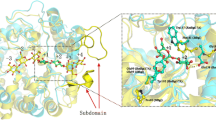
Abstract
Fusion protein combined the oligopeptide (HQAFFHA) with the C terminus of α-glucuronidase from Thermotoga maritima was produced in E. coli and purified for characterization and applications of glucuronic and glucaric acid production. The fusion protein with oligopeptide exhibited a 2.97-fold higher specific activity than individual protein. Their catalytic efficiency kcat/Km and kcat increased from 469.3 ± 2.6 s−1 (g mL−1)−1 and 62.4 ± 0.9 s−1 to 2209.5 ± 26.3 s−1 (g mL−1)−1 and 293.9 ± 4.9 s−1, respectively. Fusion protein had similar temperature and pH profiles to those without oligopeptide, but the thermal stability decreases and the pH stability shifts to alkaline. Using beech xylan hydrolysate as a substrate, the glucuronic acid yield of fusion enzyme increased by 9.94% compared with its parent at 65 °C pH 8.5 for 10 h, and can hydrolyze corn cob xylan with xylanase to obtain glucuronic acid, and can be combined with uronate dehydrogenase to obtain high-added value glucaric acid. Homologous modeling analysis revealed the factors contributing to the high catalytic efficiency of fusion enzyme. These results show that the peptide fusion strategy described here may be useful for improving the catalytic efficiency and stability of other industrial enzymes, and has great potential for producing high value-added products from agricultural waste.





Similar content being viewed by others
References
Zhang, H. S. (1990). Study on the hemicellulose and its structure. Guangzhou Chemistry, 1, 57–66.
Zhu, Y. P., Li, X. T., Sun, B. G., Song, H. L., Li, E., & Song, H. X. (2012). Properties of an alkaline-tolerant, thermostable xylanase from streptomyces chartreusis L1105, suitable for xylooligosaccharide production. Journal of Food Science, 77, C506–C511.
Zhang, H., & Sang, Q. (2015). Production and extraction optimization of xylanase and β-mannanase by Penicillium chrysogenum QML-2 and primary application in saccharification of corn cob. Biochemical Engineering Journal, 97, 101–110.
Christakopoulos, P., Katapodis, P., Kalogeris, E., Kekos, D., & Skaltsa, H. (2003). Antimicrobial activity of acidic xylo-oligosaccharides produced by family 10 and 11 endoxylanases. International Journal of Biological Macromolecules, 31, 171–175.
Tuncer, M., & Ball, A. S. (2010). Co-operative actions and degradation analysis of purified xylan-degrading enzymes from Thermomonospora fusca BD25 on oat-spelt xylan. Journal of Applied Microbiology, 94, 1030–1035.
Ruile, P., Winterhalter, C., & Liebl, W. (1997). Isolation and analysis of a gene encoding -glucuronidase, an enzyme with a novel primary structure involved in the breakdown of xylan. Molecular Microbiology, 23, 267–279.
Sunna, A., & Antranikian, G. (1997). Xylanolytic enzymes from fungi and bacteria. Critical Reviews in Biotechnology, 17, 39–67.
Zhou, L. (2012). Thoughts on the domestic present condition of straw utilization. Journal of Anhui Agricultural Sciences, 40, 15853–15855.
Wu, J. L., Xue, Y., Li, H. L., Gan, L. H., Liu, J., & Long, M. N. (2014). Research progress of α-glucuronidase, an enzyme for degrading hemicellulose side-chain. Advances in New Renewable Energy, 2, 327–333.
Johnson, K. G., Silva, M. C., Mackenzie, C. R., Schneider, H., & Fontana, J. D. (1989). Microbial degradation of hemicellulosic materials. Applied Biochemistry & Biotechnology, 20–21, 245–258.
Xue, Y. M., Mao, Z. G., & Shao, W. L. (2004). Expression and purification of thermostable alpha-glucuronidase from Thermotoga maritima. Chinese Journal of Biotechnology, 20, 554–560.
Dong, Y. Y., Li, Y. X., Shen, Y. H., & Xue, Y. M. (2018). Optimization of hydrolysis conditions of xylanase and α-glucuronidase mixture for birch xylan by response surface methodology. Food Science, 39, 125–131.
Raji, O., Bth, J. A., Vuong, T. V., Larsbrink, J. O. L., & Master, E. R. (2020). The coordinated action of glucuronoyl esterase and α-glucuronidase promotes the disassembly of lignin-carbohydrate complexes. FEBS Letters, 595, 351–359.
Golan, G., Shallom, D., Teplitsky, A., Zaide, G., & Shoham, G. (2004). Crystal structures of geobacillus stearothermophilus α-glucuronidase complexed with its substrate and products. Journal of Biological Chemistry, 279, 3014–3024.
Margolles-Clark, E., Saloheimo, M., Siika-Aho, M., & Penttil, M. (1996). The α-glucuronidase-encoding gene of Trichoderma reesei. Gene, 172, 171–172.
Haar, R. T., Timmermans, J. W., Slaghek, T. M., Dongen, F., Schols, H. A., & Gruppen, H. (2010). TEMPO oxidation of gelatinized potato starch results in acid resistant blocks of glucuronic acid moieties. Carbohydrate Polymers, 81, 830–838.
Yan, M. A., Zhao, L. L., Liu, H. Q., & Yuan, H. (2011). Progress in the preparation of glucuronic acid by catalytic oxidative methods from starch and its derivatives. Applied Chemical Industry, 40, 1244–1247.
Kim, Y. G., Hwang, J., Choi, H., & Lee, S. (2018). Development of a column-switching HPLC-MS/MS method and clinical application for determination of ethyl glucuronide in hair in conjunction with AUDIT for detecting high-risk alcohol consumption. Pharmaceutics, 10, 84–95.
Lee, C. C., Kibblewhite, R. E., Paavola, C. D., Orts, W. J., & Wagschal, K. (2016). Production of glucaric acid from hemicellulose substrate by rosettasome enzyme assemblies. Molecular Biotechnology, 58, 489–496.
Zhou, T., Xue, Y. M., Ren, F. J., & Dong, Y. Y. (2018). Antioxidant activity of xylooligosaccharides prepared from Thermotoga maritima using recombinant enzyme cocktail of β-xylanase and α-glucuronidase. Journal of Carbohydrate Chemistry, 37, 210–224.
Hong, S. Y., Lee, J. S., Cho, K. M., Math, R. K., & Yun, H. D. (2007). Construction of the bifunctional enzyme cellulase-β-glucosidase from the hyperthermophilic bacterium Thermotoga maritima. Biotechnology Letters, 29, 931–936.
Bülow, L., & Mosbach, K. (1991). Multienzyme systems obtained by gene fusion. Trends in Biotechnology, 9, 226–231.
Sørensen, H. P., & Mortensen, K. K. (2005). Advanced genetic strategies for recombinant protein expression in Escherichia coli. Journal of Biotechnology, 115, 113–128.
Makrides, S. C. (1996). Strategies for achieving high-level expression of genes in Escherichia coli. Microbiological Reviews, 60, 512–538.
Ling, Z., Kang, Z., Liu, Y., Liu, S., Chen, J., & Du, G. (2014). Improvement of catalytic efficiency and thermostability of recombinant streptomyces griseus trypsin by introducing artificial peptide. World Journal of Microbiology and Biotechnology, 30, 1819–1827.
Yang, H. Q., Lu, X. Y., Liu, L., Li, J. H., Shin, H. D., Chen, R. R., Du, G. C., & Chen, J. (2013). Fusion of an oligopeptide to the N terminus of an alkaline alpha-amylase from Alkalimonas Amylolytica simultaneously improves the enzyme’s catalytic efficiency, thermal stability, and resistance to oxidation. Applied & Environmental Microbiology, 79, 3049–3058.
Li, Z. Y., Xue, X. L., Zhao, H., Yang, P. L., Luo, H. Y., & Zhao, J. Q. (2014). A C-terminal proline-rich sequence simultaneously broadens the optimal temperature and ph ranges and improves the catalytic efficiency of glycosyl hydrolase family 10 ruminal xylanases. Applied and Environmental Microbiology, 80, 3426–3432.
Wu, W., Xing, L., Zhou, B. H., & Lin, Z. L. (2011). Active protein aggregates induced by terminally attached self-assembling peptide ELK16 in Escherichia coli. Microbial Cell Factories, 10, 1–8.
Xing, L., Wu, W., Zhou, B. H., & Lin, Z. L. (2011). Streamlined protein expression and purification using cleavable self-aggregating tags. Microbial Cell Factories, 10, 42.
Xue, Y. M., Mao, Z. G., & Shao, W. L. (2003). Expression of xylanase B gene of Thermotoga maritima in Escherichia coli. Food and Fermentation Industries, 191, 20–25.
Wu, H. W., Pei, J. J., Wu, G. G., & Shao, W. L. (2008). Overexpression of GH10 endoxylanase XynB from Thermotoga maritima in Escherichia coli by a novel vector with potential for industrial application. Enzyme Microbial Technology, 42, 230–234.
Bradford, M. M. (1976). A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analytical Biochemistry, 72, 248–254.
Milner, Y., & Avigad, G. (1967). A copper reagent for the determination of hexuronic acids and certain ketohexoses. Carbohydrate Research, 4, 359–361.
Lineweaver, H., & Burk, D. (1934). The determination of enzyme dissociation-constant. Journal of the American Chemical Society, 56, 658–666.
He, F. L., Shen, S. W., Zeng, Y., & Liu, X. (2015). Studies on Alkali Extraction of Xylan from Corncobs and Preparation of Xylooligosaccharides by Xylanase Hydrolysis. Packaging and Food Machinery, 33, 28–33.
Li, Y. X., Xue, Y. M., Cao, Z. G., Zhou, T., & Alnadari, F. (2018). Characterization of a uronate dehydrogenase from Thermobispora bispora for production of glucaric acid from hemicellulose substrate. World Journal of Microbiology & Biotechnology, 34, 102.
Melton, L. D., & Smith, B. G. (2001). Determination of the uronic acid content of plant cell walls using a colorimetric assay. Current Protocols in Food Analytical Chemistry, 1, E3.3.1-E3.3.4.
Telke, A. A., Ghatge, S. S., Kang, S. H., Thangapandian, S., Lee, K. W., Shin, H. D., Um, Y., & Kim, S. W. (2012). Construction and characterization of chimeric cellulases with enhanced catalytic activity towards insoluble cellulosic substrates. Bioresource Technology, 112, 10–17.
Kim, T. W., Chokhawala, H. A., Nadler, D., Blanch, H. W., & Clark, D. S. (2010). Binding modules alter the activity of chimeric cellulases: Effects of biomass pretreatment and enzyme source. Biotechnology and Bioengineering, 107, 601–611.
Xue, Y. M., Song, X. F., & Yu, J. J. (2009). Overexpression of β-glucosidase from Thermotoga maritima for the production of highly purified aglycone isoflavones from soy flour. World Journal of Microbiology & Biotechnology, 25, 2165–2172.
Liu, X. F., Chi, J. X., Lei, W. P., & Liu, C. G. (2020). Screening of lactic acid bacteria with high bile salt hydrolase activity and studying of its influencing factors. Food and Fermentation Industries, 46, 63–68.
Merbouh, N., Thaburet, J. F., Ibert, M., Marsais, F., & Bobbitt, J. M. (2001). Facile nitroxide-mediated oxidations of D-glucose to D-glucaric acid. Carbohydrate Research, 336, 75–78.
Kiely, D. E., Chen, L., & Lin, T. H. (1994). Hydroxylated nylons based on unprotected esterified D-glucaric acid by simple condensation reactions. Journal of the American Chemical Society, 116, 571–578.
Werpy, T., Petersen, G., Aden, A., Bozell, J. J., & Jones, S. (2004) Top value added chemicals from biomass: I. Results of screening for potential candidates from sugars and synthesis gas. Synthetic Fuels 69.
Zhang, Q., Wan, Z., Yu, I. K. M., & Tsang, D. C. W. (2021). Sustainable production of high-value gluconic acid and glucaric acid through oxidation of biomass-derived glucose: A critical review. Journal of Cleaner Production, 312, 127745.
Chen, H., & Zhou, J. (2009). Prediction-methods researqch of protein structure based on homologous modelling. Henan Science, 27, 1108–1110.
Bi, Y. F., Xie, G. Q., Gao, R. J., Lu, J., & Cao, S. G. (2007). Effect of specific hydrogen bond on activity and thermostability of hyperthermophilic esterase APE1547. Chemical Journal of Chinese Universities, 28, 1914–1916.
Funding
This work was supported by grants from “National Key Research and Development Project” of China (Grant No. 2019YFA0706900).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhao, H., Xue, Y., Xue, M. et al. Fusion of Oligopeptide to the C Terminus of α-Glucuronidase from Thermotoga maritima Improves the Catalytic Efficiency for Hemicellulose Biotransformation. Mol Biotechnol 65, 741–751 (2023). https://doi.org/10.1007/s12033-022-00569-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12033-022-00569-y