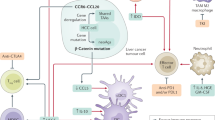
Abstract
Hepatocellular carcinoma has still been one of the cancer with increasing incidence and highest mortality rate in the world. Although many new promising developments have been defined in hepatocarcinogenesis, with a short survival the treatment of patients with advanced hepatocellular carcinoma is an emerging issue. On the recent decade, only one anti-angiogenic agent sorafenib improved overall survival with costing a hardly manageable toxicity. Novel immunotherapeutic agents, especially immune checkpoint inhibitors are on the edge of more effective but less toxic treatments for these patients. In this article the activity of immune checkpoint inhibitors, anti-CTLA-4 and anti-PD1 antibodies for the treatment of patients with advanced hepatocellular cancer will be reviewed.
Similar content being viewed by others
References
International Agency for Research on Cancer. GLOBOCAN. Estimated Global Cancer Incidence, Mortality and Prevalence Worldwide in. 2012:2012. http://globocan.iarc.fr/Pages/fact_sheets_population.aspx. Accessed August 04, 2017
Whittaker S, et al. The role of signaling pathways in the development and treatment of hepatocellular carcinoma. Oncogene. 2010;29:4989–5005.
Llovet JM, et al. Design and endpoints of clinical trials in hepatocellular carcinoma. J Natl Cancer Inst. 2008;100:698–711.
Subramanian S, et al. A review of hepatocellular carcinoma (HCC) staging systems. Chin Clin Oncol. 2013;2:33.
Llovet JM, et al. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med. 2008;359:378–90.
Kane RC, et al. Sorafenib for the treatment of unresectable hepatocellular carcinoma. Oncologist. 2009;14:95–100.
Cheng A-L et al. Phase III trial of lenvatinib (LEN) vs sorafenib (SOR) in first-line treatment of patients (pts) with unresectable hepatocellular carcinoma (uHCC). J Clin Oncol 35, 2017 (suppl; abstr 4001).
Zhu AX, et al. Ramucirumab versus placebo as second-line treatment in patients with advanced hepatocellular carcinoma following first-line therapy with sorafenib (REACH): a randomised, double-blind, multicentre, phase 3 trial. Lancet Oncol. 2015;16:859–70.
Bruix J, et al. Efficacy and safety of regorafenib versus placebo in patients with hepatocellular carcinoma (HCC) progressing on sorafenib: results of the international, randomized phase 3 RESORCE trial. Annals of Oncol, 27, 2016 suppl LBA-3, ii140–ii141.
Chen DS, Mellman I. Oncology meets immunology: the cancer-immunity cycle. Immunity. 2013;39(1):1–10.
Breous E, Thimme R. Potential of immunotherapy for hepatocellular carcinoma. J Hepatol. 2011;54(4):830–4.
Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer. 2012;12(4):252–64.
Weber J. Immune checkpoint proteins: a new therapeutic paradigm for cancer—preclinical background: CTLA-4 and PD-1 blockade. Semin Oncol. 2010;37(5):430–9.
Mizukoshi E, Nakamoto Y, Arai K, et al. Comparative analysis of various tumor-associated antigen-specific t-cell responses in patients with hepatocellular carcinoma. Hepatology. 2011;53:1206–16.
Flecken T, Schmidt N, Hild S, et al. Immunodominance and functional alterations of tumor-associated antigen-specific CD8+ T-cell responses in hepatocellular carcinoma. Hepatology. 2014;59:1415–26.
Umemoto Y, et al. Prognostic impact of programmed cell death 1 ligand 1 expression in human leukocyte antigen class I-positive hepatocellular carcinoma after curative hepatectomy. J Gastroenterol. 2015;50(1):65–75.
Grakoui A, et al. Turning on the off switch: regulation of anti-viral T cell responses in the liver by the PD-1/PD-L1 pathway. J Hepatol. 2006;45(4):468–72.
Schmidt J, et al. T-cell responses in hepatitis B and C virus infection: similarities and differences. Emerg Microbes Infect. 2013;2(3):e15.
Sangro B, Gomez-Martin C, de la Mata M, et al. A clinical trial of CTLA-4 blockade with tremelimumab in patients with hepatocellular carcinoma and chronic hepatitis C. J Hepatol. 2013;59:81–8.
Duffy AG, Ulahannan SV, Makorova-Rusher O, et al. Tremelimumab in combination with ablation in patients with advanced hepatocellular carcinoma. J Hepatol. 2017;66:545–51.
El-Khoueiry AB, et al. Phase 1/2 safety and antitumor activity of nivolumab in patients with advanced hepatocellular carcinoma: interim analysis of the CheckMate 040 dose-escalation study. Poster presentation at ASCO. 2016; poster 4012
Sangro B, Melero I, Yau T et al. Safety and antitumor activity of nivolumab in patients with advanced hepatocellular carcinoma: interim analysis of dose-expansion cohorts from the phase 1/2 checkmate 040 study. In:Presented at the 52nd Annual Meeting of the American Society of Clinical Oncology (ASCO); June 3–7, 2016; Chicago, IL, USA, poster 4078.
Crocenzi T S. Nivolumab (nivo) in sorafenib (sor)-naive and -experienced pts with advanced hepatocellular carcinoma (HCC): CheckMate 040 study. J Clin Oncol 35, 2017 (suppl; abstr 4013).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sahin, B. Enlighting the Shadow for Advanced Hepatocellular Carcinoma: Immunotherapy with Immune Checkpoint Inhibitors. J Gastrointest Canc 48, 288–290 (2017). https://doi.org/10.1007/s12029-017-9996-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12029-017-9996-8