scott D, Roy S (2012) Alpha-synuclein inhibits intersynaptic vesicle mobility and maintains recycling-pool homeostasis. The Journal of neuroscience : the official journal of the Society for Neuroscience 32(30):10129–10135. https://doi.org/10.1523/JNEUROSCI.0535-12.2012
Cameron HA, Woolley CS, McEwen BS, Gould E (1993) Differentiation of newly born neurons and glia in the dentate gyrus of the adult rat. Neuroscience 56(2):337–344
Toni N, Laplagne DA, Zhao C, Lombardi G, Ribak CE, Gage FH, Schinder AF (2008) Neurons born in the adult dentate gyrus form functional synapses with target cells. Nat Neurosci 11(8):901–907. https://doi.org/10.1038/nn.2156
Parent JM, Jessberger S, Gage FH, Gong C (2007) Is neurogenesis reparative after status epilepticus? Epilepsia 48(Suppl 8):69–71
Braak H, Del Tredici K, Rub U, de Vos RA, Jansen Steur EN, Braak E (2003) Staging of brain pathology related to sporadic Parkinson's disease. Neurobiol Aging 24(2):197–211
Mayer RJ, Lowe J, Lennox G, Landon M, MacLennan K, Doherty FJ (1989) Intermediate filament-ubiquitin diseases: implications for cell sanitization. Biochem Soc Symp 55:193–201
Galloway PG, Mulvihill P, Perry G (1992) Filaments of Lewy bodies contain insoluble cytoskeletal elements. Am J Pathol 140(4):809–822
Langston JW, Ballard P, Tetrud JW, Irwin I (1983) Chronic parkinsonism in humans due to a product of meperidine-analog synthesis. Science 219(4587):979–980
Przedborski S, Jackson-Lewis V (1998) Mechanisms of MPTP toxicity. Movement disorders: official journal of the Movement Disorder Society 13(Suppl 1):35–38
Betarbet R, Sherer TB, MacKenzie G, Garcia-Osuna M, Panov AV, Greenamyre JT (2000) Chronic systemic pesticide exposure reproduces features of Parkinson's disease. Nat Neurosci 3(12):1301–1306. https://doi.org/10.1038/81834
Cappelletti G, Pedrotti B, Maggioni MG, Maci R (2001) Microtubule assembly is directly affected by MPP(+)in vitro. Cell Biol Int 25(10):981–984. https://doi.org/10.1006/cbir.2001.0772
Cartelli D, Casagrande F, Busceti CL, Bucci D, Molinaro G, Traficante A, Passarella D, Giavini E et al (2013) Microtubule alterations occur early in experimental parkinsonism and the microtubule stabilizer epothilone D is neuroprotective. Sci Rep 3:1837. https://doi.org/10.1038/srep01837
Burke D, Gasdaska P, Hartwell L (1989) Dominant effects of tubulin overexpression in Saccharomyces cerevisiae. Mol Cell Biol 9(3):1049–1059
Weinstein B, Solomon F (1990) Phenotypic consequences of tubulin overproduction in Saccharomyces cerevisiae: differences between alpha-tubulin and beta-tubulin. Mol Cell Biol 10(10):5295–5304
Hirsch EC, Hunot S (2009) Neuroinflammation in Parkinson's disease: a target for neuroprotection? Lancet Neurol 8(4):382–397. https://doi.org/10.1016/S1474-4422(09)70062-6
McGeer PL, McGeer EG (2008) The alpha-synuclein burden hypothesis of Parkinson disease and its relationship to Alzheimer disease. Exp Neurol 212(2):235–238. https://doi.org/10.1016/j.expneurol.2008.04.008
Acknowledgments
The authors would like to thank Mr. Soraci Francesco for secretarial and administrative assistance, Medici Maria Antonietta for excellent technical assistance, and Miss Malvagni Valentina for editorial support with the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure Statement
Salvatore Cuzzocrea is co-inventor on patent WO2013121449 A8 (Epitech Group Srl) which deals with methods and compositions for the modulation of amidases capable of hydrolysing N-acylethanolamines employable in the treatment of inflammatory diseases. This invention is wholly unrelated to the present study. Moreover, Prof. Cuzzocrea is also, with Epitech Group, a co-inventor on the following patent: EP 2821083; MI2014 A001495; 102015000067344 that are however unrelated to the study.
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Crupi, R., Impellizzeri, D., Cordaro, M. et al. N-palmitoylethanolamide Prevents Parkinsonian Phenotypes in Aged Mice. Mol Neurobiol 55, 8455–8472 (2018). https://doi.org/10.1007/s12035-018-0959-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-018-0959-2