Abstract
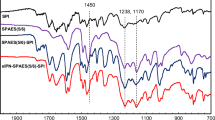
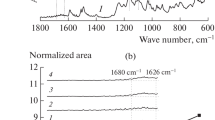
This study reports the preparation of a –COOH-containing sulphonated copolymer (SPI-COOH-70) and its composite membranes. The composite membranes (SPI/SS-X) were prepared by using the solution casting route by adding different weight percentages of 3-(trihydroxysilyl) propane-1-sulphonic acid into SPI-COOH-70 solution. The proton exchange membrane properties such as morphology, ion-exchange capacity, water uptake and proton conductivity of the prepared composite membranes were studied as a function of sulphopropylated polysilsesquioxane (SiOPS) filler and temperature. All the SPI/SS-X composite membranes showed high-thermal stability and chemical stability, which are attributed to the presence of polar phosphine oxide and the wholly aromatic nature of the copolymers. The homogeneous distribution of the SiOPS nanoparticles in the polymer matrix observed from the scanning electron microscopy and atomic force microscopy images are attributed to the formation of covalent bonds between the –OH and –COOH groups of SiOPS and SPI-COOH-70, respectively. The composite membranes with 10 wt% SiOPS exhibited much higher proton conductivity (205 mS cm−1), which is much higher than that of the pristine copolymer membrane (114 mS cm−1) under similar experimental conditions. The improvement in proton conductivity is attributed to the presence of pendant sulphonic acid groups in the nanofiller, which provides a better proton transport pathway in the composite membranes.
Graphic abstract











Similar content being viewed by others
References
Carrette L, Friedrich K A and Stimming U 2001 Fuel Cells 1 5
Miyahara T, Hayano T, Matsuno S, Watanabe M and Miyatake K 2012 ACS Appl. Mater. Interfaces 4 2881
Mauritz K A and Moore R B 2004 Chem. Rev. 104 4535
Harrison W L, Wang F, Mecham J B, Bhanu V A, Hill M, Kim Y S et al 2012 J. Polym. Sci. Part A: Polym. Chem. 41 2264
Kim D S, Robertson G P, Kim Y S and Guiver M D 2009 Macromolecules 42 957
Wang F, Hickner M, Ji Q, Harrison W, Mecham J, Zawodzinski T A et al 2001 Macromol. Symp. 175 387
Wang F, Hickner M, Kim Y S, Zawodzinski T A and McGrath J E 2002 J. Membr. Sci. 197 231
Kim Y S, Einsla B R, Sankir M, Harrison W and Pivovar B S 2006 Polymer 47 4026
Zhang N, Li J, Wang X, Xia Z and Liu H 2009 J. Appl. Polym. Sci. 114 304
Li N, Shin D W, Hwang D S, Lee Y M and Guiver M D 2010 Macromolecules 43 9810
Mohanty A K, Mistri E A, Ghosh A and Banerjee S 2012 J. Membr. Sci. 145 409
Mukherjee R, Banerjee S, Komber H and Voit B 2014 RSC Adv. 4 46723
Gao Y, Robertson G P, Guiver M D, Mikhailenko S D, Li X and Kaliaguine S 2005 Macromolecules 38 3237
Bai Z and Dang T D 2006 Macromol. Rapid Commun. 27 1271
Shin D W, Lee S Y, Kang N R, Lee K H, Guiver M D and Lee Y M 2013 Macromolecules 46 3452
Xing P, Robertson G P, Guiver M D, Mikhailenko S D and Kaliaguine S 2004 Macromolecules 37 7960
Shang X, Tian S, Kong L and Meng Y 2005 J. Membr. Sci. 266 94
Xing P, Robertson G P, Guiver M D, Mikhailenko S D and Kaliaguine S 2005 Polymer 46 3257
Liu B, Kim D S, Murphy J, Robertson G P, Guiver M D, Mikhailenko S et al 2006 J. Membr. Sci. 280 54
Zhang G, Fu T, Shao K, Li X, Zhao C, Na H et al 2009 J. Power Sources 189 875
Mistri E A, Mohanty A K, Banerjee S, Komber H and Voit B 2013 J. Membr. Sci. 441 168
Mandal A K, Bisoi S, Banerjee S, Komber H and Voit B 2017 Eur. Polym. J. 95 581
Singh A, Mukherjee R, Banerjee S, Komber H and Voit B 2014 J. Membr. Sci. 469 225
Saha S, Mukherjee R, Singh A and Banerjee S 2016 Polym. Eng. Sci. 57 312
Miyake N, Wainright J S and Savinell R F 2001 J. Electrochem. Soc. 148 A898
Mistri E A and Banerjee S 2014 RSC Adv. 4 22398
Adjemian K T, Srinivasan S, Benziger J and Bocarsly A B 2002 J. Power Sources 109 356
Sacca A, Carbone A, Passalacqua E, D’epifanio A, Licoccia S, Traversa E et al 2005 J. Power Sources 152 16
Casciola M, Bagnasco G, Donnadio A, Micoli L, Pica M, Sganappa M et al 2009 Fuel Cells 9 394
Kim Y S, Wang F, Hickner M A, Zawodzinski T A and McGrath J E 2003 J. Membr. Sci. 212 263
Wang Z, Ni H, Zhao C, Li X, Fu T and Na H 2006 J. Polym. Sci. Part B: Polym. Phys. 44 1967
Smitha B, Sridhar S and Khan A A 2005 J. Polym. Sci. Part B: Polym. Phys. 43 1538
Rhee C H, Kim H K, Chang H and Lee J S 2005 Chem. Mater. 17 1691
Su Y H, Liu Y L, Sun Y M, Lai J Y, Wang D M, Gao Y et al 2007 J. Membr. Sci. 296 21
Xu K, Chanthad C, Gadinski M R, Hickner M A and Wang Q 2009 ACS Appl. Mater. Interfaces 1 2573
Miyatake K, Tombe T, Chikashige Y, Uchida H and Watanabe M 2007 Angew. Chem. Int. Ed. 46 6646
Mandal A K, Bisoi S and Banerjee S 2019 ACS Appl. Polym. Mater. 1 893
Lin C H, Chang S L, Peng L A, Peng S P and Chuang Y H 2010 Polymer 51 3899
Mauritz K A and Warren R M 1989 Macromolecules 22 1730
Lufrano F, Squadrito G and Patti A 2000 J. Appl. Polym. Sci. 77 1250
Danilczuk M, Coms F D and Schlick S 2009 J. Phys. Chem. B 113 8031
So S Y, Yoon Y J, Kim T H, Yoon K and Hong Y T 2011 J. Membr. Sci. 381 204
Acknowledgement
AKM acknowledges CSIR, New Delhi, for the research assistantship.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
MANDAL, A.K., GHORAI, A. & BANERJEE, S. Sulphonated polysilsesquioxane–polyimide composite membranes: proton exchange membrane properties. Bull Mater Sci 43, 192 (2020). https://doi.org/10.1007/s12034-020-02158-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12034-020-02158-8