Abstract
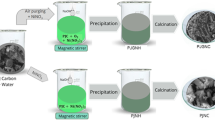
Hierarchical mesoporous NiO nanoflakes (NiOs) have been synthesized in high yield via a simple, economical and environmentally friendly hydrothermal route. The as-prepared NiOs were characterized by powder X-ray diffraction (PXRD), scanning electronic microscopy (SEM), transmission electron microscopy (TEM), selected area electron diffraction patterns (SAED), X-ray energy dispersive spectroscopy (EDS) and nitrogen adsorption–desorption techniques (Brunauer–Emmett–Teller, BET). Adsorption of heavy metal ions onto the as-prepared sample from aqueous solutions was investigated using differential pulse anodic stripping voltametry (DPASV) technique and discussed. The product possesses a BET surface area of 69.27 m2 g−1. It is found that NiOs exhibited the excellent performance for the removal of Hg(II), Pb(II) and Cd(II) from aqueous solution. The equilibrium adsorption data of Hg(II), Pb(II) and Cd(II) on the as-prepared NiOs were analyzed by Langmuir and Freundlich models, suggesting that the Langmuir model provides the better correlation of the experimental data. The adsorption capacities for removal of Hg(II), Pb(II) and Cd(II) were determined using the Langmuir equation and found to be 1324 .5, 1428 .9 and 1428 .5 mg g−1, respectively. Adsorption kinetics of all the metal ions followed pseudo second-order model. Moreover, NiOs can be recycled by simple acid treatment, which could retain the high removal efficiency in three successive cycles. This study suggests that nanoflakes could be explored as a new adsorbent with high efficiency and recyclability for removing heavy metal ions from aqueous solution.















Similar content being viewed by others
References
Mondal M K 2009 J. Environ. Manage. 90 3266
Hamidpour M, Kalbasi Afyuni M, Shariatmadari H, Holm P E and Hansen H C B 2010 J. Hazard. Mater. 181 686
Zein R, Suhaili R, Earnestly F, Indrawati and Munaf E 2010 J. Hazard. Mater. 181 52
Shafaei A, Ashtiani F Z and Kaghazchi T 2007 Chem. Eng. J. 133 311
Ho Y S and Wang C C 2008 J. Hazard. Mater. 156 398
Davis S A, Burkett S L, Mendelson N H and Mann S 1997 Nature 385 420
Walsh D, Arcelli L, Ikoma T, Tanaka J and Mann S 2003 Nat. Mater. 2 386
Yu J G, Su Y R and Cheng B 2007a Adv. Funct. Mater. 17 1984
Yu J G, Zhang L J, Cheng B and Su Y R 2007b J. Phys. Chem. C. 111 10582
Yogesh K K, Muralidhara H B, Arthoba N Y, Balasubramanyam J and Hanumanthappa H 2013a Powder Technol. 246 125
Liu J, Du S F, Wei L Q, Liu H D, Tian Y J and Chen Y F 2006 Mater. Lett. 60 3601
Yu J G, Liu W and Yu H G 2008 Cryst. Growth Des. 8 930
Yu J G and Qi L F 2009 J. Hazard. Mater. 169 221
Cai W Q, Yu J G, Cheng B, Su B L and Jaroniec M 2009 J. Phys. Chem. C. 113 14739
Yogesh K K, Muralidhara H B, Arthoba N Y, Balasubramanyam J and Hanumanthappa H 2013b Desalin. Water Treat. 51 1
Yogesh K K, Muralidhara H B, Arthoba N Y, Balasubramanyam J and Hanumanthappa H 2013c Powder Technol. 239 208
Izaki Y, Mugikura Y, Watanabe T, Kawase M and Selman J R 1998 J. Power. Sources 75 236
Ichiyanagi Y, Wakabayashi N, Yamazaki J, Yamada S and Kimishima Y 2003 Phys. B 329 862
Ai L and Zeng Y 2013 Chem. Eng. J. 215 269
Kasuga T 2006 Thin Solid Films 496 141
Hongxia Q, Zhiqiang W, Hua Y, Lin Z and Xiaoyan Y 2009 J. Nanomater. 2009 1
Lopez T, Manjarrez J, Rembao D, Vinogradova E, Moreno A and Gonzalez R D 2006 Mater. Lett. 60 2903
Bahari Y M M, Sadrnezhaad S K and Hosseini D 2008 J. Nanomater. 2008 4
Xu S and Wang Z L 2000 Nano Res. DOI: 10.1007/s12274-011-0160-7 10.1007/s12274-011-0160-7
Yakui B, Tengfei Y, Qing G, Guoan C, Ruiting Z, Yakui B, Tengfei Y, Qing G, Guoan C and Ruiting Z 2012 Powder Technol. 227 35
Rahman M S and Islam M R 2009 Chem. Eng. J. 149 273
Anbia M and Ghassemian Z 2011 Chem. Eng. J. 89 2770
Nian L, Lide Z, Yong Z C, Yue T and Huimin W 2011 J. Hazard. Mater. 189 265
Senthil Kumar P, Ramalingam S, Dinesh Kirupha S A, Murugesan A, Vidhyadevi C and Sivanesan C 2010 Chem. Eng. J. 133 311
Meng W W, Chi C K, Buenda D Rogel, Maria L P Dalida 2010 Carbohyr. Polym. 80 891
Cheng T W, Lee M L, Ko M S, Ueng T H and Yang S F 2012 Appl. Clay. Sci. 56 90
Li S X, Zheng F Y, Huang Y and Ni J C 2011 J. Hazard. Mater. 186 423
Xue M, Guanbo H, Weisong X, Wei W, Yu L and Jianping G 2012 Carbon 50 4856
Divya C and Nalini S 2008 Bioresour. Technol. 99 9021
Hardiljeet K B, Meera J and Denis M O 2011 J. Hazard. Mater. 186 458
Hasanur J, Debarati C and Papita S 2009 Clean-Soil, Air, Water 37 704
Neha C, Shweta G, Nahar S, Sukhvir S, Saikh S I, Kedar N S and Renu P 2011 J. Colloid. Interface Sci. 363 42
Acharya J, Sahu J N, Mohanty C R and Meikap B C 2009 Chem. Eng. J. 149 249
Zou W H, Han R P, Chen Z Z, Shi J and Liu H M 2006 J. Chem. Eng. Data 51 534
Wang S G, Gong W X, Liu X W, Yao Y W, Gao B Y and Yue Q Y 2007 Sep. Puri. Technol. 58 17
Sheela T, Arthoba N Y, Vishwanatha R, Basavanna S and Venkatesha T G 2012b Powder Technol. 217 163
Zabihi M, Haghighi A A and Ahmadpour A 2010 J. Hazard. Mater. 174 251
Gulay B M and Yakup A 2008 Chem. Eng. J. 143 133
Jeriffa D C 2002 Inter. J. Indus. Chem. 3 1
Sheela T and Arthoba N Y 2012a Chem. Eng. J. 191 123
Chunhua X and Caiping Y 2009 Chem. Eng. J. 155 844
Meena A K, Mishra G K, Rai P K, Rajagopal C and Nagar P N 2005 J. Hazard. Mater. 122 161
Acknowledgements
We are grateful to the Visvesvaraya Technological University (VTU) for providing financial support under the research grant scheme (Project No. VTU/Aca/2010-11/a-9/11353). We are also grateful to the authorities of the KS Institute of Technology, Bangalore, for their support and IIT Kanpur, IIT Bombay and PPRI Bangalore for providing instrumental facilities.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
KUMAR, K.Y., MURALIDHARA, H.B., NAYAKA, Y.A. et al. Cost effective and shape controlled approach to synthesize hierarchically assembled NiO nanoflakes for the removal of toxic heavy metal ions in aqueous solution. Bull Mater Sci 38, 271–282 (2015). https://doi.org/10.1007/s12034-014-0791-x
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12034-014-0791-x