Abstract
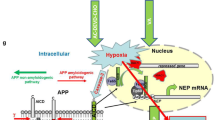
Development of the olfactory system begins early in embryogenesis and is important for the survival of new-borns in postnatal life. Olfactory malfunction in early life disrupts development of behavioural patterns while with ageing manifests development of neurodegenerative disorders. Previously, we have shown that prenatal hypoxia in rats leads to impaired olfaction in the offspring and correlates with reduced expression of a neuropeptidase neprilysin (NEP) in the brain structures involved in processing of the olfactory stimuli. Prenatal hypoxia also resulted in an increased activity of caspases in rat brain and its inhibition restored NEP content in the brain tissue and improved rat memory. In this study, we have analysed effects of intraventricular administration of a caspase inhibitor Ac-DEVD-CHO on NEP mRNA expression, the number of dendritic spines and olfactory function of rats subjected to prenatal hypoxia on E14. The data obtained demonstrated that a single injection of the inhibitor on P20 restored NEP mRNA levels and number of dendritic spines in the entorhinal and parietal cortices, hippocampus and rescued rat olfactory function in food search and odour preference tests. The data obtained suggest that caspase activation caused by prenatal hypoxia contributes to the olfactory dysfunction in developing animals and that caspase inhibition restores the olfactory deficit via upregulating NEP expression and neuronal networking. Because NEP is a major amyloid-degrading enzyme, any decrease in its expression and activity not only impairs brain functions but also predisposes to accumulation of the amyloid-β peptide and development of neurodegeneration characteristic of Alzheimer’s disease.





Similar content being viewed by others
Availability of Data and Materials
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Barresi M, Ciurleo R, Giacoppo S, Foti Cuzzola V, Celi D, Bramanti P, Marino S (2012) Evaluation of olfactory dysfunction in neurodegenerative diseases. J Neurol Sci 323:16–24. https://doi.org/10.1016/j.jns.2012.08.028
Barnes K, Turner AJ, Kenny AJ (1992) Membrane localization of endopeptidase-24.11 and peptidyl dipeptidase A (angiotensin converting enzyme) in the pig brain: a study using subcellular fractionation and electron microscopic immunocytochemistry. J Neurochem 58:2088–2096
Belyaev ND, Kellett KA, Beckett C, Makova NZ, Revett TJ, Nalivaeva NN, Hooper NM, Turner AJ (2010) The transcriptionally active amyloid precursor protein (APP) intracellular domain is preferentially produced from the 695 isoform of APP in a β-secretase-dependent pathway. J Biol Chem 285:41443–41454
Belyaev ND, Nalivaeva NN, Makova NZ, Turner AJ (2009) Neprilysin gene expression requires binding of the amyloid precursor protein intracellular domain to its promoter: implications for Alzheimer disease. EMBO Rep 10:94–100
Bertrand E, Brouillet E, Caille I, Bouillot C, Cole GM, Prochiantz A, Allinquant B (2001) A short cytoplasmic domain of the amyloid precursor protein induces apoptosis in vitro and in vivo. Mol Cell Neurosci 18:503–511. https://doi.org/10.1006/mcne.2001.1030
Carnemolla SE, Hsieh JW, Sipione R, Landis BN, Kumfor F, Piguet O, Manuel AL (2020) Olfactory dysfunction in frontotemporal dementia and psychiatric disorders: a systematic review. Neurosci Biobehav Rev 118:588–611. https://doi.org/10.1016/j.neubiorev.2020.08.002
Chang LR, Liu JP, Song YZ, Lu T, Lu G, Wu Y (2011) Expression of caspase-8 and caspase-9 in rat hippocampus during postnatal development. Microsc Res Tech 74:153–158. https://doi.org/10.1002/jemt.20886
Chen H, Tian J, Guo L, Du H (2020) Caspase inhibition rescues F1Fo ATP synthase dysfunction-mediated dendritic spine elimination. Sci Rep 10:17589. http://doi.org/10.1038/s41598-020-74613-9
Colín-Barenque L, Pedraza-Chaverri J, Medina-Campos O, Jimenez-Martínez R, Bizarro-Nevares P, González-Villalva A, Rojas-Lemus M, Fortoul TI (2015) Functional and morphological olfactory bulb modifications in mice after vanadium inhalation. Toxicol Pathol 43:282–291. https://doi.org/10.1177/0192623314548668
Doty RL (2012) Olfactory dysfunction in Parkinson disease. Nat Rev Neurol 8:329–339. https://doi.org/10.1038/nrneurol.2012.80
Dubrovskaya NM, Vasilev DS, Tumanova NL, Alekseeva OS, Nalivaeva NN (2020) Prenatal hypoxia impairs olfactory function in postnatal ontogenesis of rats. Zh Vyssh Nerv Deiat Im I P Pavlova 71: 415–427. (Article in Russian). http://doi.org/10.31857/S0044467721030035
Duffy AM, Morales-Corraliza J, Bermudez-Hernandez KM, Schaner MJ, Magagna-Poveda A, Mathews PM, Scharfman HE (2015) Entorhinal cortical defects in Tg2576 mice are present as early as 2–4 months of age. Neurobiol Aging 36:134–148. https://doi.org/10.1016/j.neurobiolaging.2014.07.001
Faul F, Erdfelder E, Lang A-G, Buchner A (2007) G*Power 3: a flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav Res Methods 39:175–191. https://doi.org/10.3758/BF03193146
Franks KM, Isaacson JS (2005) Synapse-specific downregulation of NMDA receptors by early experience: a critical period for plasticity of sensory input to olfactory cortex. Neuron 47:101–114. https://doi.org/10.1016/j.neuron.2005.05.024
Fukami S, Watanabe K, Iwata N, Haraoka J, Lu B, Gerard NP, Gerard C, Fraser P, Westaway D, St George-Hyslop P, Saido TC (2002) Aβ-degrading endopeptidase, neprilysin, in mouse brain: synaptic and axonal localization inversely correlating with Abeta pathology. Neurosci Res 43:39–56. https://doi.org/10.1016/s0168-0102(02)00015-9
Hardy J (2009) The amyloid hypothesis for Alzheimer’s disease: a critical reappraisal. J Neurochem 110:1129–1134. https://doi.org/10.1111/j.1471-4159.2009.06181.x
Kerridge C, Kozlova DI, Nalivaeva NN, Turner AJ (2015) Hypoxia affects neprilysin expression through caspase activation and an APP intracellular domain-dependent mechanism. Front Neurosci 9:426. https://doi.org/10.3389/fnins.2015.00426
Kilkenny C, Browne WJ, Cuthill IC, Emerson M, Altman DG (2010) Improving bioscience research reporting: the ARRIVE guidelines for reporting animal research. PLoS Biol 8:e1000412. https://doi.org/10.1371/journal.pbio.1000412
Kozlova DI, Vasylev DS, Dubrovskaya NM, Nalivaeva NN, Tumanova NL, Zhuravin IA (2015) Role of caspase-3 in regulation of the amyloid-degrading neuropeptidase neprilysin level in the rat cortex after hypoxia. J Evol Biochem Phys 51:480–484. https://doi.org/10.1134/S0022093015060046
Liu JP, Chang LR, Gao XL, Wu Y (2008) Different expression of caspase-3 in rat hippocampal subregions during postnatal development. Microsc Res Techn 71:633–638. https://doi.org/10.1002/jemt.20600
Louneva N, Cohen JW, Han LY, Talbot K, Wilson RS, Bennett DA, Trojanowski JQ, Arnold SE (2008) Caspase-3 is enriched in postsynaptic densities and increased in Alzheimer’s disease. Am J Pathol 173:1488–1495. https://doi.org/10.2353/ajpath.2008.080434.
Manczak M, Kandimalla R, Yin X, Reddy PH (2018) Hippocampal mutant APP and amyloid β-induced cognitive decline, dendritic spine loss, defective autophagy, mitophagy and mitochondrial abnormalities in a mouse model of Alzheimer’s disease. Hum Mol Genet 27:1332–1342. https://doi.org/10.1093/hmg/ddy042
McClendon E, Chen K, Gong X, Sharifnia E, Hagen M, Cai V, Shaver DC, Riddle A, Dean JM, Gunn AJ, Mohr C, Kaplan JS, Rossi DJ, Kroenke CD, Hohimer AR, Back SA (2014) Prenatal cerebral ischemia triggers dysmaturation of caudate projection neurons. Ann Neurol 75:508–524. https://doi.org/10.1002/ana.24100
Müller-Schwarze D, Müller-Schwarze C, Singer AG, Silverstein RM (1974) Mammalian pheromone: identification of active component in the subauricular scent of the male pronghorn. Science 183:860–862. https://doi.org/10.1126/science.183.4127.860
Murphy C (2019) Olfactory and other sensory impairments in Alzheimer disease. Nat Rev Neurol 15:11–24. https://doi.org/10.1038/s41582-018-0097-5
Nalivaeva NN, Belyaev ND, Turner AJ (2016) New insights into epigenetic and pharmacological regulation of amyloid-degrading enzymes. Neurochem Res 41:620–630. https://doi.org/10.1007/s11064-015-1703-1
Nalivaeva NN, Turner AJ, Zhuravin IA (2018) Role of prenatal hypoxia in brain development, cognitive functions, and neurodegeneration. Front Neurosci 12:825. https://doi.org/10.3389/fnins.2018.00825
Nalivaeva NN, Vasilev DS, Dubrovskaya NM, Turner AJ, Zguravin IA (2020a) Role of neprilysin in synaptic plasticity and memory. Russ J Physiol 106:1191–1208. https://doi.org/10.31857/S0869813920100076.
Nalivaeva NN, Zhuravin IA, Turner AJ (2020b) Neprilysin expression and functions in development, ageing and disease. Mech Ageing Dev 192:111363. https://doi.org/10.1016/j.mad.2020.111363
Nocera S, Simon A, Fiquet O, Chen Y, Gascuel J, Datiche F, Schneider N, Epelbaum J, Viollet C (2019) Somatostatin serves a modulatory role in the mouse olfactory bulb: neuroanatomical and behavioral evidence. Front Behav Neurosci 13:61. https://doi.org/10.3389/fnbeh.2019.00061
Pardo GVE, Lucion AB, Calcagnotto ME (2018) Postnatal development of inhibitory synaptic transmission in the anterior piriform cortex. Int J Dev Neurosci 71:1–9. https://doi.org/10.1016/j.ijdevneu.2018.07.008
Pardossi-Piquard R, Petit A, Kawarai T, Sunyach C, Alves da Costa C, Vincent B, Ring S, D’Adamio L, Shen J, Müller U, St George Hyslop P, Checler F (2005) Presenilin-dependent transcriptional control of the Aβ-degrading enzyme neprilysin by intracellular domains of βAPP and APLP. Neuron 46:541–554. https://doi.org/10.1016/j.neuron.2005.04.008
Paxinos G, Watson C (2005) The rat brain in stereotaxic coordinates. Elsevier Academic Press, Amsterdam-Boston
Pérez J, Rigo M, Kaupmann K, Bruns C, Yasuda K, Bell GI, Lübbert H, Hoyer D (1994) Localization of somatostatin (SRIF) SSTR-1, SSTR-2 and SSTR-3 receptor mRNA in rat brain by in situ hybridization. Naunyn Schmiedebergs Arch Pharmacol 349:145–160. https://doi.org/10.1007/BF00169831
Singh PJ, Tobach E (1975) Olfactory bulbectomy and nursing behavior in rat pups (Wistar DAB). Dev Psychobiol 8:151–164. https://doi.org/10.1002/dev.420080207
Sun S, Li T, Davies H, Li W, Yang J, Li S, Ling S (2016) Altered morphologies and functions of the olfactory bulb and hippocampus induced by miR-30c. Front Neurosci 10:207. https://doi.org/10.3389/fnins.2016.00207
Tumanova NL, Vasilev DS, Dubrovskaya NM, Nalivaeva NN, Zhuravin IA (2021) Effect of prenatal hypoxia on cytoarchitectonics and ultrustructural organisation of brain regions related to olfaction in rats. Cell Tiss Biol 15:482–492. https://doi.org/10.1134/S1990519X21050114
Van den Hove DL, Steinbusch HW, Scheepens A, Van de Berg WD, Kooiman LA, Boosten BJ, Prickaerts J, Blanco CE (2006) Prenatal stress and neonatal rat brain development. Neuroscience 137:145–155. https://doi.org/10.1016/j.neuroscience.2005.08.060
Vasilev DS, Dubrovskaya NM, Nalivaeva NN, Zhuravin IA (2016a) Regulation of caspase-3 content and activity in rat cortex in norm and after prenatal hypoxia. Neurochem J 10:144–150. https://doi.org/10.1134/S1819712416020100
Vasilev DS, Dubrovskaya NM, Tumanova NL, Zhuravin IA (2016b) Prenatal hypoxia in different periods of embryogenesis differentially affects cell migration, neuronal plasticity, and rat behavior in postnatal ontogenesis. Front Neurosci 10:126. https://doi.org/10.3389/fnins.2016.00126
Vasilev DS, Dubrovskaya NM, Zhuravin IA, Nalivaeva NN (2021) Developmental profile of brain neprilysin expression correlates with olfactory behaviour of rats. J Mol Neurosci 71:1772–1785. https://doi.org/10.1007/s12031-020-01786-3
Zhuravin IA, Dubrovskaya NM, Vasilev DS, Kozlova DI, Kochkina EG, Tumanova NL, Nalivaeva NN (2019) Regulation of neprilysin activity and cognitive functions in rats after prenatal hypoxia. Neurochem Res 44:1387–1398. https://doi.org/10.1007/s11064-019-02796-3
Zhuravin IA, Dubrovskaya NM, Vasilev DS, Tumanova NL, Nalivaeva NN (2011) Epigenetic and pharmacological regulation of the amyloid-degrading enzyme neprilysin results in modulation of cognitive functions in mammals. Dokl Biol Sci 438:145–148. https://doi.org/10.1134/S001249661103015X
Acknowledgements
Authors express their deep gratitude to Dr Olga S Alekseeva (IEPhB RAS) for assistance in performing prenatal hypoxia experiments. All molecular biology experiments were carried out using the facilities of the Communal Research Resource Centre for physiological, biochemical and molecular-biological research of IEPhB RAS.
Funding
The work was supported by the Russian Foundation for Basic Research, grant 19–015-00232, and Russian Federation state budget (assignment AAAA-A18-118012290373–7).
Author information
Authors and Affiliations
Contributions
All authors have been participating in design of the study and discussion of the results. Dr DS Vasilev performed tissue extraction, rt-PCR and statistical analysis of the data, and Dr NM Dubrovskaya performed animal and behavioural experiments and statistical analysis of the data. Dr NL Tumanova performed microscopy studies. Dr NN Nalivaeva has built the concept of the study and written the manuscript.
Corresponding author
Ethics declarations
Ethics Approval
All procedures were carried out in accordance with the international guidelines for work with experimental animals and the guidelines of the Russian Academy of Sciences (RAS) and approved by the Scientific Ethical Council of the Institute of Evolutionary Physiology and Biochemistry RAS.
Consent for Publication
All authors read and agreed to publish this paper. The submitted work is original and has not been published elsewhere and is not under consideration in any other journal.
Competing Interests
The authors declare no competing interests.
Additional information
This paper is a part of a special issue dedicated to the 1st ESN Virtual Conference “FUTURE PERSPECTIVES FOR EUROPEAN NEUROCHEMISTRY – A YOUNG SCIENTIST’S CONFERENCE”
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Vasilev, D., Dubrovskaya, N.M. & Nalivaeva, N.N. Caspase Inhibition Restores NEP Expression and Rescues Olfactory Deficit in Rats Caused by Prenatal Hypoxia. J Mol Neurosci 72, 1516–1526 (2022). https://doi.org/10.1007/s12031-022-01986-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-022-01986-z