Abstract
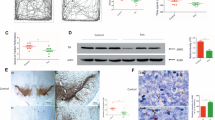
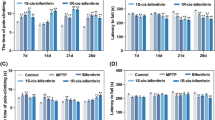
Fenpropathrin is one of the widely used pyrethroids in agriculture and household and also reported to have neurotoxic effects in rodent models. In our Parkinson’s disease (PD) clinic, there was a unique patient with a history of daily exposure to fenpropathrin for 6 months prior to developing Parkinsonian symptoms progressively. Since whether fenpropathrin is related to any dopaminergic degeneration was unknown, we aimed in this study to evaluate the neurotoxic effects of fenpropathrin on the dopaminergic system and associated mechanisms in vitro and in vivo. In cultured SH-SY5Y cells, fenpropathrin caused cell death, reactive oxygen species generation, Lewy body-associated proteins aggregation, and Lewy body-like intracytoplasmic inclusions formation. In rodent animals, two different injections of fenpropathrin were used for administrations, intraperitoneal (i.p), or stereotaxical (ST). The rats exhibited lower number of pokes 60 days after first i.p injection, while the rats in ST group showed a significant upregulation of apomorphine-evoked rotations 60 days after first injection. Dcreased tyrosine hydroxylase (TH) and vesicular monoamine transporter 2 (VMAT2) immunoreactivity, while increased dopamine transporter (DAT) immunoreactivity were observed in rats of either i.p or ST group 60 days after the last exposure to fenpropathrin. However, the number of TH-positive cells in the substantia nigra was more reduced 120 days after the first i.p injection than those of 60 days. Our data demonstrated that exposure to fenpropathrin could mimic the pathologic and pathogenetic features of PD especially in late onset cases. These results imply fenpropathrin as a DA neurotoxin and a possible environmental risk factor for PD.









Similar content being viewed by others
Abbreviations
- Fen:
-
Fenpropathrin
- PD:
-
Parkinson’s disease
- i.p:
-
Intraperitoneal injection
- ST:
-
Stereotaxical injection
- DAT:
-
Dopamine transporter
- VMAT2:
-
Vesicular monoamine transporter 2
- DA:
-
Dopamine
References
Lees AJ, Hardy J, Revesz T (2009) Parkinson’s disease. Lancet 373(9680):2055–2066. doi:10.1016/S0140-6736(09)60492-X
van der Mark M, Brouwer M, Kromhout H, Nijssen P, Huss A, Vermeulen R (2012) Is pesticide use related to Parkinson disease? Some clues to heterogeneity in study results. Environ Health Perspect 120(3):340–347. doi:10.1289/ehp.1103881
Casida JE (1980) Pyrethrum flowers and pyrethroid insecticides. Environ Health Perspect 34:189–202
Amweg EL, Weston DP, Ureda NM (2005) Use and toxicity of pyrethroid pesticides in the Central Valley, California, USA. Environ Toxicol Chem / SETAC 24(4):966–972
Wolansky MJ, Harrill JA (2008) Neurobehavioral toxicology of pyrethroid insecticides in adult animals: a critical review. Neurotoxicol Teratol 30(2):55–78. doi:10.1016/j.ntt.2007.10.005
Liu GP, Shi N (2006) The inhibitory effects of deltamethrin on dopamine biosynthesis in rat PC12 cells. Toxicol Lett 161(3):195–199. doi:10.1016/j.toxlet.2005.09.011
Gillette JS, Bloomquist JR (2003) Differential up-regulation of striatal dopamine transporter and alpha-synuclein by the pyrethroid insecticide permethrin. Toxicol Appl Pharmacol 192(3):287–293
Elwan MA, Richardson JR, Guillot TS, Caudle WM, Miller GW (2006) Pyrethroid pesticide-induced alterations in dopamine transporter function. Toxicol Appl Pharmacol 211(3):188–197. doi:10.1016/j.taap.2005.06.003
Singh AK, Tiwari MN, Upadhyay G, Patel DK, Singh D, Prakash O, Singh MP (2012) Long term exposure to cypermethrin induces nigrostriatal dopaminergic neurodegeneration in adult rats: postnatal exposure enhances the susceptibility during adulthood. Neurobiol Aging 33(2):404–415. doi:10.1016/j.neurobiolaging.2010.02.018
Weiner ML, Nemec M, Sheets L, Sargent D, Breckenridge C (2009) Comparative functional observational battery study of twelve commercial pyrethroid insecticides in male rats following acute oral exposure. Neurotoxicology 30(Suppl 1):S1–S16. doi:10.1016/j.neuro.2009.08.014
Bradberry SM, Cage SA, Proudfoot AT, Vale JA (2005) Poisoning due to pyrethroids. Toxicol Rev 24(2):93–106
Al-Makkawy HK, Madbouly MD (1999) Persistence and accumulation of some organic insecticides in Nile water and fish. Resour Conserv Recycl 27(1–2):105–115. doi:10.1016/s0921-3449(98)00090-1
Zielinska E, Kocki T, Saran T, Borbely S, Kuc D, Vilagi I, Urbanska EM, Turski WA (2005) Effect of pesticides on kynurenic acid production in rat brain slices. Ann Agric Environ Med: AAEM 12(2):177–179
Ogawa T, Matson WR, Beal MF, Myers RH, Bird ED, Milbury P, Saso S (1992) Kynurenine pathway abnormalities in Parkinson’s disease. Neurology 42(9):1702–1706
Kanawi E, Budd R, Tjeerdema RS (2013) Environmental fate and ecotoxicology of fenpropathrin. Rev Environ Contam Toxicol 225:77–93. doi:10.1007/978-1-4614-6470-9_3
Zhang Z, Cao X, Xiong N, Wang H, Huang J, Sun S, Liang Z, Wang T (2010) DNA polymerase-beta is required for 1-methyl-4-phenylpyridinium-induced apoptotic death in neurons. Apoptosis: Int J Programmed Cell Death 15(1):105–115. doi:10.1007/s10495-009-0425-8
Xiong N, Huang J, Chen C, Zhao Y, Zhang Z, Jia M, Hou L, Yang H, Cao X, Liang Z, Zhang Y, Sun S, Lin Z, Wang T (2012) Dl-3-n-butylphthalide, a natural antioxidant, protects dopamine neurons in rotenone models for Parkinson’s disease. Neurobiol Aging 33(8):1777–1791. doi:10.1016/j.neurobiolaging.2011.03.007
Salvioli S, Ardizzoni A, Franceschi C, Cossarizza A (1997) JC-1, but not DiOC6(3) or rhodamine 123, is a reliable fluorescent probe to assess delta psi changes in intact cells: implications for studies on mitochondrial functionality during apoptosis. FEBS Lett 411(1):77–82
Huang J, Hao L, Xiong N, Cao X, Liang Z, Sun S, Wang T (2009) Involvement of glyceraldehyde-3-phosphate dehydrogenase in rotenone-induced cell apoptosis: relevance to protein misfolding and aggregation. Brain Res 1279:1–8. doi:10.1016/j.brainres.2009.05.011
Xiong N, Huang J, Zhang Z, Xiong J, Liu X, Jia M, Wang F, Chen C, Cao X, Liang Z, Sun S, Lin Z, Wang T (2009) Stereotaxical infusion of rotenone: a reliable rodent model for Parkinson’s disease. PLoS One 4(11):e7878. doi:10.1371/journal.pone.0007878
Deumens R, Blokland A, Prickaerts J (2002) Modeling Parkinson’s disease in rats: an evaluation of 6-OHDA lesions of the nigrostriatal pathway. Exp Neurol 175(2):303–317. doi:10.1006/exnr.2002.7891
Sonia Angeline M, Chaterjee P, Anand K, Ambasta RK, Kumar P (2012) Rotenone-induced parkinsonism elicits behavioral impairments and differential expression of parkin, heat shock proteins and caspases in the rat. Neuroscience 220:291–301. doi:10.1016/j.neuroscience.2012.06.021
Singh S, Singh K, Patel DK, Singh C, Nath C, Singh VK, Singh RK, Singh MP (2009) The expression of CYP2D22, an ortholog of human CYP2D6, in mouse striatum and its modulation in 1-methyl 4-phenyl-1,2,3,6-tetrahydropyridine-induced Parkinson’s disease phenotype and nicotine-mediated neuroprotection. Rejuvenation Res 12(3):185–197. doi:10.1089/rej.2009.0850
Kuusisto E, Parkkinen L, Alafuzoff I (2003) Morphogenesis of Lewy bodies: dissimilar incorporation of alpha-synuclein, ubiquitin, and p62. J Neuropathol Exp Neurol 62(12):1241–1253
Olsson M, Nikkhah G, Bentlage C, Bjorklund A (1995) Forelimb akinesia in the rat Parkinson model: differential effects of dopamine agonists and nigral transplants as assessed by a new stepping test. J Neurosci: Off J Soc Neurosci 15(5 Pt 2):3863–3875
Xiong N, Jia M, Chen C, Xiong J, Zhang Z, Huang J, Hou L, Yang H, Cao X, Liang Z, Sun S, Lin Z, Wang T (2011) Potential autophagy enhancers attenuate rotenone-induced toxicity in SH-SY5Y. Neuroscience 199:292–302. doi:10.1016/j.neuroscience.2011.10.031
Watabe M, Nakaki T (2008) Mitochondrial complex I inhibitor rotenone inhibits and redistributes vesicular monoamine transporter 2 via nitration in human dopaminergic SH-SY5Y cells. Mol Pharmacol 74(4):933–940. doi:10.1124/mol.108.048546
Xie HR, Hu LS, Li GY (2010) SH-SY5Y human neuroblastoma cell line: in vitro cell model of dopaminergic neurons in Parkinson’s disease. Chin Med J 123(8):1086–1092
Jana S, Sinha M, Chanda D, Roy T, Banerjee K, Munshi S, Patro BS, Chakrabarti S (2011) Mitochondrial dysfunction mediated by quinone oxidation products of dopamine: implications in dopamine cytotoxicity and pathogenesis of Parkinson’s disease. Biochim Biophys Acta 1812(6):663–673. doi:10.1016/j.bbadis.2011.02.013
Hastings TG, Lewis DA, Zigmond MJ (1996) Role of oxidation in the neurotoxic effects of intrastriatal dopamine injections. Proc Natl Acad Sci U S A 93(5):1956–1961
Giray B, Gurbay A, Hincal F (2001) Cypermethrin-induced oxidative stress in rat brain and liver is prevented by vitamin E or allopurinol. Toxicol Lett 118(3):139–146
Abdou R, Sasaki K, Khalil W, Shah S, Murasawa Y, Shimoda M (2010) Effects of several pyrethroids on hepatic cytochrome P450 activities in rats. J Vet Med Sci / Jpn Soc Vet Sci 72(4):425–433
Gassner B, Wuthrich A, Scholtysik G, Solioz M (1997) The pyrethroids permethrin and cyhalothrin are potent inhibitors of the mitochondrial complex I. J Pharmacol Exp Ther 281(2):855–860
Gai WP, Yuan HX, Li XQ, Power JT, Blumbergs PC, Jensen PH (2000) In situ and in vitro study of colocalization and segregation of alpha-synuclein, ubiquitin, and lipids in Lewy bodies. Exp Neurol 166(2):324–333. doi:10.1006/exnr.2000.7527
Wakabayashi K, Tanji K, Odagiri S, Miki Y, Mori F, Takahashi H (2013) The Lewy body in Parkinson’s disease and related neurodegenerative disorders. Mol Neurobiol 47(2):495–508. doi:10.1007/s12035-012-8280-y
Patel S, Singh V, Kumar A, Gupta YK, Singh MP (2006) Status of antioxidant defense system and expression of toxicant responsive genes in striatum of maneb- and paraquat-induced Parkinson’s disease phenotype in mouse: mechanism of neurodegeneration. Brain Res 1081(1):9–18. doi:10.1016/j.brainres.2006.01.060
Xiong N, Long X, Xiong J, Jia M, Chen C, Huang J, Ghoorah D, Kong X, Lin Z, Wang T (2012) Mitochondrial complex I inhibitor rotenone-induced toxicity and its potential mechanisms in Parkinson’s disease models. Crit Rev Toxicol 42(7):613–632. doi:10.3109/10408444.2012.680431
Pardridge WM (2005) Tyrosine hydroxylase replacement in experimental Parkinson’s disease with transvascular gene therapy. NeuroRx: J Am Soc Exp Neurother 2(1):129–138. doi:10.1602/neurorx.2.1.129
Richardson JR, Caudle WM, Wang MZ, Dean ED, Pennell KD, Miller GW (2008) Developmental heptachlor exposure increases susceptibility of dopamine neurons to N-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)in a gender-specific manner. Neurotoxicology 29(5):855–863. doi:10.1016/j.neuro.2008.05.007
Rodriguez VM, Limon-Pacheco JH, Mendoza-Trejo MS, Gonzalez-Gallardo A, Hernandez-Plata I, Giordano M (2013) Repeated exposure to the herbicide atrazine alters locomotor activity and the nigrostriatal dopaminergic system of the albino rat. Neurotoxicology 34:82–94. doi:10.1016/j.neuro.2012.10.012
Khoshbouei H, Wang H, Lechleiter JD, Javitch JA, Galli A (2003) Amphetamine-induced dopamine efflux. A voltage-sensitive and intracellular Na+-dependent mechanism. J Biol Chem 278(14):12070–12077. doi:10.1074/jbc.M212815200
Yoon JY, Ahn SH, Oh H, Kim YS, Ryu SY, Ho WK, Lee SH (2004) A novel Na + channel agonist, dimethyl lithospermate B, slows Na + current inactivation and increases action potential duration in isolated rat ventricular myocytes. Br J Pharmacol 143(6):765–773. doi:10.1038/sj.bjp.0705969
Caudle WM, Richardson JR, Wang MZ, Taylor TN, Guillot TS, McCormack AL, Colebrooke RE, Di Monte DA, Emson PC, Miller GW (2007) Reduced vesicular storage of dopamine causes progressive nigrostriatal neurodegeneration. J Neurosci: Off J Soc Neurosci 27(30):8138–8148. doi:10.1523/JNEUROSCI. 0319-07.2007
Takahashi N, Miner LL, Sora I, Ujike H, Revay RS, Kostic V, Jackson-Lewis V, Przedborski S, Uhl GR (1997) VMAT2 knockout mice: heterozygotes display reduced amphetamine-conditioned reward, enhanced amphetamine locomotion, and enhanced MPTP toxicity. Proc Natl Acad Sci U S A 94(18):9938–9943
Tiwari MN, Singh AK, Ahmad I, Upadhyay G, Singh D, Patel DK, Singh C, Prakash O, Singh MP (2010) Effects of cypermethrin on monoamine transporters, xenobiotic metabolizing enzymes and lipid peroxidation in the rat nigrostriatal system. Free Radic Res 44(12):1416–1424. doi:10.3109/10715762.2010.512041
Acknowledgments
This work was supported by grants 30870866, 81071021, and 31171211 from the National Natural Science Foundation of China (to TW); grant 81200983 from the National Natural Science Foundation of China (to NX); grant 81100958 from the National Natural Science Foundation of China (to ZTZ); grant 81301082 from the National Natural Science Foundation of China (to JSH); grant 2012B09 from China Medical Foundation (to NX); and grant 0203201343 from Hubei Molecular Imaging Key Laboratory (to NX). The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Conflict of Interest
There are no actual or potential conflicts of interest.
Authors’ Contributions
JX, XZ, JH, CC, ZC, LL, JY, ZZ, NX, and TW contributed to the conception and design. JX, XZ, JH, and CC took care of the cell culture studies. JX, XZ, ZC, LL, and JY took care of the animal studies. JX, XZ, JH, CC, ZC, and NX analyzed and interoperated the data. JX, XZ, JH, NX, and TW coordinated all the experiments and helped to draft the manuscript. All authors read, revised, and approved the final manuscript.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Jing Xiong, Xiaowei Zhang, and Jinsha Huang equally contributed to the work.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Supplementary Fig. 1
(DOC 2278 kb)
Supplementary Fig. 2
(DOC 2020 kb)
Rights and permissions
About this article
Cite this article
Xiong, J., Zhang, X., Huang, J. et al. Fenpropathrin, a Widely Used Pesticide, Causes Dopaminergic Degeneration. Mol Neurobiol 53, 995–1008 (2016). https://doi.org/10.1007/s12035-014-9057-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-014-9057-2