Abstract
Purpose
Prior work has shown that higher circulating concentrations of fibroblast growth factor-21 (FGF-21) are associated with an increased likelihood of developing colorectal cancer. We conducted a prospective study to assess the relationship between circulating FGF-21 and odds of developing early neoplastic lesions in the colorectum.
Methods
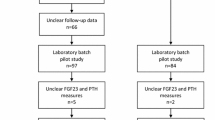
A total of 94 study participants were included from the ursodeoxycholic acid (UDCA) trial, a phase III, randomized, double-blind, placebo-controlled clinical trial of the effect of 8–10 mg/kg of body weight UDCA vs. placebo. Logistic regression analyses were conducted to evaluate the association between baseline FGF-21 concentrations and odds of developing a metachronous adenoma.
Results
Of the characteristics compared across tertiles of FGF-21 concentrations, including age, race, sex, BMI, and other variables, only a previous personal history of colorectal polyps prior to entry into the UDCA trial was statistically significantly related to FGF-21 levels, with a proportion of 26.7%, 56.7%, and 50.0% across the first, second, and third tertiles, respectively (p < 0.05). Higher circulating concentrations of FGF-21 were statistically significantly associated with greater odds of developing a metachronous colorectal adenoma. After adjusting for potential confounders and when compared with the lowest tertile of FGF-21, the adjusted ORs (95% CIs) for metachronous colorectal adenoma in the second and third tertiles were 4.72 (95% CI, 1.42–15.72) and 3.82 (95% CI, 1.15–12.68), respectively (p trend < 0.05).
Conclusion
Our results reveal for the first time that, in addition to a recently discovered association with colorectal cancer, circulating FGF-21 concentrations are significantly and directly associated with odds of developing metachronous colorectal adenoma.
Similar content being viewed by others
Data Availability
The data may be requested from the principal investigator and may be subject to IRB approval.
References
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2020. CA Cancer J Clin. 2020;70:7–30.
Siegel RL, Miller KD, Fedewa SA, Ahnen DJ, Meester RG, Barzi A, et al. Colorectal cancer statistics, 2017. CA Cancer J Clin. 2017;67(3):177–93.
Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144(5):646–74.
Lasry A, Zinger A, Ben-Neriah Y. Inflammatory networks underlying colorectal cancer. Nat Immunol. 2016;17(3):230–40.
Dela Cruz MD, Ledbetter S, Chowdhury S, Tiwari AK, Momi N, Wali RK, et al. Metabolic reprogramming of the premalignant colonic mucosa is an early event in carcinogenesis. Oncotarget. 2017;8(13):20543–57.
Qian J, Tikk K, Weigl K, Balavarca Y, Brenner H. Fibroblast growth factor 21 as a circulating biomarker at various stages of colorectal carcinogenesis. Br J Cancer. 2018;119(11):1374–82.
Degirolamo C, Sabba C, Moschetta A. Therapeutic potential of the endocrine fibroblast growth factors FGF19, FGF21 and FGF23. Nat Rev Drug Discov. 2016;15:51–69.
Knott ME, Minatta JN, Roulet L, Gueglio G, Pasik L, Ranuncolo SM, et al. Circulating fibroblast growth factor 21 (Fgf21) as diagnostic and prognostic biomarker in renal cancer. J Mol Biomarker Diag. 2016;1(Suppl 2):015. https://doi.org/10.4172/2155-9929.S2-015.
Knott ME, Ranuncolo SM, Nuñez M, Armanasco E, Puricelli LI, De Lorenzo MS. Levels of fibroblast growth factor 21 (FGF21) in serum as diagnostic biomarker in patients with breast cancer. Proceedings: AACR 106th Annual Meeting 2015; April 18-22, 2015; Philadelphia, PA. Cancer Res. 2015;75(15 Suppl):Abstract no.1577. https://doi.org/10.1158/1538-7445.AM2015-1577.
Harlid S, Myte R, Van Guelpen B (2017). The metabolic syndrome, inflammation, and colorectal cancer risk: an evaluation of large panels of plasma protein markers using repeated, prediagnostic samples. Med Inflamm. https://doi.org/10.1155/2017/4803156. Epub 2017 Mar 22.
Alberts DS, Martinez ME, Hess LM, Einspahr JG, Green SB, Bhattacharyya AK, et al. Phase III trial of ursodeoxycholic acid to prevent colorectal adenoma recurrence. J Natl Cancer Inst. 2005;97(11):846–53.
Jess T, Frisch M, Simonsen J. Trends in overall and cause-specific mortality among patients with inflammatory bowel disease from 1982 to 2010. Clin Gastroenterol Hepatol. 2013;11(1):43–8.
Burn J, Gerdes A-M, Macrae F, Mecklin J-P, Moeslein G, Olschwang S, et al. Long-term effect of aspirin on cancer risk in carriers of hereditary colorectal cancer: an analysis from the CAPP2 randomised controlled trial. Lancet. 2011;378(9809):2081–7.
Labayle D, Fischer D, Vielh P, Drouhin F, Pariente A, Bories C, et al. Sulindac causes regression of rectal polyps in familial adenomatous polyposis. Gastroenterol. 1991;101(3):635–9.
Itoh N. Hormone-like (endocrine) Fgfs: their evolutionary history and roles in development, metabolism, and disease. Cell Tissue Res. 2010;342(1):1–11.
Youm Y-H, Horvath TL, Mangelsdorf DJ, Kliewer SA, Dixit VD. Prolongevity hormone FGF21 protects against immune senescence by delaying age-related thymic involution. PNAS. 2016;113(4):1026–31.
Hanks LJ, Gutiérrez OM, Bamman MM, Ashraf A, McCormick KL, Casazza K. Circulating levels of fibroblast growth factor-21 increase with age independently of body composition indices among healthy individuals. J Clin Transl. 2015;2(2):77–82.
Amersi F, Agustin M, Ko CY. Colorectal cancer: epidemiology, risk factors, and health services. Clin Colon Rectal Surg. 2005;18(3):133–40. https://doi.org/10.1055/s-2005-916274.
Hamoya T, Fujii G, Miyamoto S, Takahashi M, Totsuka Y, Wakabayashi K, et al. Effects of NSAIDs on the risk factors of colorectal cancer: a mini review. Genes Environ. 2016;38:6. https://doi.org/10.1186/s41021-016-0033-0.
Rothwell PM, Wilson M, Elwin C-E, Norrving B, Algra A, Warlow CP, et al. Long-term effect of aspirin on colorectal cancer incidence and mortality: 20-year follow-up of five randomised trials. Lancet. 2010;76(9754):1741–50.
Yang C, Lu W, Lin T, You P, Ye M, Huang Y, et al. Activation of liver FGF21 in hepatocarcinogenesis and during hepatic stress. BMC Gastroenterol. 2013;13(1):67.
Markan KR, Naber MC, Ameka MK, Anderegg MD, Mangelsdorf DJ, Kliewer SA, et al. Circulating FGF21 is liver derived and enhances glucose uptake during refeeding and overfeeding. Diabetes. 2014;63(12):4057–63.
Akyol M, Alacacioglu A, Demir L, Kucukzeybek Y, Yildiz Y, Gumus Z, et al. The alterations of serum FGF-21 levels, metabolic and body composition in early breast cancer patients receiving adjuvant endocrine therapy. Cancer Biomark. 2017;18(4):441–9.
Chavez AO, Molina-Carrion M, Abdul-Ghani MA, Folli F, DeFronzo RA, Tripathy D. Circulating fibroblast growth factor-21 is elevated in impaired glucose tolerance and type 2 diabetes and correlates with muscle and hepatic insulin resistance. Diabetes Care. 2009;32(8):1542–6.
Mraz M, Bartlova M, Lacinova Z, Michalsky D, Kasalicky M, Haluzikova D, et al. Serum concentrations and tissue expression of a novel endocrine regulator fibroblast growth factor-21 in patients with type 2 diabetes and obesity. Clin Endocrinol. 2009;71(3):369–75.
Funding
This study was funded by the National Cancer Institute Cancer Center Support (Grant P30CA023074; NIH/NCI RO1CA140285; NIH/NCI; NIH/NCI P01CA041108).
Author information
Authors and Affiliations
Contributions
Dr. Ana Florea, Dr. Elizabeth Jacobs, and Dr. Peter W. Jurutka were responsible for the design, acquisition of data, and interpretation of data. Dr. Ana Florea and Dr. Elizabeth Jacobs were responsible for data analyses. Dr. Lindsay Kohler, Dr. Robin Harris, and Dr. Yann Klimentidis made substantial contributions to manuscript preparation and interpretation of data. All authors assisted with the drafting and critical revisions of the article by making important intellectual contributions, approved the work to be published, and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethics Approval
This study has been approved by the University of Arizona Institutional Review Board.
Consent to Participate
All participants provided written and signed informed consent forms.
Consent to Publish
All participants provided written and signed informed consent forms.
Code Availability
No special or custom code was used.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Florea, A., Harris, R.B., Klimentidis, Y.C. et al. Circulating Fibroblast Growth Factor-21 and Risk of Metachronous Colorectal Adenoma. J Gastrointest Canc 52, 940–946 (2021). https://doi.org/10.1007/s12029-020-00515-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12029-020-00515-8