Abstract
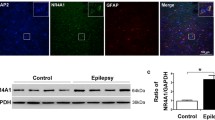
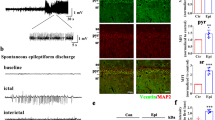
Abnormally synchronized synaptic transmission in the brain leads to epilepsy. Neuroligin-1 (NL1) is a synaptic cell adhesion molecule localized at excitatory synapses. NL1 modulates synaptic transmission and determines the properties of neuronal networks in the mammalian central nervous system. We showed that the expression of NL1 and its binding partner neurexin-1β was increased in temporal lobe epileptic foci in patients and lithium-pilocarpine-treated epileptic rats. We investigated electrophysiological and behavioral changes in epileptic rats after lentivirally mediated NL1 knockdown in the hippocampus to determine whether NL1 suppression prevented seizures and, if so, to explore the probable underlying mechanisms. Our behavioral studies revealed that NL1 knockdown in epileptic rats reduced seizure severity and increased seizure latency. Whole-cell patch-clamp recordings of CA1 pyramidal neurons in hippocampal slices from NL1 knockdown epileptic rats revealed a decrease in spontaneous action potential frequency and a decrease in miniature excitatory postsynaptic current (mEPSC) frequency but not amplitude. The amplitude of N-methyl-d-aspartate receptor (NMDAR)-dependent EPSCs was also selectively decreased. Notably, NL1 knockdown reduced total NMDAR1 expression and the surface/total ratio in the hippocampus of epileptic rats. Taken together, these data indicate that NL1 knockdown in epileptic rats may reduce the frequency and severity of seizures and suppress neuronal hyperexcitability via changes in postsynaptic NMDARs.





Similar content being viewed by others
References
Sander JW (2003) The epidemiology of epilepsy revisited. Curr Opin Neurol 16(2):165–170. doi:10.1097/01.wco.0000063766.15877.8e
Fisher RS, van Boas EW, Blume W, Elger C, Genton P, Lee P, Engel J Jr (2005) Epileptic seizures and epilepsy: definitions proposed by the International League Against Epilepsy (ILAE) and the International Bureau for Epilepsy (IBE). Epilepsia 46(4):470–472. doi:10.1111/j.0013-9580.2005.66104.x
Esclapez M, Hirsch JC, Ben-Ari Y, Bernard C (1999) Newly formed excitatory pathways provide a substrate for hyperexcitability in experimental temporal lobe epilepsy. J Comp Neurol 408(4):449–460. doi:10.1002/(SICI)1096-9861(19990614)408:4<449::AID-CNE1>3.0.CO;2-R
Dalva MB, McClelland AC, Kayser MS (2007) Cell adhesion molecules: signalling functions at the synapse. Nat Rev Neurosci 8(3):206–220. doi:10.1038/nrn2075
Krueger DD, Tuffy LP, Papadopoulos T, Brose N (2012) The role of neurexins and neuroligins in the formation, maturation, and function of vertebrate synapses. Curr Opin Neurobiol 22(3):412–422. doi:10.1016/j.conb.2012.02.012
Bottos A, Rissone A, Bussolino F, Arese M (2011) Neurexins and neuroligins: synapses look out of the nervous system. Cell Mol Life Sci. doi:10.1007/s00018-011-0664-z
Ichtchenko K, Hata Y, Nguyen T, Ullrich B, Missler M, Moomaw C, Sudhof TC (1995) Neuroligin 1: a splice site-specific ligand for beta-neurexins. Cell 81(3):435–443
Bolliger MF, Frei K, Winterhalter KH, Gloor SM (2001) Identification of a novel neuroligin in humans which binds to PSD-95 and has a widespread expression. Biochem J 356(Pt 2):581–588
Arac D, Boucard AA, Ozkan E, Strop P, Newell E, Sudhof TC, Brunger AT (2007) Structures of neuroligin-1 and the neuroligin-1/neurexin-1 beta complex reveal specific protein-protein and protein-Ca2+ interactions. Neuron 56(6):992–1003. doi:10.1016/j.neuron.2007.12.002
Chubykin AA, Atasoy D, Etherton MR, Brose N, Kavalali ET, Gibson JR, Sudhof TC (2007) Activity-dependent validation of excitatory versus inhibitory synapses by neuroligin-1 versus neuroligin-2. Neuron 54(6):919–931. doi:10.1016/j.neuron.2007.05.029
Levinson JN, Chery N, Huang K, Wong TP, Gerrow K, Kang R, Prange O, Wang YT, El-Husseini A (2005) Neuroligins mediate excitatory and inhibitory synapse formation: involvement of PSD-95 and neurexin-1beta in neuroligin-induced synaptic specificity. J Biol Chem 280(17):17312–17319. doi:10.1074/jbc.M413812200
Chih B, Engelman H, Scheiffele P (2005) Control of excitatory and inhibitory synapse formation by neuroligins. Science 307(5713):1324–1328. doi:10.1126/science.1107470
Wittenmayer N, Korber C, Liu H, Kremer T, Varoqueaux F, Chapman ER, Brose N, Kuner T, Dresbach T (2009) Postsynaptic Neuroligin1 regulates presynaptic maturation. Proc Natl Acad Sci U S A 106(32):13564–13569. doi:10.1073/pnas.0905819106
Stan A, Pielarski KN, Brigadski T, Wittenmayer N, Fedorchenko O, Gohla A, Lessmann V, Dresbach T, Gottmann K (2010) Essential cooperation of N-cadherin and neuroligin-1 in the transsynaptic control of vesicle accumulation. Proc Natl Acad Sci U S A 107(24):11116–11121. doi:10.1073/pnas.0914233107
Pan Y, Liu G, Fang M, Shen L, Wang L, Han Y, Shen D, Wang X (2010) Abnormal expression of netrin-G2 in temporal lobe epilepsy neurons in humans and a rat model. Exp Neurol 224(2):340–346. doi:10.1016/j.expneurol.2010.04.001
Fukata Y, Adesnik H, Iwanaga T, Bredt DS, Nicoll RA, Fukata M (2006) Epilepsy-related ligand/receptor complex LGI1 and ADAM22 regulate synaptic transmission. Science 313(5794):1792–1795. doi:10.1126/science.1129947
Bie B, Wu J, Yang H, Xu JJ, Brown DL, Naguib M (2014) Epigenetic suppression of neuroligin 1 underlies amyloid-induced memory deficiency. Nat Neurosci 17(2):223–231. doi:10.1038/nn.3618
Sudhof TC (2008) Neuroligins and neurexins link synaptic function to cognitive disease. Nature 455(7215):903–911. doi:10.1038/nature07456
Harrison V, Connell L, Hayesmoore J, McParland J, Pike MG, Blair E (2011) Compound heterozygous deletion of NRXN1 causing severe developmental delay with early onset epilepsy in two sisters. Am J Med Genet A 155A(11):2826–2831. doi:10.1002/ajmg.a.34255
Etherton MR, Tabuchi K, Sharma M, Ko J, Sudhof TC (2011) An autism-associated point mutation in the neuroligin cytoplasmic tail selectively impairs AMPA receptor-mediated synaptic transmission in hippocampus. EMBO J. doi:10.1038/emboj.2011.182
Ching MS, Shen Y, Tan WH, Jeste SS, Morrow EM, Chen X, Mukaddes NM, Yoo SY, Hanson E, Hundley R, Austin C, Becker RE, Berry GT, Driscoll K, Engle EC, Friedman S, Gusella JF, Hisama FM, Irons MB, Lafiosca T, LeClair E, Miller DT, Neessen M, Picker JD, Rappaport L, Rooney CM, Sarco DP, Stoler JM, Walsh CA, Wolff RR, Zhang T, Nasir RH, Wu BL (2010) Deletions of NRXN1 (neurexin-1) predispose to a wide spectrum of developmental disorders. Am J Med Genet B Neuropsychiatr Genet 153B(4):937–947. doi:10.1002/ajmg.b.31063
Brooks-Kayal A (2010) Epilepsy and autism spectrum disorders: are there common developmental mechanisms? Brain Dev 32(9):731–738. doi:10.1016/j.braindev.2010.04.010
Kim J, Jung SY, Lee YK, Park S, Choi JS, Lee CJ, Kim HS, Choi YB, Scheiffele P, Bailey CH, Kandel ER, Kim JH (2008) Neuroligin-1 is required for normal expression of LTP and associative fear memory in the amygdala of adult animals. Proc Natl Acad Sci U S A 105(26):9087–9092. doi:10.1073/pnas.0803448105
Woldbye DP, Angehagen M, Gotzsche CR, Elbrond-Bek H, Sorensen AT, Christiansen SH, Olesen MV, Nikitidou L, Hansen TV, Kanter-Schlifke I, Kokaia M (2010) Adeno-associated viral vector-induced overexpression of neuropeptide Y Y2 receptors in the hippocampus suppresses seizures. Brain 133(9):2778–2788. doi:10.1093/brain/awq219
Fang M, Xi ZQ, Wu Y, Wang XF (2011) A new hypothesis of drug refractory epilepsy: neural network hypothesis. Med Hypotheses 76(6):871–876. doi:10.1016/j.mehy.2011.02.039
Gjorlund MD, Nielsen J, Pankratova S, Li S, Korshunova I, Bock E, Berezin V (2012) Neuroligin-1 induces neurite outgrowth through interaction with neurexin-1beta and activation of fibroblast growth factor receptor-1. FASEB J 26(10):4174–4186. doi:10.1096/fj.11-202242
Schnell E, Long TH, Bensen AL, Washburn EK, Westbrook GL (2014) Neuroligin-1 knockdown reduces survival of adult-generated newborn hippocampal neurons. Front Neurosci 8:71. doi:10.3389/fnins.2014.00071
Smith BN, Dudek FE (2002) Network interactions mediated by new excitatory connections between CA1 pyramidal cells in rats with kainate-induced epilepsy. J Neurophysiol 87(3):1655–1658
Penzes P, Cahill ME, Jones KA, VanLeeuwen JE, Woolfrey KM (2011) Dendritic spine pathology in neuropsychiatric disorders. Nat Neurosci 14(3):285–293. doi:10.1038/nn.2741
Song JY, Ichtchenko K, Sudhof TC, Brose N (1999) Neuroligin 1 is a postsynaptic cell-adhesion molecule of excitatory synapses. Proc Natl Acad Sci U S A 96(3):1100–1105
Lehmann TN, Gabriel S, Kovacs R, Eilers A, Kivi A, Schulze K, Lanksch WR, Meencke HJ, Heinemann U (2000) Alterations of neuronal connectivity in area CA1 of hippocampal slices from temporal lobe epilepsy patients and from pilocarpine-treated epileptic rats. Epilepsia 41(Suppl 6):S190–S194
Perez Y, Morin F, Beaulieu C, Lacaille JC (1996) Axonal sprouting of CA1 pyramidal cells in hyperexcitable hippocampal slices of kainate-treated rats. Eur J Neurosci 8(4):736–748
Kalia LV, Kalia SK, Salter MW (2008) NMDA receptors in clinical neurology: excitatory times ahead. Lancet Neurol 7(8):742–755. doi:10.1016/S1474-4422(08)70165-0
Churn SB, Sombati S, Jakoi ER, Severt L, DeLorenzo RJ (2000) Inhibition of calcium/calmodulin kinase II alpha subunit expression results in epileptiform activity in cultured hippocampal neurons. Proc Natl Acad Sci U S A 97(10):5604–5609. doi:10.1073/pnas.080071697080071697
Lau CG, Zukin RS (2007) NMDA receptor trafficking in synaptic plasticity and neuropsychiatric disorders. Nat Rev Neurosci 8(6):413–426. doi:10.1038/nrn2153
Seeburg PH (1993) The TINS/TiPS Lecture. The molecular biology of mammalian glutamate receptor channels. Trends Neurosci 16(9):359–365
Tovar KR, Westbrook GL (1999) The incorporation of NMDA receptors with a distinct subunit composition at nascent hippocampal synapses in vitro. J Neurosci 19(10):4180–4188
Blundell J, Blaiss CA, Etherton MR, Espinosa F, Tabuchi K, Walz C, Bolliger MF, Sudhof TC, Powell CM (2010) Neuroligin-1 deletion results in impaired spatial memory and increased repetitive behavior. J Neurosci 30(6):2115–2129. doi:10.1523/JNEUROSCI.4517-09.2010
Budreck EC, Kwon OB, Jung JH, Baudouin S, Thommen A, Kim HS, Fukazawa Y, Harada H, Tabuchi K, Shigemoto R, Scheiffele P, Kim JH (2013) Neuroligin-1 controls synaptic abundance of NMDA-type glutamate receptors through extracellular coupling. Proc Natl Acad Sci U S A 110(2):725–730. doi:10.1073/pnas.1214718110
Acknowledgments
This work was supported by National Natural Science Foundation Of China (grant numbers are 81401070, 81471319, and 81301630). The authors sincerely thank the patients and their families for their participation in this study. We thank the Xinqiao Hospital of the Third Military Medical University, Beijing Tiantan Hospital and Xuanwu Hospital of the Capital University of Medical Sciences for their support in brain tissue procurement, and the National Institutes of Health of China and the Ethics Committee on Human Research of the Chongqing Medical University.
Conflict of Interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fang, M., Wei, JL., Tang, B. et al. Neuroligin-1 Knockdown Suppresses Seizure Activity by Regulating Neuronal Hyperexcitability. Mol Neurobiol 53, 270–284 (2016). https://doi.org/10.1007/s12035-014-8999-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-014-8999-8