Abstract
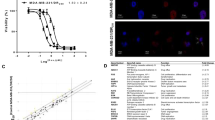
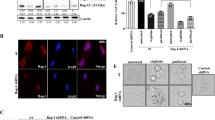
The B7-H7 is the newest addition to the B7 family of proteins that is present in the majority of malignancies. In this respect, the goal of the work was to investigate the impact of B7-H7 inhibition on breast cancer cells when paclitaxel and small interference RNA (siRNA) were combined. B7-H7 siRNA was used with Paclitaxel to treat MCF-7 cells. The IC50 of Paclitaxel and the cell survival was then assessed through using MTT assay. Investigation was conducted using flow cytometry to both the induction of apoptosis and the cell cycle. In addition, the clonogenic capacity of MCF-7 cells was investigated. Furthermore, qRT-PCR, was used to evaluate expression of genes. Our results demonstrated that suppressing B7-H7 sensitizes MCF-7 cells to Paclitaxel by triggering apoptosis and altering the expression of critical apoptosis mediator genes. In addition, the cell cycle was stopped in the sub-G1 and also G2-M phases as a result of the combination therapy leading prevention of developing colonies by MCF-7 cells. B7-H7 silencing improved the chemosensitivity of MCF-7 cells to Paclitaxel and demonstrated antiproliferative effects. After the adequate study has been conducted, this strategy may be regarded as a possible alternative treatment option for this cancer.







Similar content being viewed by others
Data Availability
Data is available upon request when necessary.
References
Momenimovahed, Z., & Salehiniya, H. (2017). Incidence, mortality and risk factors of cervical cancer in the world. Biomedical Research and Therapy, 4(12), 1795–1811.
Benson, J. R., & Jatoi, I. (2012). The global breast cancer burden. Future oncology, 8(6), 697–702.
Ferley, J., Soerjomataram, I., Ervik, M., Dikshit, R., Eser, S., & Mathers, C. (2017). GLOBOCAN 2012 v1. 2. Cancer incidence and mortality worldwide: IARC cancer base No. 11: International Agency for Research on Cancer. 2014.
Eslamkhah, S., Alizadeh, N., Safaei, S., Mokhtarzadeh, A., Amini, M., Baghbanzadeh, A., & Baradaran, B. (2021). Micro RNA-34a sensitizes MCF-7 breast cancer cells to carboplatin through the apoptosis induction. Gene Reports, 25, 101361.
Ries, L. A. G., Melbert, D., Krapcho, M., Stinchcomb, D. G., Howlader, N., Horner, M. J., Mariotto, A., Miller, B. A., Feuer, E. J., Altekruse, S. F., & Lewis, D. R. (2008). SEER cancer statistics review, 1975–2005 (p. 2999). National Cancer Institute.
Yedjou, C. G., Sims, J. N., Miele, L., Noubissi, F., Lowe, L., Fonseca, D. D., Alo, R. A., Payton, M., & Tchounwou, P. B. (2019). Health and racial disparity in breast cancer. In A. Ahmad (Ed.), Breast cancer metastasis and drug resistance (pp. 31–49). Springer.
Glass, A. G., & Hoover, R. N. (1990). Rising incidence of breast cancer: relationship to stage and receptor status. JNCI: Journal of the National Cancer Institute, 82(8), 693–696.
Li, C. I., Daling, J. R., & Malone, K. E. (2003). Incidence of invasive breast cancer by hormone receptor status from 1992 to 1998. Journal of Clinical Oncology, 21(1), 28–34.
Bigaard, J., Stahlberg, C., Jensen, M. B., Ewertz, M., & Kroman, N. (2012). Breast cancer incidence by estrogen receptor status in Denmark from 1996 to 2007. Breast Cancer Research and Treatment, 136(2), 559–564.
Brédart, A., De Pauw, A., Anota, A., Tüchler, A., Dick, J., Müller, A., Kop, J. L., Rhiem, K., Schmutzler, R., Devilee, P., & Stoppa-Lyonnet, D. (2021). Information needs on breast cancer genetic and non-genetic risk factors in relatives of women with a BRCA1/2 or PALB2 pathogenic variant. The Breast, 60, 38–44.
Wright, N., Akinyemiju, T., Subhedar, P., Rida, P., & Aneja, R. (2019). Targeting risk factors for reducing the racially disparate burden in breast cancer. Frontiers in Bioscience-Scholar, 11(1), 136–160.
Wunderle, M., Olmes, G., Nabieva, N., Häberle, L., Jud, S. M., Hein, A., Rauh, C., Hack, C. C., Erber, R., Ekici, A. B., & Hoyer, J. (2018). Risk, prediction and prevention of hereditary breast cancer–large-scale genomic studies in times of big and smart data. Geburtshilfe und Frauenheilkunde, 78(05), 481–492.
Dan, V. M., Raveendran, R. S., & Baby, S. (2021). Resistance to intervention: Paclitaxel in breast cancer. Mini Reviews in Medicinal Chemistry, 21(10), 1237–1268.
Shadbad, M. A., Asadzadeh, Z., Hosseinkhani, N., Derakhshani, A., Alizadeh, N., Brunetti, O., Silvestris, N., & Baradaran, B. (2021). A systematic review of the tumor-infiltrating CD8+ T-Cells/PD-L1 axis in high-grade glial tumors: Toward personalized immuno-oncology. Frontiers in Immunology, 12, 734956. https://doi.org/10.3389/fimmu.2021.734956
Howard, F. M., Villamar, D., He, G., Pearson, A. T., & Nanda, R. (2022). The emerging role of immune checkpoint inhibitors for the treatment of breast cancer. Expert Opinion on Investigational Drugs, 31(6), 531–548.
Alizadeh, N., Kazemi, T., Hemmat, N., Jafarlou, M., & Baradaran, B. (2023). The combination of PD-L1 and CTLA-4 suppression significantly decreased the expression levels of cancer stem cell factors in the pancreatic cancer cell line. ImmunoAnalysis, 3, 6. https://doi.org/10.34172/ia.2023.06
Solinas, C., Gombos, A., Latifyan, S., Piccart-Gebhart, M., Kok, M., & Buisseret, L. (2017). Targeting immune checkpoints in breast cancer: an update of early results. ESMO Open, 2(5), e000255.
Janakiram, M., Shah, U. A., Liu, W., Zhao, A., Schoenberg, M. P., & Zang, X. (2017). The third group of the B7-CD 28 immune checkpoint family: HHLA 2, TMIGD 2, B7x, and B7–H3. Immunological Reviews, 276(1), 26–39.
Yan, S., Yim, L. Y., Lu, L., Lau, C. S., & Chan, V. S. F. (2014). MicroRNA regulation in systemic lupus erythematosus pathogenesis. Immune Network, 14(3), 138–148.
Ni, L., & Dong, C. (2017). New B7 family checkpoints in human cancers. Molecular Cancer Therapeutics, 16(7), 1203–1211.
Amir Taghavi, B., Alizadeh, N., Saeedi, H., Karim Ahangar, N., Derakhshani, A., Hajiasgharzadeh, K., Silvestris, N., Baradaran, B., & Brunetti, O. (2022). Targeted therapy of B7 family checkpoints as an innovative approach to overcome cancer therapy resistance: A review from chemotherapy to immunotherapy. Molecules, 27(11), 3545.
Janakiram, M., Chinai, J. M., Fineberg, S., Fiser, A., Montagna, C., Medavarapu, R., Castano, E., Jeon, H., Ohaegbulam, K. C., Zhao, R., & Zhao, A. (2015). Expression, clinical significance, and receptor identification of the newest B7 family member HHLA2 ProteinHHLA is the newest immune checkpoint in human cancers. Clinical Cancer Research, 21(10), 2359–2366.
Wouters, M. C., Laumont, C. M., Chen, B., Han, S. J., Matuszewska, K., Potts, K., Boudreau, J. E., & Krawczyk, C. M. (2017). The summit for cancer immunotherapy (Summit4CI), June 26–29, 2016 Halifax, Canada. Springer.
Siegel, R. L., Miller, K. D., & Jemal, A. (2019). Cancer statistics, 2019. CA: A Cancer Journal for Clinicians, 69(1), 7–34.
Mansoori, B., Mohammadi, A., Asadzadeh, Z., Shirjang, S., Minouei, M., AbediGaballu, F., Shajari, N., Kazemi, T., Gjerstorff, M. F., Duijf, P. H., & Baradaran, B. (2019). HMGA2 and Bach-1 cooperate to promote breast cancer cell malignancy. Journal of Cellular Physiology, 234(10), 17714–17726.
Bhatt, R. S., Berjis, A., Konge, J. C., Mahoney, K. M., Klee, A. N., Freeman, S. S., Chen, C. H., Jegede, O. A., Catalano, P. J., Pignon, J. C., & Sticco-Ivins, M. (2021). KIR3DL3 is an inhibitory receptor for HHLA2 that mediates an alternative immunoinhibitory pathway to PD1KIR3DL3 is an immunoinhibitory receptor for HHLA2. Cancer Immunology Research, 9(2), 156–169.
Dolatkhah, K., Alizadeh, N., Mohajjel-Shoja, H., Abdoli Shadbad, M., Hajiasgharzadeh, K., Aghebati-Maleki, L., Baghbanzadeh, A., Hosseinkhani, N., Ahangar, N. K., & Baradaran, B. (2022). B7 immune checkpoint family members as putative therapeutics in autoimmune disease: An updated overview. International Journal of Rheumatic Diseases, 25(3), 259–271. https://doi.org/10.1111/1756-185X.14273
Saunders, D. E., Lawrence, W. D., Christensen, C., Wappler, N. L., Ruan, H., & Deppe, G. (1997). Paclitaxel-induced apoptosis in MCF-7 breast-cancer cells. International Journal of Cancer, 70(2), 214–220.
Chen, L., Zhu, D., Feng, J., Zhou, Y., Wang, Q., Feng, H., Zhang, J., & Jiang, J. (2019). Overexpression of HHLA2 in human clear cell renal cell carcinoma is significantly associated with poor survival of the patients. Cancer Cell International, 19(1), 1–12.
Aborehab, N. M., Elnagar, M. R., & Waly, N. E. (2021). Gallic acid potentiates the apoptotic effect of paclitaxel and carboplatin via overexpression of Bax and P53 on the MCF-7 human breast cancer cell line. Journal of Biochemical and Molecular Toxicology, 35(2), e22638.
Quispe-Soto, E. T., & Calaf, G. M. (2016). Effect of curcumin and paclitaxel on breast carcinogenesis. International Journal of Oncology, 49(6), 2569–2577.
Sun, W., Li, S., Tang, G., Sun, S., Luo, Y., Bai, R., Han, L., Jiang, X., Gao, Y., Huang, Z., & Zhang, J. (2021). HHLA2 deficiency inhibits non-small cell lung cancer progression and THP-1 macrophage M2 polarization. Cancer Medicine, 10(15), 5256–5269.
Acknowledgements
This study was supported by the Immunology Research Center, Tabriz University of Medical Sciences, Tabriz, Iran.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Taghavi, B.A., Salehi, M., Mokhtarzadeh, A. et al. Suppression of B7-H7 Enhanced MCF-7 Cancer Cell Line’s Chemosensitivity to Paclitaxel. Mol Biotechnol (2024). https://doi.org/10.1007/s12033-024-01145-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12033-024-01145-2