Abstract
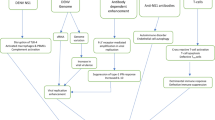
Following the global spread of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection, the importance of investigation of the pathogenesis and immunological characteristics of COVID-19 became quite clear. Currently, there are reports indicating that COVID-19 is able to induce autoimmune responses. Abnormal immune reactions are a cornerstone in the pathogenicity of both conditions. Detection of autoantibodies in COVID-19 patients may suggest a link between COVID-19 and autoimmunity. In this study, we focused on the similarities and possible differences between COVID-19 and autoimmune disorders to explore the relationship between them. Comparing the pathogenicity of SARS-CoV-2 infection with autoimmune conditions revealed significant immunological properties of COVID-19 including the presence of several autoantibodies, autoimmunity-related cytokines, and cellular activities that could be useful in future clinical studies aiming at managing this pandemic.

Similar content being viewed by others
References
Wang C, Pan R, Wan X, Tan Y, Xu L, Ho CS, et al. Immediate psychological responses and associated factors during the initial stage of the 2019 coronavirus disease (COVID-19) epidemic among the general population in China. Int J Environ Res Public Health. 2020;17(5):1729.
Mahase E. China coronavirus: WHO declares international emergency as death toll exceeds 200. BMJ Brit Med J (Online). 2020;368.
Thienemann F, Pinto F, Grobbee DE, Boehm M, Bazargani N, Ge J, et al. World heart federation briefing on prevention: Coronavirus disease 2019 (COVID-19) in low-income countries. Global Heart. 2020;15(1).
Rokni M, Hamblin MR, Rezaei N. Cytokines and COVID-19: Friends or foes? Hum Vaccin Immunother. 2020;16(10):2363–5.
Chetaille Nézondet AL, Poubelle PE, Pelletier M. The evaluation of cytokines to help establish diagnosis and guide treatment of autoinflammatory and autoimmune diseases. J Leukoc Biol. 2020;108(2):647–57.
Getts DR, Chastain EM, Terry RL, Miller SD. Virus infection, antiviral immunity, and autoimmunity. Immunol Rev. 2013;255(1):197–209.
Ehrenfeld M, Tincani A, Andreoli L, Cattalini M, Greenbaum A, Kanduc D, et al. Covid-19 and autoimmunity. Autoimmun Rev. 2020;19(8):102597.
Ruan Q, Yang K, Wang W, Jiang L, Song J. Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China. Intensive Care Med. 2020;46(5):846–8.
Wang J, Jiang M, Chen X, Montaner LJ. Cytokine storm and leukocyte changes in mild versus severe SARS-CoV-2 infection: Review of 3939 COVID-19 patients in China and emerging pathogenesis and therapy concepts. J Leukoc Biol. 2020;108(1):17–41.
Rodríguez Y, Novelli L, Rojas M, De Santis M, Acosta-Ampudia Y, Monsalve DM, et al. Autoinflammatory and autoimmune conditions at the crossroad of COVID-19. J Autoimmun. 2020;114:102506.
Ragab D, Salah Eldin H, Taeimah M, Khattab R, Salem R. The COVID-19 cytokine storm: What we know so far. Front Immunol. 2020;11:1446.
Mehta P, McAuley DF, Brown M, Sanchez E, Tattersall RS, Manson JJ. COVID-19: Consider cytokine storm syndromes and immunosuppression. Lancet. 2020;395(10229):1033–4.
Tay MZ, Poh CM, Rénia L, MacAry PA, Ng LFP. The trinity of COVID-19: Immunity, inflammation and intervention. Nat Rev Immunol. 2020;20(6):363–74.
Hussein HM, Rahal EA. The role of viral infections in the development of autoimmune diseases. Crit Rev Microbiol. 2019;45(4):394–412.
Harley JB, James JA. Everyone comes from somewhere: Systemic lupus erythematosus and Epstein-Barr virus induction of host interferon and humoral anti-Epstein-Barr nuclear antigen 1 immunity. Arthritis Rheum. 2010;62(6):1571–5.
Jog NR, Young KA, Munroe ME, Harmon MT, Guthridge JM, Kelly JA, et al. Association of Epstein-Barr virus serological reactivation with transitioning to systemic lupus erythematosus in at-risk individuals. Ann Rheum Dis. 2019;78(9):1235–41.
Lucchese G, Flöel A. Molecular mimicry between SARS-CoV-2 and respiratory pacemaker neurons. Autoimmun Rev. 2020;19(7):102556.
Marino Gammazza A, Légaré S, Lo Bosco G, Fucarino A, Angileri F, Conway de Macario E, et al. Human molecular chaperones share with SARS-CoV-2 antigenic epitopes potentially capable of eliciting autoimmunity against endothelial cells: Possible role of molecular mimicry in COVID-19. Cell Stress Chaperones. 2020;25(5):737–41.
Lucchese G, Flöel A. SARS-CoV-2 and Guillain-Barré syndrome: Molecular mimicry with human heat shock proteins as potential pathogenic mechanism. Cell Stress Chaperones. 2020;25(5):731–5.
Venkatakrishnan AJ, Kayal N, Anand P, Badley AD, Church GM, Soundararajan V. Benchmarking evolutionary tinkering underlying human-viral molecular mimicry shows multiple host pulmonary-arterial peptides mimicked by SARS-CoV-2. Cell Death Discov. 2020;6(1):96.
Kanduc D. From anti-SARS-CoV-2 immune responses to COVID-19 via molecular mimicry. Antibodies. 2020;9(3):33–8.
Muller S, Radic M. Oxidation and mitochondrial origin of NET DNA in the pathogenesis of lupus. Nat Med. 2016;22(2):126–7.
Apel F, Zychlinsky A, Kenny EF. The role of neutrophil extracellular traps in rheumatic diseases. Nat Rev Rheumatol. 2018;14(8):467–75.
Wigerblad G, Kaplan MJ. NETs spread ever wider in rheumatic diseases. Nat Rev Rheumatol. 2020;16(2):73–4.
Ali RA, Gandhi AA, Meng H, Yalavarthi S, Vreede AP, Estes SK, et al. Adenosine receptor agonism protects against NETosis and thrombosis in antiphospholipid syndrome. Nat Commun. 2019;10(1):1916.
Skendros P, Mitsios A, Chrysanthopoulou A, Mastellos DC, Metallidis S, Rafailidis P, et al. Complement and tissue factor-enriched neutrophil extracellular traps are key drivers in COVID-19 immunothrombosis. J Clin Invest. 2020;130(11):6151–7.
McDonald B, Davis RP, Kim SJ, Tse M, Esmon CT, Kolaczkowska E, et al. Platelets and neutrophil extracellular traps collaborate to promote intravascular coagulation during sepsis in mice. Blood. 2017;129(10):1357–67.
Englert H, Rangaswamy C, Deppermann C, Sperhake JP, Krisp C, Schreier D, et al. Defective NET clearance contributes to sustained FXII activation in COVID-19-associated pulmonary thrombo-inflammation. EBioMedicine. 2021;67:103382.
Noubouossie DF, Reeves BN, Strahl BD, Key NS. Neutrophils: Back in the thrombosis spotlight. Blood. 2019;133(20):2186–97.
Bonaventura A, Vecchié A, Dagna L, Martinod K, Dixon DL, Van Tassell BW, et al. Endothelial dysfunction and immunothrombosis as key pathogenic mechanisms in COVID-19. Nat Rev Immunol. 2021;21(5):319–29.
Lee JS, Shin EC. The type I interferon response in COVID-19: Implications for treatment. Nat Rev Immunol. 2020;20(10):585–6.
Northcott M, Jones S, Koelmeyer R, Bonin J, Vincent F, Kandane-Rathnayake R, et al. Type 1 interferon status in systemic lupus erythematosus: A longitudinal analysis. Lupus Sci Med. 2022;9(1):e000625.
da Silva RP, Gonçalves JIB, Zanin RF, Schuch FB, de Souza APD. Circulating type I interferon levels and COVID-19 severity: A systematic review and meta-analysis. Front Immunol. 2021;12:657363.
Crow MK. Type I interferon in the pathogenesis of lupus. J Immunol. 2014;192(12):5459–68.
Merrill JT, Erkan D, Winakur J, James JA. Emerging evidence of a COVID-19 thrombotic syndrome has treatment implications. Nat Rev Rheumatol. 2020;16(10):581–9.
Mahajan A, Herrmann M, Muñoz LE. Clearance deficiency and cell death pathways: A model for the pathogenesis of SLE. Front Immunol. 2016;7:35.
Setiati S, Harimurti K, Safitri ED, Ranakusuma RW, Saldi SRF, Azwar MK, et al. Risk factors and laboratory test results associated with severe illness and mortality in COVID-19 patients: A systematic review. Acta Med Indones. 2020;52(3):227–45.
Ziadi A, Hachimi A, Admou B, Hazime R, Brahim I, Douirek F, et al. Lymphopenia in critically ill COVID‐19 patients: A predictor factor of severity and mortality. Int J Lab Hematol. 2021;43(1):38–43.
Wang J, Li Q, Yin Y, Zhang Y, Cao Y, Lin X, et al. Excessive neutrophils and neutrophil extracellular traps in COVID-19. Front Immunol. 2021;43(1):e38–46.
Satış H, Özger HS, Yıldız PA, Hızel K, Gulbahar Ö, Erbaş G, et al. Prognostic value of interleukin-18 and its association with other inflammatory markers and disease severity in COVID-19. Cytokine. 2021;137:155302.
Vassallo M, Manni S, Pini P, Blanchouin E, Ticchioni M, Seitz-Polski B, et al. Patients with Covid-19 exhibit different immunological profiles according to their clinical presentation. Int J Infect Dis. 2020;101:174–9.
Sun Y, Dong Y, Wang L, Xie H, Li B, Chang C, et al. Characteristics and prognostic factors of disease severity in patients with COVID-19: The Beijing experience. J Autoimmun. 2020;112:102473.
Dotan A, Muller S, Kanduc D, David P, Halpert G, Shoenfeld Y. The SARS-CoV-2 as an instrumental trigger of autoimmunity. Autoimmun Rev. 2021;20(4):102792.
Liu Y, Sawalha AH, Lu Q. COVID-19 and autoimmune diseases. Curr Opin Rheumatol. 2021;33(2):155–62.
Wang JY, Roehrl MW, Roehrl VB, Roehrl MH. A master autoantigen-ome links alternative splicing, female predilection, and COVID-19 to autoimmune diseases. J Transl Autoimmun. 2022;5:100147.
Wang JY, Zhang W, Roehrl VB, Roehrl MW, Roehrl MH. An autoantigen-ome from HS-sultan B-lymphoblasts offers a molecular map for investigating autoimmune sequelae of COVID-19. bioRxiv. 2021;3(12):24–33.
Khandpur R, Carmona-Rivera C, Vivekanandan-Giri A, Gizinski A, Yalavarthi S, Knight JS, et al. NETs are a source of citrullinated autoantigens and stimulate inflammatory responses in rheumatoid arthritis. Sci Transl Med. 2013;5(178):178ra40.
Zuo Y, Yalavarthi S, Navaz SA, Hoy CK, Harbaugh A, Gockman K, et al. Autoantibodies stabilize neutrophil extracellular traps in COVID-19. JCI Insight. 2021;6(15):12–23.
Torres-Ruiz J, Absalón-Aguilar A, Nuñez-Aguirre M, Pérez-Fragoso A, Carrillo-Vázquez DA, Maravillas-Montero JL, et al. Neutrophil extracellular traps contribute to COVID-19 hyperinflammation and humoral autoimmunity. Cells. 2021;10(10):32–44.
Richter AG, Shields AM, Karim A, Birch D, Faustini SE, Steadman L, et al. Establishing the prevalence of common tissue-specific autoantibodies following severe acute respiratory syndrome coronavirus 2 infection. Clin Exp Immunol. 2021;205(2):99–105.
Jin M, Tong Q. Rhabdomyolysis as potential late complication associated with COVID-19. Emerg Infect Dis. 2020;26(7):1618.
Saud A, Naveen R, Aggarwal R, Gupta L. COVID-19 and myositis: What we know so far. Curr Rheumatol Rep. 2021;23(8):1–16.
Gokhale Y, Patankar A, Holla U, Shilke M, Kalekar L, Karnik ND, et al. Dermatomyositis during COVID-19 pandemic (a case series): Is there a cause effect relationship? J Assoc Physicians India. 2020;68(11):20–4.
Pascolini S, Granito A, Muratori L, Lenzi M, Muratori P. Coronavirus disease associated immune thrombocytopenia: Causation or correlation? J Microbiol Immunol Infect. 2021;54(3):531.
Rahi MS, Jindal V, Reyes S-P, Gunasekaran K, Gupta R, Jaiyesimi I. Hematologic disorders associated with COVID-19: A review. Ann Hematol. 2021;100(2):309–20.
Nagu P, Parashar A, Behl T, Mehta V. CNS implications of COVID-19: A comprehensive review. Rev Neurosci. 2021;32(2):219–34.
Scappaticcio L, Pitoia F, Esposito K, Piccardo A, Trimboli P. Impact of COVID-19 on the thyroid gland: An update. Rev Endocr Metab Disord. 2021;22(4):803–15.
Finsterer J, Scorza FA. Infectious and immune-mediated central nervous system disease in 48 COVID-19 patients. J Clin Neurosci. 2021;90:140–3.
Ismail II, Al-Hashel J, Alroughani R, Ahmed SF. A case report of multiple sclerosis after COVID-19 infection: Causality or coincidence? Neuroimmunol Rep. 2021;1:100008.
Ismail II, Al-Hashel J, Alroughani R, Ahmed SF. Association of multiple sclerosis and COVID-19 infection: A case report. MultScler Relat Disord. 2022;14(3):59–63.
Rajdev K, Victor N, Buckholtz ES, Hariharan P, Saeed MA, Hershberger DM, et al. A case of Guillain-Barré syndrome associated with COVID-19. J Investig Med High Impact Case Rep. 2020;8:2324709620961198.
Rajdev K, Victor N, Buckholtz ES, Hariharan P, Saeed MA, Hershberger DM, et al. A case of Guillain-Barré syndrome associated with COVID-19. J Investig Med High Impact Case Rep. 2020;8:2324709620961198.
Palao M, Fernández-Díaz E, Gracia-Gil J, Romero-Sánchez C, Díaz-Maroto I, Segura T. Multiple sclerosis following SARS-CoV-2 infection. Mult Scler Relat Disord. 2020;45:102377.
Zhou S, Jones-Lopez EC, Soneji DJ, Azevedo CJ, Patel VR. Myelin oligodendrocyte glycoprotein antibody–associated optic neuritis and myelitis in COVID-19.J Neuro-ophthalmol. 2020;12(2):23–9.
Knack RS, Hanada T, Knack RS, Mayr K. Hashimoto’s thyroiditis following SARS-CoV-2 infection. BMJ Case Rep CP. 2021;14(8):e244909.
Mateu-Salat M, Urgell E, Chico A. SARS-COV-2 as a trigger for autoimmune disease: Report of two cases of Graves’ disease after COVID-19. J Endocrinol Invest. 2020;43(10):1527–8.
Trovato CM, Montuori M, Pietropaoli N, Oliva S. COVID-19 and celiac disease: A pathogenetic hypothesis for a celiac outbreak. Int J Clin Pract. 2021;75(9):e14452.
Guerra I, Algaba A, Jiménez L, Mar Aller M, Garza D, Bonillo D, et al. Incidence, clinical characteristics, and evolution of SARS-CoV-2 infection in patients with inflammatory bowel disease: A single-center study in Madrid, Spain. Inflamm Bowel Dis. 2021;27(1):25–33.
Bril F, Al Diffalha S, Dean M, Fettig DM. Autoimmune hepatitis developing after coronavirus disease 2019 (COVID-19) vaccine: Causality or casualty? J Hepatol. 2021;75(1):222–4.
Bartoli A, Gitto S, Sighinolfi P, Cursaro C, Andreone P. Primary biliary cholangitis associated with SARS-CoV-2 infection. J Hepatol. 2021;74(5):1245–6.
Masuko K. Will the COVID-19 pandemic trigger future occurrence of autoimmunity like Sjögren’s syndrome? Int J Rheum Dis. 2021;24(7):963–5.
Toor SM, Saleh R, Sasidharan Nair V, Taha RZ, Elkord E. T-cell responses and therapies against SARS-CoV-2 infection. Immunology. 2021;162(1):30–43.
Koga T, Ichinose K, Kawakami A, Tsokos GC. Current insights and future prospects for targeting IL-17 to treat patients with systemic lupus erythematosus. Front Immunol. 2020;11:624971.
Bhattacharjee S, Banerjee M. Immune thrombocytopenia secondary to COVID-19: A systematic review. SN Compr Clin Med. 2020;2(11):2048–58.
Fathi N, Rezaei N. Lymphopenia in COVID-19: Therapeutic opportunities. Cell Biol Int. 2020;44(9):1792–7.
Cavalcante-Silva LHA, Carvalho DCM, de Almeida Lima É, Galvao JG, da Silva JSdF, de Sales-Neto JM, et al. Neutrophils and COVID-19: The road so far. Int Immunopharmacol. 2021;90:107233.
Iba T, Levy JH, Levi M, Thachil J. Coagulopathy in COVID-19. J Thromb Haemost. 2020;18(9):2103–9.
Muhammed L, Baheerathan A, Cao M, Leite MI, Viegas S. MuSK antibody-associated myasthenia gravis with SARS-CoV-2 infection: A case report. Ann Intern Med. 2021;174(6):872–3.
Kabeerdoss J, Danda D. Understanding immunopathological fallout of human coronavirus infections including COVID-19: Will they cross the path of rheumatologists? Int J Rheum Dis. 2020;23(8):998–1008.
Ladani AP, Loganathan M, Danve A. Managing rheumatic diseases during COVID-19. Clin Rheumatol. 2020;39(11):3245–54.
Akiyama S, Hamdeh S, Micic D, Sakuraba A. Prevalence and clinical outcomes of COVID-19 in patients with autoimmune diseases: A systematic review and meta-analysis. Ann Rheum Dis. 2021;80(3):384–91.
Owlia S, Owlia MB. Glucocorticoids in COVID19; a friend not foe. J Res Med Sci. 2020;25:45.
Cabanillas F, Morales J, Conde JG, Bertrán-Pasarell J, Fernández R, Hernandez-Silva Y, et al. Single-arm, open-label phase 2 trial of preemptive methylprednisolone to avert progression to respiratory failure in high-risk patients with COVID-19. medRxiv. 2021;23(4):33–9.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Saeedi, N., Gohari, N.S.F., Ghalibaf, A.A.M. et al. COVID-19 infection: a possible induction factor for development of autoimmune diseases?. Immunol Res 71, 547–553 (2023). https://doi.org/10.1007/s12026-023-09371-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12026-023-09371-7