Abstract
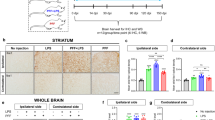
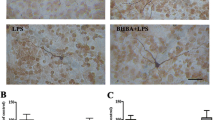
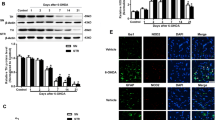
Microglia-associated inflammatory processes have been strongly implicated in the development and progression of Parkinson’s disease (PD). Specifically, microglia are activated in response to lipopolysaccharide (LPS) and become chronic source of cytokines and reactive oxygen species (ROS) production. Nicotinamide adenine dinucleotide phosphate (NADPH) oxidase complex is responsible for extracellular as well as intracellular production of ROS by microglia and its expression is upregulated in PD. Therefore, targeting NADPH oxidase complex activation using an NADPH oxidase inhibitor, i.e., apocyanin seems to be an effective approach. The aim of present study was to investigate the neuroprotective effects of apocyanin in a LPS-induced PD model. LPS (5 μg) was injected intranigral and apocyanin was administered daily at a dose of 10 mg/kg b.wt (i.p.) during the experiment. LPS when injected into the substantia nigra (SN) reproduced the characteristic hallmark features of PD in rats. It elicited an inflammatory response characterized by glial cell activation (Iba-1, GFAP). Furthermore, LPS upregulated the gene expression of nuclear factor-κB (NFκB), iNOS, and gp91PHOX and resulted in an elevated total ROS production as well as NADPH oxidase activity. Subsequently, this resulted in dopaminergic loss as depicted by decreased tyrosine hydroxylase (TH) expression with substantial loss in neurotransmitter dopamine and its metabolites, whereas treatment with apocyanin significantly reduced the number of glial fibrillary acidic protein (GFAP) and Iba-1-positive cells in LPS-treated animals. It also mitigated microglial activation-induced inflammatory response and elevation in NADPH oxidase activity, thus reducing the extracellular as well as intracellular ROS production. The present study indicated that targeting NADPH oxidase can inhibit microglial activation and reduce a broad spectrum of toxic factors generation (i.e., cytokines, ROS, and reactive nitrogen species [RNS]), thus offering a hope in halting the progression of PD.







Similar content being viewed by others
Abbreviations
- AD:
-
Alzheimer’s disease
- ALS:
-
Amyotrophic lateral sclerosis
- DA:
-
Dopamine
- DCFH-DA:
-
2′-7′-Dichlorodihydrofluorescein diacetate
- GFAP:
-
Glial fibrillary acidic protein
- IL-1β:
-
Interleukin-1β
- LPO:
-
Lipid peroxidation
- LPS:
-
Lipopolysaccharide
- MDA:
-
Malondialdehyde
- NADPH oxidase:
-
Nicotinamide adenine dinucleotide phosphate-oxidase
- NFκB:
-
Nuclear factor-κB
- NO:
-
Nitric oxide
- PBS:
-
Phosphate-buffered saline
- PD:
-
Parkinson’s disease
- PHOX:
-
Phagocytic oxidase
- PMF:
-
Post-mitochondrial fraction
- PMS:
-
Post mitochondrial supernatant
- RNS:
-
Reactive nitrogen species
- ROS:
-
Reactive oxygen species
- SN:
-
Substantia nigra
- SOD:
-
Superoxide dismutase
- TH:
-
Tyrosine hydroxylase
- TNF-α:
-
Tumor necrosis factor-alpha
References
BlocK ML, Hong JS (2005) Microglia and inflammation-mediated neurodegeneration: multiple triggers with a common mechanism. Prog Neurobiol 76(2):77–98
McGeer, McGeer (2007) NSAIDs and Alzheimer disease: epidemiological, animal model and clinical studies. Neurobiol Aging 28(5):639–647
Block ML, Zecca Hong JS (2007) Microglia and inflammation-mediated neurodegeneration multiple triggers with a common mechanism. Prog Neurobiol 76:77–98
McGuire SO, Ling ZD, Lipton JW, Sortwell CE, Collier TJ, Carvey PM (2001) Tumor necrosis factor alpha is toxic to embryonic mesencephalic dopamine neurons. Exp Neurol 169:219–230
Lawson LJ, Perry VH, Dri P, Gordon S (1990) Heterogeneity in the distribution and morphology of microglia in the normal adult mouse. Neuroscience 39:151–170
Tansey MG, Goldberg MS (2010) Neuroinflammation in Parkinson’s disease: its role in neuronal death and implications for therapeutic intervention. Neurobiol Dis 37:510–518
Tsang AHK, Chung KKK (2009) Oxidative and nitrosative stress in Parkinson’s disease. Biochim Biophys Acta 1792(7):643–650
McGeer PL, Itagaki S, Boyes BE, McGeer EG (1988) Reactive microglia are positive for HLA-DR in the substantia nigra of Parkinson’s and Alzheimer’s disease brains. Neurology 38(8):1285–1291
McGeer PL, McGeer EG (2008) Glial reactions in Parkinson’s disease. Mov Disord 23(4):474–483
Hirsch EC, Hunot S (2009) Neuroinflammation in Parkinson’s disease: a target for neuroprotection? Lancet Neurol 8(4):382–397
Chung ES, Boka E, Chung YC, Baika HH, Jina BK (2012) Cannabinoids prevent lipopolysaccharide-induced neurodegeneration in the rat substantia nigra in vivo through inhibition of microglial activation and NADPH oxidase. Brain Res. doi:10.1016/j.brainres.2012.02.058
Miller RL et al (2009) Oxidative and inflammatory pathways in Parkinson’s disease. Neurochem Res 34:55–65
Kavya R et al (2006) Nitric oxide synthase regulation and diversity: implications in Parkinson’s disease. Nitric Oxide 15:280–294
Wu DC et al (2003) NADPH oxidase mediates oxidative stress in the 1-methyl-4-phenyl-1,2,3,6 tetrahydropyridine model of Parkinson’s disease. Proc Natl Acad Sci U S A 100:6145–6150
Gao HM et al (2003) Critical role of microglial NADPH oxidase-derived free radicals in the in vitro MPTP model of Parkinson’s disease. FASEB J 17:1954–1956
Koppula S et al (2012) Reactive oxygen species and inhibitors of inflammatory enzymes, NADPH oxidase, and iNOS in experimental models of Parkinson’s disease. Mediat Inflamm. doi:10.1155/2012/823902
Luchtefeld R, Luo R, Stine K, Alt ML, Chernovitz PA, Smith RE (2008) Dose formulation and analysis of diapocynin. J Agric Food Chem 56:301–306
Ximenes VF, Kanegae MPP, Rissato SR, Galhiane MS (2007) The oxidation of apocynin catalyzed by myeloperoxidase: proposal for NADPH oxidase inhibition. Arch Biochem Biophys 457:134–141
Barbieri SS, Cavalca V, Eligini S, Brambilla M, Caiani A, Tremoli E, Colli S (2004) Apocyanin prevents cyclooxygenase 2 expression in human monocytes through NADPH oxidase and glutathione redox-dependent mechanisms. Free Radic Biol Med 37:156–165
Kanegae MPP, da Fonseca LM et al (2007) The reactivity of ortho-methoxy-substituted catechol radical. Biochem Pharmacol 74:457–464
Zhang W, Wang T, Pei Z et al (2005) Aggregated α-synuclein activates microglia a process leading to disease progression in Parkinson’s disease. FASEB J 19(6):533–542
Wang Q, Tompkins KD, Simonyi A, Korthuis RJ, Sun AY, Sun GY (2006) Apocynin protects against global cerebral ischemia-reperfusion-induced oxidative stress and injury in the gerbil hippocampus. Brain Res 1090(1):182–189
Bravo WLT, Jadhav V, Titova E, Zhang JH, Tang J (2007) NADPH oxidase inhibition improves neurological outcomes in surgically-induced brain injury. Neurosci Lett 414(3):228–232
Lull ME, Levesque S, Surace MJ, Block ML (2011) Chronic apocynin treatment attenuates beta amyloid plaque size and microglial number in hAPP(751) SL mice. PLoS One 6(5):e20153
Tang XN, Cairns B, Cairns N, Yenari MA (2008) Apocynin improves outcome in experimental stroke with a narrow dose range. Neuroscience 154(2):556–562
Titova E, Ostrowski RP, Sowers LC, Zhang JH, Tang J (2007) Effects of apocynin and ethanol on intracerebral haemorrhage-induced brain injury in rats. Clin Exp Pharmacol Physiol 34(9):845–850
Church W (2005) Column chromatography analysis of brain tissue: an advanced laboratory exercise for neuroscience majors. J Undergrad Neurosci Educ 3:A36–A41
Kalonia H, Mishra J, Kumar A (2012) Targeting neuro-inflammatory cytokines and oxidative stress by minocycline attenuates quinolinic-acid-induced Huntington’s disease-like symptoms in rats. Neurotox Res 22:310–320
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Follin’s phenol reagent. J Biol Chem 193:265–275
Wills ED (1966) Mechanism of lipid peroxide formation in animal tissue. Biochem J 99:667
Raddassi K (1993) Role of calcium in the activation of mouse peritoneal macrophages: induction of NO synthase by calcium ionophores and thapsigargin. Cell Immunol 153:443–455
Best TM (1999) Free radical activity, antioxidant enzyme, and glutathione changes with muscle stretch injury in rabbits. J Appl Physiol 87:74–82
Humanson GL (1961) Basic procedures—animal tissue technique. Animal tissue techniques. W. H. Freeman & Co., San Francisco, pp 130–132, Part-1
Liu M, Bing G (2011) Lipopolysaccharide animal models for Parkinson’s disease. Parkinsons Dis 327089
Qin L et al (2004) NADPH oxidase mediates lipopolysaccharide-induced neurotoxicity and proinflammatory gene expression in activated microglia. J Biol Chem 279:1415–1421
Qin et al (2007) Systemic LPS causes chronic neuroinflammation and progressive neurodegeneration. Glia 55(5):453–462
Block ML et al (2007) Microglia-mediated neurotoxicity: uncovering the molecular mechanisms. Nat Rev Neurosci 8:57–69
Koutsilieri E et al (2002) Free radicals in Parkinson’s disease. J Neurol 249(2):1–5
Choi SH et al (2005) Thrombin-induced oxidative stress contributes to the death of hippocampal neurons in vivo: role of microglial NADPH oxidase. J Neurosci 25:4082–4090
Cross AR, Segal AW (2004) The NADPH oxidase of professional phagocytes—prototype of the NOX electron transport chain systems. Biochim Biophys Acta 1657:1–22
Hernandez-Romero MC et al (2008) Simvastatin prevents the inflammatory process and the dopaminergic degeneration induced by the intranigral injection of lipopolysaccharide. J Neurochem 105:445–459
Ghosh A, Roy A, Liu X, Kordower JH, Mufson EJ, Hartley DM, Ghosh S, Mosley RL et al (2007) Selective inhibition of NF-kappaB activation prevents dopaminergic neuronal loss in a mouse model of Parkinson’s disease. Proc Natl Acad Sci U S A 104:18754–18759
Chung YC et al (2010) The role of neuroinflammation on the pathogenesis of Parkinson’s disease. BMB Rep 43:225–232
Allan S, Rothwell N (2001) Cytokines and acute neurodegeneration. Nat Rev Neurosci 2:734–744
Rothwell NJ, Luheshi GN (2000) Interleukin 1 in the brain: biology, pathology and therapeutic target. Trends Neurosci 23:618–625
Choi SH, Aid S, Kim HW, Jackson SH, Bosetti F (2012) Inhibition of NADPH oxidase promotes alternative and anti-inflammatory microglial activation during neuroinflammation. J Neurochem 120:292–301
Philippens IHCHM, Wubben JA, Finsen B, Hart BA (2013) Oral treatment with NADPH antagonist apocyanin mitigates clinical and pathological feature of parkinsonism in the MPTP-marmoset model. J Neuroimmune Pharmacol 8:715–726
Garman RH (2011) Histology of the central nervous system. Toxicol Pathol 39:22–35
Acknowledgments
The study was carried out with the funds provided by Indian Council of Medical Research (ICMR) India.
Conflict of Interest
The authors do not have any competing interests in the manuscript.
Compliance with Ethical Standards
The authors read and have abided by the statement of the ethical standards for manuscripts submitted to this journal.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sharma, N., Nehru, B. Apocyanin, a Microglial NADPH Oxidase Inhibitor Prevents Dopaminergic Neuronal Degeneration in Lipopolysaccharide-Induced Parkinson’s Disease Model. Mol Neurobiol 53, 3326–3337 (2016). https://doi.org/10.1007/s12035-015-9267-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-015-9267-2