Abstract
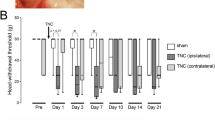
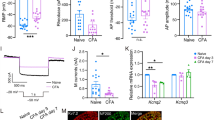
Recent clinical studies have revealed sex differences in response to transient receptor potential vanilloid 1 (TRPV1) agonist-induced pain. However, the mechanism of these differences in TRPV1-related chronic pain remains unclear. In the present study, we investigate the effects of inflammation and gonadal hormones on TRPV1 expression in trigeminal ganglia. Inflammatory pain was modeled by injecting complete Freund’s adjuvant (CFA) into the left masseter muscle in rats. TRPV1 mRNA and protein levels in the trigeminal ganglia of male and female rats following CFA injection were assessed. CFA-induced changes in TRPV1 mRNA and protein expression in the trigeminal ganglia from orchidectomized (ODX) male rats and testosterone-replaced ODX rats were examined. Additionally, TRPV1 mRNA levels in the trigeminal ganglia from ovariectomized (OVX) female and ODX male rats treated with tamoxifen were assessed. We found that the levels of TRPV1 mRNA and protein in the trigeminal ganglia from female rats following CFA injection were significantly higher than in the ganglia from naïve female rats. CFA-induced inflammatory hyperalgesia did not alter TRPV1 expression in the trigeminal ganglia from male rats. The TRPV1 mRNA and protein expression levels in the ODX male trigeminal ganglia were significantly upregulated on day 3 following the initiation of inflammation. However, CFA-induced inflammatory pain had no significant effect on TRPV1 mRNA or protein expression in testosterone-replaced ODX rats. Furthermore, tamoxifen was unable to inhibit the upregulation of TRPV1 expression in OVX female and ODX male rats after CFA injection. In summary, these data indicate that gender differences in TRPV1 function may be, in part, mediated by sex-dependent TRPV1 expression in sensory ganglia. Testosterone plays a key role in the inhibition of TRPV1 expression in this rat chronic inflammatory pain model.






Similar content being viewed by others
Abbreviations
- CB1:
-
Cannabinoid type 1 receptor
- CFA:
-
Complete Freund’s adjuvant
- MOR:
-
μ-Opioid receptor
- ODX:
-
Orchidectomized
- OVX:
-
Ovariectomized
- TG:
-
Trigeminal ganglia
- TRPV1:
-
Transient receptor potential vanilloid 1
References
Aloisi AM, Ceccarelli I, Carlucci M, Suman A, Sindaco G, Mameli S, Paci V, Ravaioli L, Passavanti G, Bachiocco V, Pari G (2011) Hormone replacement therapy in morphine-induced hypogonadic male chronic pain patients. Reprod Biol Endocrinol 9(1):26. https://doi.org/10.1186/1477-7827-9-26
Ambalavanar R, Moutanni A, Dessem D (2006) Inflammation of craniofacial muscle induces widespread mechanical allodynia. Neurosci Lett 399(3):249–254. https://doi.org/10.1016/j.neulet.2006.02.003
Bai X, Zhang X, Li Y, Lu L, Li B, He X (2015) Sex differences in peripheral mu-opioid receptor mediated analgesia in rat orofacial persistent pain model. PLoS One 10(3):e0122924. https://doi.org/10.1371/journal.pone.0122924
Caterina MJ, Leffler A, Malmberg AB, Martin WJ, Trafton J, Petersen-Zeitz KR, Koltzenburg M, Basbaum AI, Julius D (2000) Impaired nociception and pain sensation in mice lacking the capsaicin receptor. Science 288(5464):306–313. https://doi.org/10.1126/science.288.5464.306
Claiborne J, Nag S, Mokha SS (2006) Activation of opioid receptor like-1 receptor in the spinal cord produces sex-specific antinociception in the rat: estrogen attenuates antinociception in the female, whereas testosterone is required for the expression of antinociception in the male. J Neurosci 26(50):13048–13053. https://doi.org/10.1523/JNEUROSCI.4783-06.2006
Durga A, Sepahpanah F, Regozzi M, Hastings J, Crane DA (2011) Prevalence of testosterone deficiency after spinal cord injury. PM R 3(10):929–932. https://doi.org/10.1016/j.pmrj.2011.07.008
Ferrell WR, Wong BB, Lockhart JC, Ramsay JE (2004) Gender differences in regional cutaneous microcirculatory responses to capsaicin. Fundam Clin Pharmacol 18(2):195–200. https://doi.org/10.1111/j.1472-8206.2004.00218.x
Frot M, Feine JS, Bushnell MC (2004) Sex differences in pain perception and anxiety. A psychophysical study with topical capsaicin. Pain 108(3):230–236. https://doi.org/10.1016/j.pain.2003.11.017
Gazerani P, Andersen OK, Arendt-Nielsen L (2005) A human experimental capsaicin model for trigeminal sensitization. Gender-specific differences. Pain 118(1):155–163. https://doi.org/10.1016/j.pain.2005.08.009
Gazerani P, Andersen OK, Arendt-Nielsen L (2007) Site-specific, dose-dependent, and sex-related responses to the experimental pain model induced by intradermal injection of capsaicin to the foreheads and forearms of healthy humans. J Orofac Pain 21(4):289–302
Hartmann EM, Handwerker HO, Forster C (2015) Gender differences in itch and pain-related sensations provoked by histamine, cowhage and capsaicin. Acta Derm Venereol 95(1):25–30. https://doi.org/10.2340/00015555-1894
Jensen MT, Petersen KL (2006) Gender differences in pain and secondary hyperalgesia after heat/capsaicin sensitization in healthy volunteers. J Pain 7(3):211–217. https://doi.org/10.1016/j.jpain.2005.10.013
Kim YS, Chu Y, Han L, Li M, Li Z, Lavinka PC, Sun S, Tang Z, Park K, Caterina MJ, Ren K, Dubner R, Wei F, Dong X (2014) Central terminal sensitization of TRPV1 by descending serotonergic facilitation modulates chronic pain. Neuron 81(4):873–887. https://doi.org/10.1016/j.neuron.2013.12.011
Kobayashi H, Yoshiyama M, Zakoji H, Takeda M, Araki I (2009) Sex differences in the expression profile of acid-sensing ion channels in the mouse urinary bladder: a possible involvement in irritative bladder symptoms. BJU Int 104(11):1746–1751. https://doi.org/10.1111/j.1464-410X.2009.08658.x
Lee KS, Asgar J, Zhang Y, Chung MK, Ro JY (2013) The role of androgen receptor in transcriptional modulation of cannabinoid receptor type 1 gene in rat trigeminal ganglia. Neuroscience 254:395–403. https://doi.org/10.1016/j.neuroscience.2013.09.014
Lee KS, Zhang Y, Asgar J, Auh QS, Chung MK, Ro JY (2016) Androgen receptor transcriptionally regulates mu-opioid receptor expression in rat trigeminal ganglia. Neuroscience 331:52–61. https://doi.org/10.1016/j.neuroscience.2016.06.023
Loyd DR, Sun XX, Locke EE, Salas MM, Hargreaves KM (2012) Sex differences in serotonin enhancement of capsaicin-evoked calcitonin gene-related peptide release from human dental pulp. Pain 153(10):2061–2067. https://doi.org/10.1016/j.pain.2012.06.018
McGaraughty S, Chu KL, Brown BS, Zhu CZ, Zhong C, Joshi SK, Honore P, Faltynek CR, Jarvis MF (2008) Contributions of central and peripheral TRPV1 receptors to mechanically evoked and spontaneous firing of spinal neurons in inflamed rats. J Neurophysiol 100(6):3158–3166. https://doi.org/10.1152/jn.90768.2008
Montico F, Hetzl AC, Candido EM, Favaro WJ, Cagnon VH (2011) Hormonal therapy in the senescence: prostatic microenvironment structure and adhesion molecules. Micron 42(6):642–655. https://doi.org/10.1016/j.micron.2011.03.004
Nag S, Mokha SS (2009) Testosterone is essential for alpha(2)-adrenoceptor-induced antinociception in the trigeminal region of the male rat. Neurosci Lett 467(1):48–52. https://doi.org/10.1016/j.neulet.2009.10.016
Nguelefack TB, Dutra RC, Paszcuk AF, de Andrade EL, Calixto JB (2015) TRPV1 channel inhibition contributes to the antinociceptive effects of Croton macrostachyus extract in mice. BMC Complement Altern Med 15(1):293. https://doi.org/10.1186/s12906-015-0816-z
Niu KY, Zhang Y, Ro JY (2012) Effects of gonadal hormones on the peripheral cannabinoid receptor 1 (CB1R) system under a myositis condition in rats. Pain 153(11):2283–2291. https://doi.org/10.1016/j.pain.2012.07.037
Okun A, DeFelice M, Eyde N, Ren J, Mercado R, King T, Porreca F (2011) Transient inflammation-induced ongoing pain is driven by TRPV1 sensitive afferents. Mol Pain 7:4. https://doi.org/10.1186/1744-8069-7-4
Peroni RN, Orliac ML, Becu-Villalobos D, Huidobro-Toro JP, Adler-Graschinsky E, Celuch SM (2004) Sex-linked differences in the vasorelaxant effects of anandamide in vascular mesenteric beds: role of oestrogens. Eur J Pharmacol 493(1-3):151–160. https://doi.org/10.1016/j.ejphar.2004.04.031
Pogatzki-Zahn EM, Shimizu I, Caterina M, Raja SN (2005) Heat hyperalgesia after incision requires TRPV1 and is distinct from pure inflammatory pain. Pain 115(3):296–307. https://doi.org/10.1016/j.pain.2005.03.010
Ro JY, Lee JS, Zhang Y (2009) Activation of TRPV1 and TRPA1 leads to muscle nociception and mechanical hyperalgesia. Pain 144(3):270–277. https://doi.org/10.1016/j.pain.2009.04.021
Rosario ER, Aqeel R, Brown MA, Sanchez G, Moore C, Patterson D (2013) Hypothalamic-pituitary dysfunction following traumatic brain injury affects functional improvement during acute inpatient rehabilitation. J Head Trauma Rehabil 28(5):390–396. https://doi.org/10.1097/HTR.0b013e318250eac6
Szabo A, Helyes Z, Sandor K, Bite A, Pinter E, Nemeth J, Banvolgyi A, Bolcskei K, Elekes K, Szolcsanyi J (2005) Role of transient receptor potential vanilloid 1 receptors in adjuvant-induced chronic arthritis: in vivo study using gene-deficient mice. J Pharmacol Exp Ther 314(1):111–119. https://doi.org/10.1124/jpet.104.082487
Wang S, Joseph J, Diatchenko L, Ro JY, Chung MK (2016) Agonist-dependence of functional properties for common nonsynonymous variants of human transient receptor potential vanilloid 1. Pain 157(7):1515–1524. https://doi.org/10.1097/j.pain.0000000000000556
Wilkinson CW, Pagulayan KF, Petrie EC, Mayer CL, Colasurdo EA, Shofer JB, Hart KL, Hoff D, Tarabochia MA, Peskind ER (2012) High prevalence of chronic pituitary and target-organ hormone abnormalities after blast-related mild traumatic brain injury. Front Neurol 3:11. https://doi.org/10.3389/fneur.2012.00011
Yoshiyama M, Araki I, Kobayashi H, Zakoji H, Takeda M (2010) Functional roles of TRPV1 channels in lower urinary tract irritated by acetic acid: in vivo evaluations of the sex difference in decerebrate unanesthetized mice. Am J Phys Renal Phys 298(6):F1351–F1359. https://doi.org/10.1152/ajprenal.00695.2009
Zhang X, Zhang Y, Asgar J, Niu KY, Lee J, Lee KS, Schneider M, Ro JY (2014) Sex differences in mu-opioid receptor expression in trigeminal ganglia under a myositis condition in rats. Eur J Pain 18(2):151–161. https://doi.org/10.1002/j.1532-2149.2013.00352.x
Funding
These studies were supported by grants from the National Natural Science Foundation of China (81400530), the Technology Foundation for Selected Overseas Chinese Scholar, the Ministry of Personnel of China ([2014]240), and the Natural Science Foundation of Liaoning Province (201602811). The funders had no roles in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Bai, X., Zhang, X. & Zhou, Q. Effect of Testosterone on TRPV1 Expression in a Model of Orofacial Myositis Pain in the Rat. J Mol Neurosci 64, 93–101 (2018). https://doi.org/10.1007/s12031-017-1009-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-017-1009-7