Abstract
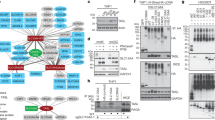
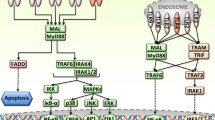
Stimulation of Toll-like receptor (TLR) 4 leads to the activation of both MyD88-dependent and MyD88-independent pathways through the recruitment of adaptors TIRAP/MyD88 and TRIF/TRAM, respectively. However, the molecular basis of the TLR4 Toll/interleukin-1 receptor (TIR) domain in recruiting these downstream adaptors is still not entirely clear. Here, we identify three amino acid residues (714P in the BB loop, 696L in the αA helix and 721N in the αB sheet) conserved in all MyD88-recruited TLRs, but not the TLR3 TIR domain, as being critical for TLR4 responsiveness to LPS. These results were based on the substitution of each residue with a residue of the opposite type (hydrophilic/hydrophobic). However, the responsiveness of the TLR4 mutants to LPS was only partially decreased when each residue was replaced with a residue having the same hydrophilicity/hydrophobicity. This result is likely associated with an alteration in the BB-loop conformation of each TLR4 mutant and its ability to recruit the downstream adaptor TRAM. Thus, we identified three amino acids essential for TLR4 signaling, and their replacement with a residue of the same or opposite hydrophilicity/hydrophobicity greatly affected TLR4 signaling. This study furthers our understanding of the molecular mechanism by which the TLR4 TIR domain modulates TLR4 signaling and also provides new insight for the design of antisepsis therapy.






Similar content being viewed by others
References
Kawai T, Akira S. The role of pattern-recognition receptors in innate immunity: update on Toll-like receptors. Nat Immunol. 2010;11(5):373–84.
O’Neill LA, Golenbock D, Bowie AG. The history of Toll-like receptors—redefining innate immunity. Nat Rev Immunol. 2013;13(6):453–60.
Dunzendorfer S, Lee HK, Soldau K, Tobias PS. TLR4 is the signaling but not the lipopolysaccharide uptake receptor. J Immunol. 2004;173(2):1166–70.
Poltorak A, He X, Smirnova I, Liu MY, Van Huffel C, Du X, et al. Defective LPS signaling in C3H/HeJ and C57BL/10ScCr mice: mutations in Tlr4 gene. Science. 1998;282(5396):2085–8.
Arbour NC, Lorenz E, Schutte BC, Zabner J, Kline JN, Jones M, et al. TLR4 mutations are associated with endotoxin hyporesponsiveness in humans. Nat Genet. 2000;25(2):187–91.
Roger T, Froidevaux C, Le Roy D, Reymond MK, Chanson AL, Mauri D, et al. Protection from lethal gram-negative bacterial sepsis by targeting Toll-like receptor 4. Proc Natl Acad Sci USA. 2009;106(7):2348–52.
Dunne A, O’Neill LA. The interleukin-1 receptor/Toll-like receptor superfamily: signal transduction during inflammation and host defense. Sci STKE. 2003;2003(171):re3.
O’Neill LA, Bowie AG. The family of five: TIR-domain-containing adaptors in Toll-like receptor signalling. Nat Rev Immunol. 2007;7(5):353–64.
Zughaier SM, Zimmer SM, Datta A, Carlson RW, Stephens DS. Differential induction of the Toll-like receptor 4-MyD88-dependent and -independent signaling pathways by endotoxins. Infect Immun. 2005;73(5):2940–50.
Xu Y, Tao X, Shen B, Horng T, Medzhitov R, Manley JL, et al. Structural basis for signal transduction by the Toll/interleukin-1 receptor domains. Nature. 2000;408(6808):111–5.
Dunne A, Ejdeback M, Ludidi PL, O’Neill LA, Gay NJ. Structural complementarity of Toll/interleukin-1 receptor domains in Toll-like receptors and the adaptors Mal and MyD88. J Biol Chem. 2003;278(42):41443–51.
Toshchakov VY, Fenton MJ, Vogel SN. Cutting edge: differential inhibition of TLR signaling pathways by cell-permeable peptides representing BB loops of TLRs. J Immunol. 2007;178(5):2655–60.
Jiang Z, Georgel P, Li C, Choe J, Crozat K, Rutschmann S, et al. Details of Toll-like receptor: adapter interaction revealed by germ-line mutagenesis. Proc Natl Acad Sci USA. 2006;103(29):10961–6.
Sali A, Overington JP. Derivation of rules for comparative protein modeling from a database of protein structure alignments. Protein Sci. 1994;3(9):1582–96.
Akira S, Takeda K. Toll-like receptor signalling. Nat Rev Immunol. 2004;4(7):499–511.
Yanagimoto S, Tatsuno K, Okugawa S, Kitazawa T, Tsukada K, Koike K, et al. A single amino acid of Toll-like receptor 4 that is pivotal for its signal transduction and subcellular localization. J Biol Chem. 2009;284(6):3513–20.
Ronni T, Agarwal V, Haykinson M, Haberland ME, Cheng G, Smale ST. Common interaction surfaces of the Toll-like receptor 4 cytoplasmic domain stimulate multiple nuclear targets. Mol Cell Biol. 2003;23(7):2543–55.
Qiu Y, Ding Y, Zou L, Tan Z, Liu T, Fu X, et al. Divergent roles of amino acid residues inside and outside the BB loop affect human Toll-like receptor (TLR)2/2, TLR2/1 and TLR2/6 responsiveness. PLoS One. 2013;8(4):e61508.
Verstak B, Arnot CJ, Gay NJ. An alanine-to-proline mutation in the BB-loop of TLR3 Toll/IL-1R domain switches signalling adaptor specificity from TRIF to MyD88. J Immunol. 2013;191(12):6101–9.
Li C, Zienkiewicz J, Hawiger J. Interactive sites in the MyD88 Toll/interleukin (IL) 1 receptor domain responsible for coupling to the IL1beta signaling pathway. J Biol Chem. 2005;280(28):26152–9.
Randow F, Seed B. Endoplasmic reticulum chaperone gp96 is required for innate immunity but not cell viability. Nat Cell Biol. 2001;3(10):891–6.
Yang Y, Liu B, Dai J, Srivastava PK, Zammit DJ, Lefrancois L, et al. Heat shock protein gp96 is a master chaperone for Toll-like receptors and is important in the innate function of macrophages. Immunity. 2007;26(2):215–26.
Wakabayashi Y, Kobayashi M, Akashi-Takamura S, Tanimura N, Konno K, Takahashi K, et al. A protein associated with Toll-like receptor 4 (PRAT4A) regulates cell surface expression of TLR4. J Immunol. 2006;177(3):1772–9.
Shen H, Tesar BM, Walker WE, Goldstein DR. Dual signaling of MyD88 and TRIF is critical for maximal TLR4-induced dendritic cell maturation. J Immunol. 2008;181(3):1849–58.
Orr MT, Duthie MS, Windish HP, Lucas EA, Guderian JA, Hudson TE, et al. MyD88 and TRIF synergistic interaction is required for TH1-cell polarization with a synthetic TLR4 agonist adjuvant. Eur J Immunol. 2013;43(9):2398–408.
Janssen E, Ozcan E, Liadaki K, Jabara HH, Manis J, Ullas S, et al. TRIF signaling is essential for TLR4-driven IgE class switching. J Immunol. 2014;192(6):2651–8.
Gaddis DE, Michalek SM, Katz J. TLR4 signaling via MyD88 and TRIF differentially shape the CD4+ T cell response to Porphyromonas gingivalis hemagglutinin B. J Immunol. 2011;186(10):5772–83.
Brieger A, Rink L, Haase H. Differential regulation of TLR-dependent MyD88 and TRIF signaling pathways by free zinc ions. J Immunol. 2013;191(4):1808–17.
Kagan JC, Su T, Horng T, Chow A, Akira S, Medzhitov R. TRAM couples endocytosis of Toll-like receptor 4 to the induction of interferon-beta. Nat Immunol. 2008;9(4):361–8.
Kissner TL, Moisan L, Mann E, Alam S, Ruthel G, Ulrich RG, et al. A small molecule that mimics the BB-loop in the Toll interleukin-1 (IL-1) receptor domain of MyD88 attenuates staphylococcal enterotoxin B-induced pro-inflammatory cytokine production and toxicity in mice. J Biol Chem. 2011;286(36):31385–96.
Couture LA, Piao W, Ru LW, Vogel SN, Toshchakov VY. Targeting Toll-like receptor (TLR) signaling by Toll/interleukin-1 receptor (TIR) domain-containing adapter protein/MyD88 adapter-like (TIRAP/Mal)-derived decoy peptides. J Biol Chem. 2012;287(29):24641–8.
Avbelj M, Horvat S, Jerala R. The role of intermediary domain of MyD88 in cell activation and therapeutic inhibition of TLRs. J Immunol. 2011;187(5):2394–404.
Acknowledgments
This work is supported by the Natural Science Foundation of China for WX (81000747) and the Natural Science Foundation of Military for WX (CWS12J093). We are grateful to Professor Kensuke Miyake for providing us with plasmid pEF-BOS-TLR4 and pEF-BOS-MD-2 and Dr. Xin Du for pFLAG-CMV1-CD14.
Conflict of interest
The authors declare that there is no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Ding, Y., Qiu, Y., Zou, L. et al. Three conserved MyD88-recruiting TLR residues exert different effects on the human TLR4 signaling pathway. Immunol Res 62, 213–221 (2015). https://doi.org/10.1007/s12026-015-8652-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12026-015-8652-2