Abstract
Purpose
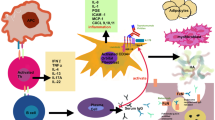
Thyroid eye disease (TED) is the foremost extrathyroidal manifestation of Graves’ disease (GD). Currently, available treatments do not entirely prevent the long-term consequences of TED and have distinct disadvantages. Therefore, this systematic review explored available evidence regarding the efficacy of statins in preventing and treating TED.
Methods
Relevant studies investigating statin usage in patients with GD or TED were identified by searching Medline (Pubmed and Ovid), Scopus, Web of Science, ProQuest, and Cochrane Library databases (from the database inception to September 2023). The review was done according to the PRISMA statement. Web searching was done independently by two investigators. Two researchers independently extracted the data, and any disagreement was adjudicated by consensus. Based on the study design, the studies’ quality appraisal was done using the Newcastle-Ottawa Scale (NOS) and Version 2 of the Cochrane risk-of-bias tool (RoB2).
Results
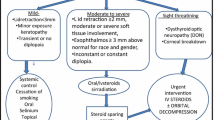
The literature search identified 145 publications, of which four met the inclusion criteria (Three retrospective cohort studies and one randomized clinical trial) and were reviewed in full text. The two retrospective cohort studies demonstrated the beneficial effects of statins on TED in newly diagnosed GD Stein et al. showed that statins, regardless of the type, prevent or delay TED (HR: 0.74 (0.65–0.84)), especially in men or treatment duration of more than one year. Nilsson et al. fascinatingly revealed that at least 60 days of statin usage in the preceding year could decrease the risk of TED development by around 40%. One RCT showed a higher treatment response for active moderate-to-severe TED in patients with hypercholesterolemia who took atorvastatin 20 mg in addition to ivGC for 24 weeks without any increase in serious side effects. The retrospective study revealed that the need for reconstructive surgery was reduced in patients with severe TED who received statin therapy.
Conclusion
Statin therapy could be a potential adjunctive modality for preventing and treating TED.
Trial registration
PROSPERO registration number: CRD42022315522.


Similar content being viewed by others
References
G.J. Kahaly et al. Prospective trial of functional thyrotropin receptor antibodies in Graves disease. J. Clin. Endocrinol. Metab. 105(4), e1006–e1014 (2020)
J.P. Brito et al. Antithyroid drugs-the most common treatment for Graves’ disease in the United States: A nationwide population-based study. Thyroid 26(8), 1144–1145 (2016)
Y.H. Chin et al. Prevalence of thyroid eye disease in Graves’ disease: A meta‐analysis and systematic review. Clin. Endocrinol. 93(4), 363–374 (2020)
S. Ippolito et al. Change in newly diagnosed Graves’ disease phenotype between the twentieth and the twenty-first centuries: meta-analysis and meta-regression. J. Endocrinol. Invest 44(8), 1707–1718 (2021)
M. Tanda et al. Prevalence and natural history of Graves’ orbitopathy in a large series of patients with newly diagnosed graves’ hyperthyroidism seen at a single center. J. Clin. Endocrinol. Metab. 98(4), 1443–1449 (2013)
A. Naselli et al. Evidence that baseline levels of low-density lipoproteins cholesterol affect the clinical response of Graves’ ophthalmopathy to parenteral corticosteroids. Front. Endocrinol. 11, 609895 (2020)
G. Lanzolla et al. Cholesterol serum levels and use of statins in Graves’ orbitopathy: A new starting point for the therapy. Front Endocrinol. (Lausanne) 10, 933 (2019)
L. Bartalena, A. Pinchera, C. Marcocci, Management of Graves’ ophthalmopathy: reality and perspectives. Endocr. Rev. 21(2), 168–199 (2000)
W.M. Wiersinga, Quality of life in Graves’ ophthalmopathy. Best. Pract. Res. Clin. Endocrinol. Metab. 26(3), 359–370 (2012)
K.A. Ponto et al. Public health relevance of Graves’ orbitopathy. J. Clin. Endocrinol. Metab. 98(1), 145–152 (2013)
L. Bartalena et al. The 2021 European Group on Graves’ orbitopathy (EUGOGO) clinical practice guidelines for the medical management of Graves’ orbitopathy. Eur. J. Endocrinol. 185(4), G43–G67 (2021)
L. Bartalena et al. Relation between therapy for hyperthyroidism and the course of Graves’ ophthalmopathy. N. Engl. J. Med. 338(2), 73–78 (1998)
F. Traisk et al. Thyroid-associated ophthalmopathy after treatment for Graves’ hyperthyroidism with antithyroid drugs or iodine-131. J. Clin. Endocrinol. Metab. 94(10), 3700–3707 (2009)
S.H. Acharya et al. Radioiodine therapy (RAI) for Graves’ disease (GD) and the effect on ophthalmopathy: a systematic review. Clin. Endocrinol. 69(6), 943–950 (2008)
D. Villagelin et al. Outcomes in relapsed Graves’ disease patients following radioiodine or prolonged low dose of methimazole treatment. Thyroid 25(12), 1282–1290 (2015)
L. Tallstedt et al. Occurrence of ophthalmopathy after treatment for Graves’ hyperthyroidism. N. Engl. J. Med. 326(26), 1733–1738 (1992)
Marcocci et al. The course of Graves’ ophthalmopathy is not influenced by near total thyroidectomy: a case‐control study. Clin. Endocrinol. 51(4), 503–508 (1999)
R. Malboosbaf, F. Azizi, Long-term treatment with antithyroid drugs: Efficacy and safety. Int. J. Endocrinol. Metab. 18(Suppl), e101487 (2020)
F. Azizi et al. Long-term follow-up of Graves’ orbitopathy after treatment with short-term or long-term methimazole or radioactive iodine. Endocrine Pract. 29(4), 240–246 (2023).
F. Azizi, R. Malboosbaf, Safety of long-term antithyroid drug treatment? A systematic review. J. Endocrinol. Invest 42(11), 1273–1283 (2019)
L. Bartalena, M.L. Tanda, Current concepts regarding Graves’ orbitopathy. J. Intern. Med. 292(5), 692–716 (2022)
A.L. Reynolds, M.A. Del Monte, S.M. Archer, The effect of oral statin therapy on strabismus in patients with thyroid eye disease. J. Aapos 22(5), 340–343 (2018)
M. Ludgate, G. Baker, Unlocking the immunological mechanisms of orbital inflammation in thyroid eye disease. Clin. Exp. Immunol. 127(2), 193–198 (2002)
V. Croons et al. Effect of statins on the viability of macrophages and smooth muscle cells. J. Cardiovascular Pharmacol. 55(3), 269–275 (2010)
A. Nilsson, K. Tsoumani, T. Planck, Statins decrease the risk of orbitopathy in newly diagnosed patients with Graves disease. J. Clin. Endocrinol. Metab. 106(5), 1325–1332 (2021)
Y.-H. Wei et al. Simvastatin and ROCK inhibitor Y-27632 inhibit myofibroblast differentiation of Graves’ ophthalmopathy-derived orbital fibroblasts via RhoA-mediated ERK and p38 signaling pathways. Front. Endocrinol. 11, 607968 (2021)
J.S. Yoon, D.O. Kikkawa, Thyroid eye disease: From pathogenesis to targeted therapies. Taiwan J. Ophthalmol. 12(1), 3–11 (2022)
R. Bahn, Current insights into the pathogenesis of Graves’ ophthalmopathy. Horm. Metab. Res. 47(10), 773–778 (2015)
G. Lanzolla et al. Statins for Graves’ orbitopathy (STAGO): a phase 2, open-label, adaptive, single centre, randomised clinical trial. Lancet Diabetes Endocrinol. 9(11), 733–742 (2021)
A. Endo, A historical perspective on the discovery of statins. Proc. Jpn Acad. Ser. B Phys. Biol. Sci. 86(5), 484–493 (2010)
T. Cai et al. Associations between statins and adverse events in primary prevention of cardiovascular disease: systematic review with pairwise, network, and dose-response meta-analyses. BMJ 374, n1537 (2021)
E. Myasoedova et al. Effect of statin use on the risk of rheumatoid arthritis: a systematic review and meta-analysis. in Seminars in arthritis and rheumatism. 2020. Elsevier
P. Morfino et al. Treatment of cardiac fibrosis: from neuro-hormonal inhibitors to CAR-T cell therapy. Heart Fail. Rev. 28(2), 555–569 (2023)
E.M. Lambert et al. Statins: cause of fibrosis or the opposite? Effect of cardiovascular drugs in idiopathic pulmonary fibrosis. Respiratory Med. 176, 106259 (2021)
R. Gilbert et al. Statins as anti-inflammatory agents: A potential therapeutic role in sight-threatening non-infectious uveitis. Porto Biomed. J. 2(2), 33–39 (2017)
T. Kandelouei et al. Effect of statins on serum level of hs-CRP and CRP in patients with cardiovascular diseases: A systematic review and meta-analysis of randomized controlled trials. Mediat. Inflamm. 2022, 8732360 (2022)
M.C. Proute et al. The effect of Statin therapy on inflammatory biomarkers: A systematic review. Cureus 13(9), e18273 (2021)
A. Liberati et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. Ann. Intern. Med. 151(4), W-65–W-94 (2009)
G. Wells et al. Newcastle-Ottawa quality assessment scale cohort studies. University of Ottawa, 2014.
J.A. Sterne et al. RoB 2: a revised tool for assessing risk of bias in randomised trials. bmj 366, 4898 (2019)
J.D. Stein et al. Risk factors for developing thyroid-associated ophthalmopathy among individuals with Graves disease. JAMA Ophthalmol. 133(3), 290–296 (2015)
I. Campi, L. Fugazzola, How can we prevent disease relapse in Graves’ orbitopathy after immunosuppressive treatment? Expert Rev. Endocrinol. Metab. 17(4), 269–274 (2022)
G. Lanzolla et al. Relationship between serum cholesterol and Graves’ orbitopathy (GO): a confirmatory study. J. Endocrinol. Investig. 41(12), 1417–1423 (2018)
E. Sabini et al. High serum cholesterol is a novel risk factor for Graves’ orbitopathy: Results of a cross-sectional study. Thyroid 28(3), 386–394 (2018)
M. Bifulco, E. Ciaglia, Statin reduces orbitopathy risk in patients with Graves’ disease by modulating apoptosis and autophagy activities. Endocrine 53(3), 649–650 (2016)
T. Park et al. StAtin Cost Effectiveness: A systematic review including the recent literature. Value Health 20(9), A616 (2017)
J. Beltowski, G. Wojcicka, A. Jamroz-Wisniewska, Adverse effects of statins-mechanisms and consequences. Curr. Drug Saf. 4(3), 209–228 (2009)
E. Sabini et al. Statins are not a risk factor for liver damage associated with intravenous glucocorticoid pulse therapy for Graves’ orbitopathy. J. Endocrinol. Invest 39(11), 1323–1327 (2016)
Author contributions
RM and ZM conceptualized the research question, contributed to the literature search, and full text screening assessed the quality, extracted the data, and wrote the review. MK and FA participated in the review and editing of the manuscript. ZE contributed to the literature search. All authors contributed to the article and approved the submitted version.
Funding
The study was supported by the Iran University of Medical Sciences (Grant number:23307).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Malboosbaf, R., Maghsoomi, Z., Emami, Z. et al. Statins and thyroid eye disease (TED): a systematic review. Endocrine (2024). https://doi.org/10.1007/s12020-023-03680-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12020-023-03680-5