Abstract
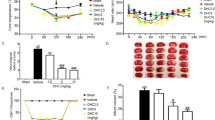
N-acetylcysteine (NAC), a precursor of glutathione that reduces reperfusion-induced injury, has been shown protection when it was administered pre-ischemia. However, less is known about the effect when it was given post-ischemia and there is no positive result associated with anti-oxidant in clinical trials. This study investigated the neuro- and vaso-protection of post-ischemia NAC administration as well as combining NAC with normobaric hyperoxia (NBO). Male Sprague–Dawley rats were exposed to NBO or normoxia during 2-h occlusion of the middle cerebral artery, followed by 48-h reperfusion. NAC or vehicle was intraperitoneally administered to rats immediately before reperfusion onset. NAC and NBO treatments produced 1.2 and 30 % reduction of infarction volume, respectively, and combination treatment showed greater reduction (59.8 %) as well as more decrease of hemispheric swelling volume. Of note, combination therapy showed improved neurological assessment and motor function which were sustained for 7 days after reperfusion. We also determined that the combination therapy showed greater inhibitory effects on tight junction protein degradation accompanied by Evan’s blue extravasation, hypoxia-inducible factor-1α (HIF-1α) and vascular endothelial growth factor (VEGF) induction, and poly ADP-ribose polymerase (PARP)-1 activation in ischemic brain tissue. Our results showed that although post-ischemia NAC administration had limited protection, combination treatment of NAC plus NBO effectively prevented blood–brain barrier (BBB) damage and significantly improved the outcome of brain injury, providing new evidence to support the concept that “cocktail” treatment targeting different stages provides better neuro- and vaso-protection than current individual treatment that has all failed in their clinical trials.






Similar content being viewed by others
Abbreviations
- BBB:
-
Blood–brain barrier
- EB:
-
Evan’s blue
- Et:
-
Ethidium
- Het:
-
Hydroethidine
- HIF-1α:
-
Hypoxia-inducible factor-1α
- I:
-
Ischemic hemisphere
- MCAO:
-
Middle cerebral artery occlusion
- NAC:
-
N-acetylcysteine
- NBO:
-
Normobaric hyperoxia
- NI:
-
Nonischemic hemisphere
- PAR:
-
Poly ADP-ribose
- PARP:
-
Poly ADP-ribose polymerase
- PFA:
-
Paraformaldehyde
- TTC:
-
Triphenyl-2,3,4-tetrazolium-chloride
- VEGF:
-
Vascular endothelial growth factor
References
Ramos-Cabrer P, Campos F, Sobrino T, Castillo J (2011) Targeting the ischemic penumbra. Stroke 42(1 Suppl):S7–S11
Dirnagl U, Iadecola C, Moskowitz MA (1999) Pathobiology of ischaemic stroke: an integrated view. Trends Neurosci 22(9):391–397
Schaller B, Graf R (2004) Cerebral ischemia and reperfusion: the pathophysiologic concept as a basis for clinical therapy. J Cereb Blood Flow Metab 24(4):351–371
Reza Noorian A, Nogueira R, Gupta R (2011) Neuroprotection in acute ischemic stroke. J Neurosurg Sci 55(2):127–138
Zhang L, Zhang ZG, Chopp M (2012) The neurovascular unit and combination treatment strategies for stroke. Trends Pharmacol Sci 33(8):415–422
Pan J, Konstas AA, Bateman B, Ortolano GA, Pile-Spellman J (2007) Reperfusion injury following cerebral ischemia: pathophysiology, MR imaging, and potential therapies. Neuroradiology 49(2):93–102
Khan M, Sekhon B, Jatana M, Giri S, Gilg AG, Sekhon C, Singh I, Singh AK (2004) Administration of N-acetylcysteine after focal cerebral ischemia protects brain and reduces inflammation in a rat model of experimental stroke. J Neurosci Res 76(4):519–527
Sekhon B, Sekhon C, Khan M, Patel SJ, Singh I, Singh AK (2003) N-Acetyl cysteine protects against injury in a rat model of focal cerebral ischemia. Brain Res 971(1):1–8
Lancet T (2006) Neuroprotection: the end of an era? Lancet 368(9547):1548
O’Collins VE, Macleod MR, Donnan GA, Howells DW (2012) Evaluation of combination therapy in animal models of cerebral ischemia. J Cereb Blood Flow Metab 32(4):585–597
Jin X, Liu J, Liu W (2014) Early ischemic blood brain barrier damage: a potential indicator for hemorrhagic transformation following tissue plasminogen activator (tPA) thrombolysis? Curr Neurovasc Res 11(3):254–262
Jin X, Liu J, Liu KJ, Rosenberg GA, Yang Y, Liu W (2013) Normobaric hyperoxia combined with minocycline provides greater neuroprotection than either alone in transient focal cerebral ischemia. Exp Neurol 240:9–16
Liang J, Qi Z, Liu W, Wang P, Shi W, Dong W, Ji X, Luo Y et al (2015) Normobaric hyperoxia slows blood–brain barrier damage and expands the therapeutic time window for tissue-type plasminogen activator treatment in cerebral ischemia. Stroke 46(5):1344–1351
Singhal AB, Wang X, Sumii T, Mori T, Lo EH (2002) Effects of normobaric hyperoxia in a rat model of focal cerebral ischemia-reperfusion. J Cereb Blood Flow Metab 22(7):861–868
Henninger N, Fisher M (2006) Normobaric hyperoxia—a promising approach to expand the time window for acute stroke treatment. Cerebrovasc Dis 21(1–2):134–136
Nonaka Y, Koumura A, Hyakkoku K, Shimazawa M, Yoshimura S, Iwama T, Hara H (2009) Combination treatment with normobaric hyperoxia and cilostazol protects mice against focal cerebral ischemia-induced neuronal damage better than each treatment alone. J Pharmacol Exp Ther 330(1):13–22
Nonaka Y, Shimazawa M, Yoshimura S, Iwama T, Hara H (2008) Combination effects of normobaric hyperoxia and edaravone on focal cerebral ischemia-induced neuronal damage in mice. Neurosci Lett 441(2):224–228
Beker MC, Caglayan AB, Kelestemur T, Caglayan B, Yalcin E, Yulug B, Kilic U, Hermann DM et al (2015) Effects of normobaric oxygen and melatonin on reperfusion injury: role of cerebral microcirculation., Oncotarget
Geng X, Fu P, Ji X, Peng C, Fredrickson V, Sy C, Meng R, Ling F et al (2013) Synergetic neuroprotection of normobaric oxygenation and ethanol in ischemic stroke through improved oxidative mechanism. Stroke 44(5):1418–1425
Geng X, Parmar S, Li X, Peng C, Ji X, Chakraborty T, Li WA, Du H et al (2013) Reduced apoptosis by combining normobaric oxygenation with ethanol in transient ischemic stroke. Brain Res 1531:17–24
Liu S, Liu W, Ding W, Miyake M, Rosenberg GA, Liu KJ (2006) Electron paramagnetic resonance-guided normobaric hyperoxia treatment protects the brain by maintaining penumbral oxygenation in a rat model of transient focal cerebral ischemia. J Cereb Blood Flow Metab 26(10):1274–1284
Zhao Y, Pan R, Li S, Luo Y, Yan F, Yin J, Qi Z, Yan Y et al (2014) Chelating intracellularly accumulated zinc decreased ischemic brain injury through reducing neuronal apoptotic death. Stroke 45(4):1139–1147
Zhao BQ, Wang S, Kim HY, Storrie H, Rosen BR, Mooney DJ, Wang X, Lo EH (2006) Role of matrix metalloproteinases in delayed cortical responses after stroke. Nat Med 12(4):441–445
Liu J, Jin X, Liu KJ, Liu W (2012) Matrix metalloproteinase-2-mediated occludin degradation and caveolin-1-mediated claudin-5 redistribution contribute to blood–brain barrier damage in early ischemic stroke stage. J Neurosci 32(9):3044–3057
Semenza GL (2014) Oxygen sensing, hypoxia-inducible factors, and disease pathophysiology. Annu Rev Pathol 9:47–71
Yan J, Zhou B, Taheri S, Shi H (2011) Differential effects of HIF-1 inhibition by YC-1 on the overall outcome and blood–brain barrier damage in a rat model of ischemic stroke. PLoS One 6(11), e27798
Zhang Z, Yan J, Taheri S, Liu KJ, Shi H (2014) Hypoxia-inducible factor 1 contributes to N-acetylcysteine’s protection in stroke. Free Radic Biol Med 68:8–21
Liu W, Hendren J, Qin XJ, Shen J, Liu KJ (2009) Normobaric hyperoxia attenuates early blood–brain barrier disruption by inhibiting MMP-9-mediated occludin degradation in focal cerebral ischemia. J Neurochem 108(3):811–820
Singhal AB, Benner T, Roccatagliata L, Koroshetz WJ, Schaefer PW, Lo EH, Buonanno FS, Gonzalez RG et al (2005) A pilot study of normobaric oxygen therapy in acute ischemic stroke. Stroke 36(4):797–802
Jin X, Liu J, Yang Y, Liu KJ, Liu W (2012) Spatiotemporal evolution of blood brain barrier damage and tissue infarction within the first 3h after ischemia onset. Neurobiol Dis 48(3):309–316
Saver JL (2006) Time is brain—quantified. Stroke 37(1):263–266
Yang Y, Candelario-Jalil E, Thompson JF, Cuadrado E, Estrada EY, Rosell A, Montaner J, Rosenberg GA (2010) Increased intranuclear matrix metalloproteinase activity in neurons interferes with oxidative DNA repair in focal cerebral ischemia. J Neurochem 112(1):134–149
van Wijk SJ, Hageman GJ (2005) Poly(ADP-ribose) polymerase-1 mediated caspase-independent cell death after ischemia/reperfusion. Free Radic Biol Med 39(1):81–90
Chen C, Hu Q, Yan J, Yang X, Shi X, Lei J, Chen L, Huang H et al (2009) Early inhibition of HIF-1alpha with small interfering RNA reduces ischemic-reperfused brain injury in rats. Neurobiol Dis 33(3):509–517
Chen C, Ostrowski RP, Zhou C, Tang J, Zhang JH (2010) Suppression of hypoxia-inducible factor-1alpha and its downstream genes reduces acute hyperglycemia-enhanced hemorrhagic transformation in a rat model of cerebral ischemia. J Neurosci Res 88(9):2046–2055
Semenza GL (2000) HIF-1: mediator of physiological and pathophysiological responses to hypoxia. J Appl Physiol (1985) 88(4):1474–1480
Argaw AT, Gurfein BT, Zhang Y, Zameer A, John GR (2009) VEGF-mediated disruption of endothelial CLN-5 promotes blood–brain barrier breakdown. Proc Natl Acad Sci U S A 106(6):1977–1982
Yeh WL, Lu DY, Lin CJ, Liou HC, Fu WM (2007) Inhibition of hypoxia-induced increase of blood–brain barrier permeability by YC-1 through the antagonism of HIF-1alpha accumulation and VEGF expression. Mol Pharmacol 72(2):440–449
Liu J, Weaver J, Jin X, Zhang Y, Xu J, Liu KJ, Li W, Liu W (2015) Nitric oxide interacts with caveolin-1 to facilitate autophagy-lysosome-mediated claudin-5 degradation in oxygen-glucose deprivation-treated endothelial cells., Mol Neurobiol
Wang X, Liu Y, Sun Y, Liu W, Jin X (2016) Blood brain barrier breakdown was found in non-infarcted area after 2-h MCAO. J Neurol Sci 363:63–68
Wang X, Tsuji K, Lee SR, Ning M, Furie KL, Buchan AM, Lo EH (2004) Mechanisms of hemorrhagic transformation after tissue plasminogen activator reperfusion therapy for ischemic stroke. Stroke 35(11 Suppl 1):2726–2730
Agarwal R, Shukla GS (1999) Potential role of cerebral glutathione in the maintenance of blood–brain barrier integrity in rat. Neurochem Res 24(12):1507–1514
Li W, Maloney RE, Circu ML, Alexander JS, Aw TY (2013) Acute carbonyl stress induces occludin glycation and brain microvascular endothelial barrier dysfunction: role for glutathione-dependent metabolism of methylglyoxal. Free Radic Biol Med 54:51–61
Beauchesne E, Desjardins P, Butterworth RF, Hazell AS (2010) Up-regulation of caveolin-1 and blood–brain barrier breakdown are attenuated by N-acetylcysteine in thiamine deficiency. Neurochem Int 57(7):830–837
Sceneay J, Liu MC, Chen A, Wong CS, Bowtell DD, Moller A (2013) The antioxidant N-acetylcysteine prevents HIF-1 stabilization under hypoxia in vitro but does not affect tumorigenesis in multiple breast cancer models in vivo. PLoS One 8(6), e66388
Acknowledgments
A patent application on therapeutic effects of combination NBO with NAC on ischemic stroke has been filed to China Intellectual Property Office. We thank the financial support from the Soochow University Research starting funds, Ministry of Education of China, Shenzhen Science and Technology Innovation Commission, and Priority Academic Program Development of Jiangsu Higher Education Institutions of China.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Funding
This work was supported by Soochow University Research starting funds (Q 421500113), by Fund of Ministry of Education of China (K521507713), and by Shenzhen Science and Technology Innovation Commission (CXZZ20130516152706040 and ZDSY20140509173142601). This work was supported by A Project by the Priority Academic Program Development of Jiangsu Higher Education Institutions of China.
Additional information
Yushan Liu and Wen-Cao Liu contributed equally to this work.
Rights and permissions
About this article
Cite this article
Liu, Y., Liu, WC., Sun, Y. et al. Normobaric Hyperoxia Extends Neuro- and Vaso-Protection of N-Acetylcysteine in Transient Focal Ischemia. Mol Neurobiol 54, 3418–3427 (2017). https://doi.org/10.1007/s12035-016-9932-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-016-9932-0