Abstract
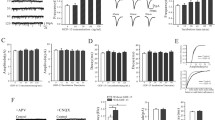
Vascular endothelial growth factor (VEGF) exerts both neuroprotective and proinflammatory effects in the brain, depending on the VEGF (A–E) and VEGF receptor (VEGFR1–3) types involved. Neonatal monosodium glutamate (MSG) treatment triggers an excitotoxic degenerative process associated with several neuropathological conditions, and VEGF messenger RNA (mRNA) expression is increased at postnatal day (PD) 14 in rat hippocampus (Hp) following the treatment. The aim of this work was to establish the changes in immunoreactivity to VEGF-A, VEGF-B, VEGFR-1 and VEGFR-2 proteins induced by neonatal MSG treatment (4 g/kg, subcutaneous, at PD1, 3, 5 and 7) in the cerebral motor cortex (CMC) and Hp. Samples collected from PD2 to PD60 from control and MSG-treated male Wistar rats were assessed by western blotting for each protein. Considering that immunoreactivity measured by western blotting is related to the protein expression level, we found that each protein in each cerebral region has a specific expression profile throughout the studied ages, and all profiles were differentially modified by MSG. Specifically, neonatal MSG treatment significantly increased the immunoreactivity to the following: (1) VEGF-A at PD8–PD10 in the CMC and at PD6–PD8 in the Hp; (2) VEGF-B at PD2, PD6 and PD10 in the CMC and at PD8–PD9 in the Hp; and (3) VEGFR-2 at PD6–PD8 in the CMC and at PD21–PD60 in the Hp. Also, MSG significantly reduced the immunoreactivity to the following: (1) VEGF-B at PD8–PD9 and PD45–PD60 in the CMC; and (2) VEGFR-1 at PD4–PD6 and PD14–PD21 in the CMC and at PD4, PD9–PD10 and PD60 in the Hp. Our results indicate that VEGF-mediated signalling is involved in the excitotoxic process triggered by neonatal MSG treatment and should be further characterized.




Similar content being viewed by others
References
Beas-Zarate C, Rivera-Huizar SV, Martinez-Contreras A, Feria-Velasco A, Armendariz-Borunda J (2001) Changes in NMDA-receptor gene expression are associated with neurotoxicity induced neonatally by glutamate in the rat brain. Neurochem Int 39(1):1–10. doi:10.1016/S0197-0186(01)00008-0
Beas-Zarate C, Ureña-Guerrero ME, Flores-Soto M, Armendariz-Borunda J, Ortuño-Sahagun D (2007) The expression and binding of kainate receptors is modified in different brain regions by glutamate neurotoxicity during postnatal rat development. Int J Dev Neurosci 25(1):53–61. doi:10.1016/j.ijdevneu.2006.10.001
Brockington A, Lewis C, Wharton S, Shaw PJ (2004) Vascular endothelial growth factor and the nervous system. Neuropathol Appl Neurobiol 30(5):427–446. doi:10.1111/j.1365-2990.2004.00600.x
Bry M, Kivelä R, Leppänen VM, Alitalo K (2014) Vascular endothelial growth factor-B in physiology and disease. Physiol Rev 94(3):779–794. doi:10.1152/physrev.00028.2013
Chaparro-Huerta V, Rivera-Cervantes MC, Torres-Mendoza BM, Beas-Zarate C (2002) Neuronal death and tumor necrosis factor-alpha response to glutamate-induced excitotoxicity in the cerebral cortex of neonatal rats. Neurosci Lett 333(2):95–98. doi:10.1016/S0304-3940(02)01006-6
Chaparro-Huerta V, Rivera-Cervantes MC, Flores-Soto ME, Gomez-Pinedo U, Beas-Zarate C (2005) Proinflammatory cytokines and apoptosis following glutamate-induced excitotoxicity mediated by p38 MAPK in the hippocampus of neonatal rats. J Neuroimmunol 165(1–2):53–62. doi:10.1016/j.jneuroim.2005.04.025
Chaparro-Huerta V, Flores-Soto ME, Gudiño-Cabrera G, Rivera-Cervantes MC, Bitzer-Quintero OK, Beas-Zarate C (2008) Role of p38 MAPK and pro-inflammatory cytokines expression in glutamate-induced neuronal death of neonatal rats. Int J Dev Neurosci 26(5):487–495. doi:10.1016/j.ijdevneu.2008.02.008
Cho Y, Shin JE, Ewan EE, Oh YM, Pita-Thomas W, Cavalli V (2015) Activating injury-responsive genes with hypoxia enhances axon regeneration through neuronal HIF-1α. Neuron 88(4):720–734. doi:10.1016/j.neuron.2015.09.050
Croll SD, Goodman JH, Scharfman HE (2004) Vascular endothelial growth factor (VEGF) in seizures: a double-edged sword. Adv Exp Med Biol 548:57–68. doi:10.1016/j.yebeh.2016.12.007
Cross MJ, Dixelius J, Matsumoto T, Claesson-Welsh L (2003) VEGF-receptor signal transduction. Trends Biochem Sci 28(9):488–494. doi:10.1016/S0968-0004(03)00193-2
De Rossi P, Harde E, Dupuis JP, Martin L, Chounlamountri N, Bardin M, Watrin C, Benetollo C, Pernet-Gallay K, Luhmann HJ, Honnorat J, Malleret G, Groc L, Acker-Palmer A, Salin PA, Meissirel C (2016) A critical role for VEGF and VEGFR2 in NMDA receptor synaptic function and fear-related behavior. Mol Psychiatry 21(12):1768–1780. doi:10.1038/mp.2015.195
Dhondt J, Peeraer E, Verheyen A, Nuydens R, Buysschaert I, Poesen K, Van Geyte K, Beerens M, Shibuya M, Haigh JJ, Meert T, Carmeliet P, Lambrechts D (2011) Neuronal FLT1 receptor and its selective ligand VEGF-B protect against retrograde degeneration of sensory neurons. FASEB J 25(5):1461–1473. doi:10.1096/fj.10-170944
Elfving B, Jakobsen JL, Madsen JC, Wegener G, Müller HK (2015) Chronic restraint stress increases the protein expression of VEGF and its receptor VEGFR-2 in the prefrontal cortex. Synapse 69(4):190–194. doi:10.1002/syn.21808
Enholm B, Paavonen K, Ristimäki A, Kumar V, Gunji Y, Klefstrom J, Kivinen L, Laiho M, Olofsson B, Joukov V, Eriksson U, Alitalo K (1997) Comparison of VEGF, VEGF-B, VEGF-C and Ang-1 mRNA regulation by serum, growth factors, oncoproteins and hypoxia. Oncogene 14(20):2475–2483
Falk T, Yue X, Zhang S, McCourt AD, Yee BJ, Gonzalez RT, Sherman SJ (2011) Vascular endothelial growth factor-B is neuroprotective in an in vivo rat model of Parkinson’s disease. Neurosci Lett 496(1):43–47. doi:10.1016/j.neulet.2011.03.088
Fournier NM, Duman RS (2012) Role of vascular endothelial growth factor in adult hippocampal neurogenesis: implications for the pathophysiology and treatment of depression. Behav Brain Res 227(2):440–449. doi:10.1016/j.bbr.2011.04.022
Guaiquil VH, Pan Z, Karagianni N, Fukuoka S, Alegre G, Rosenblatt MI (2014) VEGF-B selectively regenerates injured peripheral neurons and restores sensory and trophic functions. Proc Natl Acad Sci U S A 111(48):17272–17277. doi:10.1073/pnas.1407227111
Greenberg DA, Jin K (2013) Vascular endothelial growth factors (VEGFs) and stroke. Cell Mol Life Sci 70(10):1753–1761. doi:10.1007/s00018-013-1282-8
Han W, Song X, He R, Li T, Cheng L, Xie L, Chen H, Jiang L (2017) VEGF regulates hippocampal neurogenesis and reverses cognitive deficits in immature rats after status epilepticus through the VEGF R2 signaling pathway. Epilepsy Behav 68:159–167. doi:10.1016/j.yebeh.2016.12.007
Hao T, Rockwell P (2013) Signaling through the vascular endothelial growth factor receptor VEGFR-2 protects hippocampal neurons from mitochondrial dysfunction and oxidative stress. Free Radic Biol Med 63:421–431. doi:10.1016/j.freeradbiomed.2013.05.036
Hernandez-Ojeda M, Ureña-Guerrero ME, Gutierrez-Barajas PE, Cardenas-Castillo JA, Camins A, Beas-Zarate C (2017) KB-R7943 reduces 4-aminopyridine-induced epileptiform activity in adult rats after neuronal damage induced by neonatal monosodium glutamate treatment. J Biomed Sci 24(1):27. doi:10.1186/s12929-017-0335-y
Keifer OP, O’Connor DM, Boulis NM (2014) Gene and protein therapies utilizing VEGF for ALS. Pharmacol Ther 141(3):261–271. doi:10.1016/j.pharmthera.2013.10.009
Kieran MW, Kalluri R, Cho YJ (2012) The VEGF pathway in cancer and disease: response, resistance, and the path forward. Cold Spring Harb Perspect Med 2(12):a006593. doi:10.1101/cshperspect.a006593
Koch S, Claesson-Welsh L (2012) Signal transduction by vascular endothelial growth factor receptors. Cold Spring Harb Perspect Med 2(7):a006502. doi:10.1101/cshperspect.a006502
Krum JM, Mani N, Rosenstein JM (2008) Roles of the endogenous VEGF receptors flt-1 and flk-1 in astroglial and vascular remodeling after brain injury. Exp Neurol 212(1):108–117. doi:10.1016/j.expneurol.2008.03.019
Kubo T, Kohira R, Okano T, Ishikawa K (1993) Neonatal glutamate can destroy the hippocampal CA1 structure and impair discrimination learning in rats. Brain Res 616(1–2):311–314. doi:10.1016/0006-8993(93)90223-A
Lee C, Agoston DV (2010) Vascular endothelial growth factor is involved in mediating increased de novo hippocampal neurogenesis in response to traumatic brain injury. J Neurotrauma 27(3):541–553. doi:10.1089/neu.2009.0905
Li X, Kumar A, Zhang F, Lee C, Tang Z (2012) Complicated life, complicated VEGF-B. Trends Mol Med 18(2):119–127. doi:10.1016/j.molmed.2011.11.006
Licht T, Keshet E (2013) Delineating multiple functions of VEGF-A in the adult brain. Cell Mol Life Sci 70(10):1727–1737. doi:10.1007/s00018-013-1280-x
Lladó J, Tolosa L, Olmos G (2013) Cellular and molecular mechanisms involved in the neuroprotective effects of VEGF on motoneurons. Front Cell Neurosci 7:181. doi:10.3389/fncel.2013.00181
Lopez-Perez SJ, Vergara P, Ventura-Valenzuela JP, Ureña-Guerrero ME, Segovia J, Beas-Zárate C (2005) Modification of dopaminergic markers expression in the striatum by neonatal exposure to glutamate during development. Int J Dev Neurosci 23(4):335–342. doi:10.1016/j.ijdevneu.2004.12.010
Lopez-Perez SJ, Ureña-Guerrero ME, Morales-Villagran A (2010) Monosodium glutamate neonatal treatment as a seizure and excitotoxic model. Brain Res 1317:246–256. doi:10.1016/j.brainres.2009.12.054
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193(1):265–275
Mehta A, Prabhakar M, Kumar P, Deshmukh R, Sharma PL (2013) Excitotoxicity: bridge to various triggers in neurodegenerative disorders. Eur J Pharmacol 698(1–3):6–18. doi:10.1016/j.ejphar.2012.10.032
Mendonça MC, Soares ES, Stávale LM, Rapôso C, Coope A, Kalapothakis E, da Cruz-Höfling MA (2013) Expression of VEGF and Flk-1 and Flt-1 receptors during blood-brain barrier (BBB) impairment following Phoneutria nigriventer spider venom exposure. Toxins 5(12):2572–2588. doi:10.3390/toxins5122572
Nicoletti JN, Shah SK, McCloskey DP, Goodman JH, Elkady A, Atassi H, Hylton D, Rudge JS, Scharfman HE, Croll SD (2008) Vascular endothelial growth factor is up-regulated after status epilepticus and protects against seizure-induced neuronal loss in hippocampus. Neuroscience 151(1):232–241. doi:10.1016/j.neuroscience.2007.09.083
Nowacka MM, Obuchowicz E (2012) Vascular endothelial growth factor (VEGF) and its role in the central nervous system: a new element in the neurotrophic hypothesis of antidepressant drug action. Neuropeptides 46(1):1–10. doi:10.1016/j.npep.2011.05.005
Olney JW (2003) Excitotoxicity, apoptosis and neuropsychiatric disorders. Curr Opin Pharmacol 3(1):101–109. doi:10.1016/S1471-4892(02)00002-4
Ortuño-Sahagún D, Beas-Zárate C, Adame-Gonzalez G, Feria-Velasco A (1997) Effect of L-glutamate on cholinergic neurotransmission in various brain regions and during the development of rats, when administered perinatally. Neurochem Int 31(5):683–692. doi:10.1016/S0197-0186(97)00032-6
Park YS, Cho JH, Kim IH, Cho GS, Cho JH, Park JH, Ahn JH, Chen BH, Shin BN, Shin MC, Tae HJ, Cho YS, Lee YL, Kim YM, Won MH, Lee JC (2014) Effects of ischemic preconditioning on VEGF and pFlk-1 immunoreactivities in the gerbil ischemic hippocampus after transient cerebral ischemia. J Neurol Sci 347(1–2):179–187. doi:10.1016/j.jns.2014.09.044
Pronto-Laborinho AC, Pinto S, de Carvalho M (2014) Roles of vascular endothelial growth factor in amyotrophic lateral sclerosis. Biomed Res Int 2014:947513. doi:10.1155/2014/947513
Rivera-Cervantes MC, Flores-Soto ME, Chaparro-Huerta V, Reyes-Gómez J, Feria-Velasco A, Schliebs R, Beas-Zárate C (2009) Changes in hippocampal NMDA-R subunit composition induced by exposure of neonatal rats to L-glutamate. Int J Dev Neurosci 27(2):197–204. doi:10.1016/j.ijdevneu.2008.09.006
Roy H, Bhardwaj S, Ylä-Herttuala S (2006) Biology of vascular endothelial growth factors. FEBS Lett 580(12):2879–2887. doi:10.1016/j.febslet.2006.03.087
Ruiz de Almodovar C, Lambrechts D, Mazzone M, Carmeliet P (2009) Role and therapeutic potential of VEGF in the nervous system. Physiol Rev 89:607–648. doi:10.1007/s00018-013-1283-7
Schoch HJ, Fischer S, Marti HH (2002) Hypoxia-induced vascular endothelial growth factor expression causes vascular leakage in the brain. Brain 125(Pt 11):2549–2557
Shibuya M (2013) Vascular endothelial growth factor and its receptor system: physiological functions in angiogenesis and pathological roles in various diseases. J Biochem 153(1):13–19. doi:10.1093/jb/mvs136
Shibuya M (2014) VEGF-VEGFR signals in health and disease. Biomol Ther 22(1):1–9. doi:10.4062/biomolther.2013.113
Sköld MK, von Gertten C, Sandberg-Nordqvist AC, Mathiesen T, Holmin S (2005) VEGF and VEGF receptor expression after experimental brain contusion in rat. J Neurotrauma 22(3):353–367. doi:10.1089/neu.2005.22.353
Sun Y, Jin K, Childs JT, Xie L, Mao XO, Greenberg DA (2006) Vascular endothelial growth factor-B (VEGFB) stimulates neurogenesis: evidence from knockout mice and growth factor administration. Dev Biol 289(2):329–335. doi:10.1016/j.ydbio.2005.10.016
Sun FJ, Wei YJ, Li S, Guo W, Chen X, Liu SY, He JJ, Yin Q, Yang H, Zhang CQ (2016) Elevated expression of VEGF-C and its receptors, VEGFR-2 and VEGFR-3, in patients with mesial temporal lobe epilepsy. J Mol Neurosci 59(2):241–250. doi:10.1007/s12031-016-0714-y
Tovar-y-Romo LB, Tapia R (2012) Delayed administration of VEGF rescues spinal motor neurons from death with a short effective time frame in excitotoxic experimental models in vivo. ASN Neuro 4(2):121–129. doi:10.1042/AN20110057
Tovar-y-Romo LB, Ramirez-Jarquin UN, Lazo-Gomez R, Tapia R (2014) Trophic factors as modulators of motor neuron physiology and survival: implications for ALS therapy. Front Cell Neurosci 8:61. doi:10.3389/fncel.2014.00061
Ureña-Guerrero ME, Lopez-Perez SJ, Beas-Zarate C (2003) Neonatal monosodium glutamate treatment modifies glutamic acid decarboxylase activity during rat brain postnatal development. Neurochem Int 42(4):269–276. doi:10.1016/S0197-0186(02)00131-6
Ureña-Guerrero ME, Orozco-Suarez S, Lopez-Perez SJ, Flores-Soto ME, Beas-Zarate C (2009) Excitotoxic neonatal damage induced by monosodium glutamate reduces several GABAergic markers in the cerebral cortex and hippocampus in adulthood. Int J Dev Neurosci 27(8):845–855. doi:10.1016/j.ijdevneu.2009.07.01
Vazquez-Valls E, Flores-Soto ME, Chaparro-Huerta V, Torres-Mendoza BM, Gudiño-Cabrera G, Rivera-Cervantes MC, Pallas M, Camins A, Armendáriz-Borunda J, Beas-Zárate C (2011) HIF-1α expression in the hippocampus and peripheral macrophages after glutamate-induced excitotoxicity. J Neuroimmunol 238(1–2):12–18. doi:10.1016/j.jneuroim.2011.06.001
Vieira JM, Ruhrberg C, Schwarz Q (2010) VEGF receptor signaling in vertebrate development. Organ 6(2):97–106
Wang WY, Dong JH, Liu X, Wang Y, Ying GX, Ni ZM, Zhou CF (2005) Vascular endothelial growth factor and its receptor Flk-1 are expressed in the hippocampus following entorhinal deafferentation. Neuroscience 134(4):1167–1178. doi:10.1016/j.neuroscience.2005.04.064
Yeh WL, Lu DY, Lin CJ, Liou HC, Fu WM (2007) Inhibition of hypoxia-induced increase of blood-brain barrier permeability by YC-1 through the antagonism of HIF-1alpha accumulation and VEGF expression. Mol Pharmacol 72(2):440–449. doi:10.1124/mol.107.036418
Zhang J, Li X, Chai S, Wang X (2015) Neuroprotective effect of lentivirus mediated VEGF on rat model with cerebral ischemic injury. Int J Clin Exp Med 8(3):4094–4100
Acknowledgements
This study was partially supported by the National Council of Science and Technology of Mexico (CONACYT) through Grant No. 177594 given to CBZ, GGC and MEUG as members of the same research group and through Scholarship No. 510027 given to JLCC. In addition, regular financial support given to MEUG as a researcher of CUCBA-University of Guadalajara during the last 3 years was applied in this study.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
ESM 1
(DOCX 21 kb)
Rights and permissions
About this article
Cite this article
Castañeda-Cabral, J.L., Beas-Zarate, C., Gudiño-Cabrera, G. et al. Glutamate Neonatal Excitotoxicity Modifies VEGF-A, VEGF-B, VEGFR-1 and VEGFR-2 Protein Expression Profiles During Postnatal Development of the Cerebral Cortex and Hippocampus of Male Rats. J Mol Neurosci 63, 17–27 (2017). https://doi.org/10.1007/s12031-017-0952-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-017-0952-7